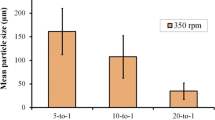
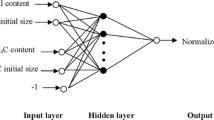
Abstract
Milling by mechanical means is vital unit operation in pharmaceutical processing which can be used for controlling particle size reduction. This approach can be also used for mechanochemical synthesis of pharmaceutical cocrystals. However, controlling the particle size of cocrystal during the milling process is challenging due to complexity of the process and different mechanisms involved. In this study, artificial neural network (ANN) approach was performed to predict the size of particles during mechanochemical synthesis of pharmaceutical cocrystals in ball milling operation. Theophylline was used as active pharmaceutical ingredient (API) and 4-aminobenzoic acid as co-former in the experiments. Different types of excipients including hydroxypropylmethylcellulose (HPMC), lactose, microcrystalline cellulose (MCC) polyvinylpyrrolidone (PVP) and polyethylene glycol (PEG) were used to see the effect of excipients on the cocrystals particle size. ANN was developed considering excipient type, excipient percentage, jar size, milling time as inputs, while median particle size (d50) was considered as the response, and representative of particle size. Two hidden layers were considered in developing ANN, and a combination of linear and nonlinear functions was used as transfer function. ANN was trained and validated with measured data, and R2 greater than 0.99 was obtained for the training and validation, with RMSE values of 1.06 and 3.89 for training and validation, respectively. The results were used to provide a design space for understanding the cocrystal particle size variation during ball milling process. It was indicated that the largest particles were formed using PEG as excipient, and increase in particle size over the milling time. The latter was attributed to the electrostatic attraction forces between the particles, thus aggregation and agglomeration of particles as the milling time exceeds a threshold. Furthermore, larger particles were obtained with the larger jar size. It turned out that faster cocrystallization rate occurred after only 5 min of milling in 25 mL jar compared to 25 min in 10 mL milling jar. The developed methodology has been shown to be robust and can be used as predictive tool for designing pharmaceutical cocrystallization using ball milling, and controlling the particle size during size reduction process.















Similar content being viewed by others
References
Shaikh R et al (2018) Pharmaceutical cocrystal drug products: an outlook on product development. Trends Pharmacol Sci 39(12):1033–1048
Kalepu S, Nekkanti V (2015) Insoluble drug delivery strategies: review of recent advances and business prospects. Acta Pharmaceutica Sin B 5(5):442–453
Karimi-Jafari M et al (2018) Creating cocrystals: a review of pharmaceutical cocrystal preparation routes and applications. Cryst Growth Des 18(10):6370–6387
Pishnamazi M et al (2019) Application of lignin in controlled release: development of predictive model based on artificial neural network for API release. Cellulose 26(10):6165–6178
Van den Mooter G (2012) The use of amorphous solid dispersions: a formulation strategy to overcome poor solubility and dissolution rate. Drug Discov Today Technol 9(2):e79–e85
Ziaee A et al (2017) Spray drying ternary amorphous solid dispersions of ibuprofen—an investigation into critical formulation and processing parameters. Eur J Pharm Biopharm 120:43–51
Korde S et al (2018) Continuous manufacturing of cocrystals using solid state shear milling technology. Cryst Growth Des 18(4):2297–2304
Crawford DE et al (2017) Organic synthesis by Twin Screw Extrusion (TSE): continuous, scalable and solvent-free. Green Chem 19(6):1507–1518
Shaikh R et al (2018) Chapter 2—The development of a pharmaceutical oral solid dosage forms. In: Singh R, Yuan Z (eds) Computer aided chemical engineering, Elsevier. pp 27–65
Zhou QT et al (2010) Understanding the influence of powder flowability, fluidization and de-agglomeration characteristics on the aerosolization of pharmaceutical model powders. Eur J Pharm Sci 40(5):412–421
Zhou QT et al (2010) Improving aerosolization of drug powders by reducing powder intrinsic cohesion via a mechanical dry coating approach. Int J Pharm 394(1):50–59
Zhou Q et al (2011) Investigation of the extent of surface coating via mechanofusion with varying additive levels and the influences on bulk powder flow properties. Int J Pharm 413(1):36–43
Zhou Q et al (2013) Effect of surface coating with magnesium stearate via mechanical dry powder coating approach on the aerosol performance of micronized drug powders from dry powder inhalers. AAPS PharmSciTech 14(1):38–44
Lakio S et al (2015) Optimizing aerosolization of a high-dose L-arginine powder for pulmonary delivery. Asian J Pharm Sci 10(6):528–540
Yu Y et al (2015) Combined toxicity of amorphous silica nanoparticles and methylmercury to human lung epithelial cells. Ecotoxicol Environ Saf 112:144–152
Kumar Akkisetty P et al (2010) Population balance model-based hybrid neural network for a pharmaceutical milling process. J Pharm Innov 5(4):161–168
Ismail HY et al (2019) ANN-Kriging hybrid model for predicting carbon and inorganic phosphorus recovery in hydrothermal carbonization. Waste Manage 85:242–252
Ismail HY et al (2019) Developing ANN-Kriging hybrid model based on process parameters for prediction of mean residence time distribution in twin-screw wet granulation. Powder Technol 343:568–577
Shirazian S et al (2017) Artificial neural network modelling of continuous wet granulation using a twin-screw extruder. Int J Pharm 521(1):102–109
Aquino G et al (2020) Novel nonlinear hypothesis for the delta parallel robot modeling. IEEE Access 8:46324–46334
Rubio JD (2009) SOFMLS: online self-organizing fuzzy modified least-squares network. IEEE Trans Fuzzy Syst 17(6):1296–1309
Chiang HS, Chen MY, Huang YJ (2019) Wavelet-based EEG processing for epilepsy detection using fuzzy entropy and associative petri net. IEEE Access 7:103255–103262
Elias I et al (2020) Hessian with mini-batches for electrical demand prediction. Appl Sci Basel 10(6):2036
Meda-Campana JA (2018) On the estimation and control of nonlinear systems with parametric uncertainties and noisy outputs. IEEE Access 6:31968–31973
Ashfahani A et al (2020) DEVDAN: deep evolving denoising autoencoder. Neurocomputing 390:297–314
Senyigit E et al (2013) Heuristic-based neural networks for stochastic dynamic lot sizing problem. Appl Soft Comput 13(3):1332–1339
Senyigit E, Atici U (2013) Artificial neural network models for lot-sizing problem: a case study. Neural Comput Appl 22(6):1039–1047
Mohammed MA et al (2018) Neural network and multi-fractal dimension features for breast cancer classification from ultrasound images. Comput Electr Eng 70:871–882
Ibrahim Obaid O et al (2018) Evaluating the performance of machine learning techniques in the classification of wisconsin breast cancer. Comput Electr Eng 7:7
Abd Ghani MK et al (2020) Decision-level fusion scheme for nasopharyngeal carcinoma identification using machine learning techniques. Neural Comput Appl 32(3):625–638
Kazemi P et al (2016) Computational intelligence modeling of granule size distribution for oscillating milling. Powder Technol 301:1252–1258
Shaikh R, Walker GM, Croker DM (2019) Continuous, simultaneous cocrystallization and formulation of Theophylline and 4-Aminobenzoic acid pharmaceutical cocrystals using twin screw melt granulation. Eur J Pharm Sci 137:104981
Gotwalt CM (2011) JMP neural network methodology. SAS Institute, Cary
Khandai M, Chakraborty DS, NiranjanPatro C (2010) Preparation and evaluation of algino-sericin mucoadhesive microspheres: an approach for sustained drug delivery. J Adv Pharm Res 1:48–60
Baláž P et al (2013) Hallmarks of mechanochemistry: from nanoparticles to technology. Chem Soc Rev 42(18):7571–7637
Shaikh R, Shirazian S, Guerin S, Sheehan E, Thompson D, Walker GM, Croker DM (2021) Understanding solid-state processing of pharmaceutical cocrystals via milling: role of tablet excipients. Int J Pharm 601:120514
Kiselak J et al (2020) “SPOCU”: scaled polynomial constant unit activation function. In: Neural computing and applications
Acknowledgements
This research was made possible by the support of Synthesis and Solid-State Pharmaceutical Centre (SSPC), funded by Science Foundation Ireland (SFI). S.S. acknowledges the supports by the Government of the Russian Federation (Act 211, contract 02.A03.21.0011) and by the Ministry of Science and Higher Education of Russia (Grant FENU-2020-0019).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
1.1 The fitting results of ANN using Tansig transfer function
The training, testing and validation results for the Tansig transfer function are shown in Fig. 16.
Also, MSE (mean squared error) versus Epoch is plotted and represented in Fig. 17 for Tansig transfer function. The comparisons between the predicted and measured data in terms of d50 are indicated in Fig. 18 for the case of Tansig transfer function.
1.2 The fitting results of ANN using Logsig transfer function
The training, testing and validation results for the Logsig transfer function are shown in Fig. 19. Also, MSE versus Epoch is plotted and represented in Fig. 20 for Logsig transfer function. The comparisons between the predicted and measured data in terms of d50 are indicated in Fig. 21 for the case of Logsig transfer function.
1.3 The fitting results of ANN using Softmax transfer function
The training, testing and validation results for the Softmax transfer function are shown in Fig. 22. Also, MSE versus Epoch is plotted and represented in Fig. 23 for Softmax transfer function. The comparisons between the predicted and measured data in terms of d50 are indicated in Fig. 24 for the case of Softmax transfer function.
The summary of ANN models is listed in Table 3.
1.4 Simulation using SPOCU transfer function
Finally, SPOCU (scaled polynomial constant unit activation function) which was developed by Kiselak et al. [37] was used in the ANN simulations. It was indicated to perform satisfactorily on a variety of problems because of genuine normalization of the output of layers. The SPOCU activation function is written as [37]:
where \(\beta \in \left( {0,1} \right), \alpha , \gamma > 0\) and:
with \(r\left( x \right) = x^{3} \left( {x^{5} - 2x^{4} + 2} \right)\) and \(1 \le c < \infty\).
A code was written in MATLAB for implementing SPOCU transfer function which can be found in the Supplementary file. The results of ANN simulation for the test data are shown in Fig. 25. Also, the MSE results versus iterations are provided in Fig. 26. The SPOCU function was implemented on a network with the properties as reported in Table 4.
A comparison between various activation functions in terms of R2 for the testing/validation dataset is listed in Table 5.
Rights and permissions
About this article
Cite this article
Shaikh, R., Shirazian, S. & Walker, G.M. Application of artificial neural network for prediction of particle size in pharmaceutical cocrystallization using mechanochemical synthesis. Neural Comput & Applic 33, 12621–12640 (2021). https://doi.org/10.1007/s00521-021-05912-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00521-021-05912-z