Abstract
Introduction
Hand-foot syndrome (HFS) significantly impacts quality of life in cancer patients undergoing capecitabine treatment. This study assessed capecitabine-associated HFS prevalence, its impacts on chemotherapy treatment, and identified risk factors in multiracial Malaysian patients.
Methods
We included adult cancer patients receiving capecitabine at Sarawak General Hospital for at least two cycles from April 1, 2021 to June 30, 2022. HFS rates, time to HFS, and proportions of HFS-related treatment modifications were determined. Characteristics between patients with and without HFS were compared and multivariable logistic regression was used to identify risk factors for all-grade HFS and grade ≥2.
Results
Among 369 patients, 185 (50.1%) developed HFS, with 14.6% experiencing grade ≥2 and 21.6% (40/185) underwent treatment modifications. Risk factors for all-grade HFS include older age (OR 1.03 95%CI 1.01, 1.06), prior chemotherapy (OR 2.09 95%CI 1.22, 3.58), higher capecitabine dose (OR 2.96 95%CI 1.62, 5.38), prolonged treatment (OR 1.36 95%CI 1.21, 1.51), folic acid intake (OR 3.27 95%CI 1.45, 7.35) and lower neutrophil count (OR 0.77 95%CI 0.66, 0.89). For HFS grade ≥2, older age (OR 1.04 95%CI 1.01, 1.08), female sex (OR 2.10 95%CI 1.05, 4.18), Chinese race (OR 2.10 95%CI 1.06, 4.18), and higher capecitabine dose (OR 2.62 95%CI 1.28, 5.35) are significant risk factors. Use of calcium channel blockers were associated with reduced risks of all-grade HFS (OR 0.27, 95%CI 0.12, 0.60) and grade ≥2 (OR 0.21 95%CI 0.06, 0.78).
Conclusion
This study provides real-world data on capecitabine-induced HFS in Malaysian patients and identifies risk factors that may offer insights into its understanding and management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Capecitabine, as the prodrug of 5-fluorouracil (5-FU), is used either as monotherapy or in combination with other agents in adjuvant or palliative treatment for cancers. Although oral capecitabine has comparatively reduced gastrointestinal and bone marrow toxicities than intravenous fluorouracil [1], it is associated with a higher rate of hand-foot syndrome (HFS) [2].
HFS, also known as palmar-plantar erythrodysesthesia, is a well-documented and common adverse cutaneous reaction associated with chemotherapy. HFS is characterised by a range of symptoms including erythema, dysesthesia, pain, skin cracking, desquamation, and ulceration, primarily affecting the palms and soles of cancer patients undergoing capecitabine-based chemotherapy [3, 4]. HFS is typically graded using National Cancer Institute Common Terminology Criteria for Adverse Events (NCI-CTCAE, v5.0) grading scale [5]. While not posing a life-threatening risk, HFS can inflict pain and debilitation, disrupting patients' daily activities [6] and affecting their overall quality of life [7]. It may also lead to treatment interruptions and dose reductions [8, 9]. A study showed that 17-24% of metastatic colorectal cancer patients taking capecitabine experienced treatment modifications due to HFS [9].
A pooled analysis of clinical trials reported that up to 63% of the breast cancer patients taking capecitabine experienced HFS as an adverse event [10]. In a recent meta-analysis of clinical trials and observational studies, 68.3% of colorectal cancer patients experienced HFS after treatment with capecitabine alone, and 55.8% had HFS when treated with a combination of capecitabine and oxaliplatin. Moreover, 6% of them developed severe HFS. [11]. The study also highlighted disparities in HFS prevalence across different countries, with China having the highest rate at 55.1%, followed by Japan, Korea, and Italy [11]. However, to our knowledge, no study has reported the HFS prevalence in the Malaysian population.
There is a lack of real-world data on capecitabine-associated HFS and its impacts among the Malaysian patients. Sarawak General Hospital, as the largest hospital in Sarawak region, Malaysia, serves a population of around 2.5 million people. It is also providing cares to 80 to 90 per cent of cancer patients in Sarawak [12]. In this study, we aimed to evaluate the prevalence of HFS in cancer patients receiving capecitabine treatment at SGH, including the time to HFS onset and worsening, as well as any dose-reductions and treatment interruptions due to HFS. We also sought to explore the underlying demographic, clinical characteristics, and laboratory parameters as potential risk factors associated with the development of HFS associated with capecitabine treatment.
Methods
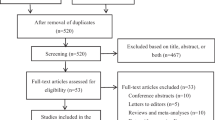
In this retrospective, cross-sectional study, we included adult cancer patients aged 18 years and above who were receiving cancer care at Sarawak General Hospital and collected their capecitabine prescription from the hospital pharmacy between April 1, 2021, and June 30, 2022. We included patients who had received capecitabine monotherapy or combination therapy with another agent for a minimum of two treatment cycles. To ensure sufficient capecitabine exposure and avoid underestimation of HFS rate, we excluded those who prematurely discontinued their treatment (≤1 cycle) or lacked records indicating treatment continuation in their medical notes.
Upon initiation of capecitabine treatment, patients at our centre received urea cream as standard care for preventing HFS. Besides that, pharmacist counselling was given on non-pharmacological preventive measures, including avoiding exposure to extreme temperatures (e.g., washing hands with hot water) and minimising friction, trauma, and pressure on palms and soles [13]. In cases where HFS worsened to grade 2 or 3, capecitabine treatment might be interrupted and/or the dose reduced.
We retrieved information related to the patients' demographics, anthropometric measurements, comorbidities, concomitant medications, chemotherapy regimens, primary cancer site, cancer staging, Eastern Cooperative Oncology Group (ECOG) performance status, and their laboratory investigations at baseline before the first cycle of capecitabine treatment from their medical notes. We extracted the results of full blood counts, liver and renal function tests from the laboratory reports. Creatinine clearance was calculated using the Cockcroft-Gault method: ([140 – age, year] × [weight, kg]) x [0.85 if female]) / (72 x [serum creatinine, μmol/L] x 0.0113 [conversion factor to mg/dl]) [14]. The degree of HFS was assessed by attending doctors upon completion of every capecitabine treatment cycle and graded from 1 to 3 using the NCI-CTCAE, version 5 [5].
In our analysis, we calculated the HFS prevalence among the cancer patients receiving capecitabine, the median number of treatment cycles until the onset of HFS or worsened HFS of grade ≥2, and the proportions of HFS-related treatment interruptions and dose reductions. We collected baseline demographic and clinical characteristics, along with laboratory results, and compared between patients who developed HFS and those who did not. We conducted multivariable logistic regression analysis to explore the associations of demographic and clinical characteristics, with the occurrence of HFS of all-grade or grade ≥2. We employed another multivariable regression model for baseline laboratory findings, with age, sex, race, and prior chemotherapy history added as potential confounders to explore laboratory risk factors associated with the occurrence or worsening of HFS. We included both clinically and statistically significant variables (variables with p<0.10 from the univariable analysis were included) in the multivariable models. We used jamovi software, version 2.3 (The jamovi project, Sydney, Australia) and SPSS Statistics for Windows, version 16 (SPSS Inc., Chicago, Ill., USA) to perform our analyses. We set a p-value less than 0.05 as the statistical significance level.
However, we excluded breast cancer, cancer treatment intent, and capecitabine regime (monotherapy or combination therapy) from the multivariable analyses due to their high correlation with patient’s sex (breast cancer in females), cancer staging (palliative treatment intent in patients who had cancer stage IV), and capecitabine dose (patients receive lower capecitabine dose [2000mg/m2/day] when it is given in combination therapy), respectively. In addition, we also opted to include neutrophil count instead of total white blood cell count due to the high correlation between these two measures.
Results
We included a total of 369 patients receiving capecitabine for analysis. The majority were female (50.4%), Chinese (37.7%), with a mean age of 57.0 years (standard deviation 11.7). Nearly two-thirds (65.9%) of the patients had colorectal cancer, followed by breast (16.5%) and gastric cancers (7.9%). Most of them were at stage III (42.2%) and IV (44.7%) and had an ECOG performance status of ≤1 (84.3%). Around 48.0% of the patients were taking capecitabine with palliative treatment intent. Slightly more than half (54.2%) were taking capecitabine in combination regimen, with majority (49.1%) receiving capecitabine in combination with oxaliplatin. Most patients were following the dosage regimen of 2000mg/m2/day (53.9%) or less (16.3%), with a median treatment duration of six cycles, as of the time of data collection. In terms of comorbidities, the majority had hypertension (44.4%) and dyslipidaemia (24.7%) and were taking calcium channel blockers (CCB) (16.8%) and statins (12.7%) (Table 1).
About half (50.1%) of the patients receiving capecitabine experienced HFS of all-grade, with 14.6% experiencing HFS of grade ≥2. The median number of treatment cycles until the onset of HFS was three cycles, and until HFS of grade ≥2 was five cycles. Among those who had HFS (n=185), about 22% had their chemotherapy treatment affected; 37 (20.0%) experienced treatment interruption, and 22 (11.9%) had their dosage reduced (Table 1).
When comparing patients with HFS to those without the condition, we noted that the patients who had HFS were significantly more likely to be female and Chinese but less likely to be of Iban descent. Additionally, compared to patients who did not develop HFS, those with HFS had a higher proportion of patients diagnosed with breast cancer but had a smaller percentage of patients with colorectal cancer. Furthermore, individuals with HFS were more likely to be at stage IV of their cancer and had a history of prior chemotherapy. They were also more likely to be undergoing palliative chemotherapy, receiving capecitabine monotherapy, prescribed at a higher dose of 2500mg/m2/day, and have taken a significantly longer duration of chemotherapy. Besides that, we noted that individuals who had HFS were significantly less likely to be taking CCB and more likely to be taking supplementary folic acid (Table 1). In terms of laboratory parameters, capecitabine patients who had HFS had significantly lower counts of total white blood cells, specifically neutrophils, red blood cells, and platelets, along with a reduced globulin level at baseline, in comparison to those who did not develop HFS (Table 3).
In the multivariable logistic regression analysis, we found that older age (OR 1.03, 95%CI 1.01, 1.06), prior treatment of chemotherapy (OR 2.09, 95% CI 1.22, 3.58), a higher capecitabine dose (OR 2.96, 95% CI 1.62, 5.38), and a longer treatment duration in cycles (OR 1.36, 95% CI 1.21, 1.51) were significantly associated with the occurrence of HFS in capecitabine patients. Our results also showed supplementary folic acid intake was significantly associated with higher risk of all-grade HFS (OR 3.27 95%CI 1.45, 7.35). Conversely, the concurrent use of CCB (OR 0.27, 95% CI 0.12, 0.60) was significantly associated with a reduced likelihood of developing HFS (Table 4). For HFS of grade ≥2, risk factors that showed significant associations were older age (OR 1.04, 95%CI 1.01, 1.08), female sex (OR 2.10, 95%CI 1.05, 4.18), Chinese race (OR 2.10, 95%CI 1.06 to 4.18), a higher capecitabine dose (OR 2.62, 95%CI 1.28, 5.35), and the absence of CCB in concomitant medications (OR 0.21, 95%CI 0.06, 0.78) (Table 4). In terms of laboratory investigations, only lower neutrophil count (OR 0.77 95%CI 0.66, 0.89) showed a significant association with the development of all-grade HFS in the multivariable analysis. None of the laboratory tests showed a significant association with the occurrence of grade ≥2 HFS, after controlling for other variables (Table 5). The univariable analysis findings can be found in the supplementary tables (Supplementary materials, Table S1 and Table S2).
Discussion
The HFS prevalence in our study (all grades, 50.1%; grade ≥2, 14.6%) is consistent with previous findings in other populations [8, 10, 11]. Despite urea cream being routinely prescribed for HFS at our centre and pharmacist counselling given, we still observed around 30% of the HFS cases worsened, and 20% required capecitabine treatment adjustments. Randomised trial evaluating urea cream's efficacy for HFS has shown mixed results [15, 16]. Other potential treatments for HFS includes pyridoxine, celecoxib, and vitamin E [17,18,19], however, dose interruption or reduction remains the main approach to mitigate the condition [3, 4]. Future studies are warranted to determine treatment options to improve HFS management.
A higher capecitabine dose was significantly associated with an increased HFS rate in our study. Furthermore, HFS was more likely to occur in later capecitabine treatment cycles, consistent with previous studies demonstrating a higher occurrence with prolonged capecitabine treatment [20, 21]. HFS showed a dose-related and cumulative exposure association in patients receiving capecitabine in this study. Besides that, our univariable analysis suggested a lower HFS risk in the combination therapy group compared to those receiving capecitabine alone (Supplementary material, Table S1). However, as our patients received lower capecitabine dose when it is given in combination with other agents, the effect is confounded by the lower prescribed dose.
In addition, consistent with other studies [22, 23], our study showed a significant association between HFS and increasing age. Older patients often present with pre-existing comorbidities and are prescribed with multiple concurrent medications [22, 24]. Furthermore, a study showed that elderly patients may have reduced drug clearance, leading to higher capecitabine exposure compared to younger individuals [25]. These factors could contribute to the increased risk of HFS occurrence in older individuals.
We also found that female patients were more prone to develop HFS of grade ≥2. Sex difference in capecitabine toxicity has been previously reported [22, 26], possibly attributed to varying body compositions between males and females [27, 28]. A study showed that female colorectal cancer patients had more dose-limiting toxicity than male patients when given capecitabine based on their body surface area [26]. There is a growing call for sex-specific dosing strategies in cancer therapy [27, 29]. Future research could address this disparity between males and females, exploring alternative capecitabine dosing strategies beyond flat dose and body-surface-area-based dosing [27, 28].
Furthermore, studies have shown racial and ethnic differences in chemotherapy toxicities [30, 31]. In a study, African Americans and Hispanics demonstrated significantly higher capecitabine dose reductions due to side effects compared to non-Hispanic Caucasians, with African Americans also showing a non-significant increase in HFS risk relative to Caucasians [30]. Sarawak has a multiracial population comprising predominantly Ibans, Chinese, and Malays. Our findings indicate that Chinese patients had a significantly higher risk of grade ≥2 HFS compared to other racial groups. These disparities in HFS susceptibility may be attributed to genetic polymorphisms in capecitabine metabolism [32, 33], that leads to variation in toxicity profiles [34] across different races. However, a study in Singapore did not demonstrate such racial association with HFS risk, despite having a similar multiracial setting with a predominantly Chinese patient population (81%) [35]. Another potential explanation for the observed difference in HFS risk could be the racial variation in adherence to capecitabine treatment [36].
Our finding revealed a significant association of supplementary folic acid intake with increased HFS risk, consistent with a previous research by Yap et al. showing that folate levels are independent predictors of grade ≥2 HFS [35]. Besides that, an observational study in colorectal cancer patients found that use of folic acid supplements during capecitabine treatment significantly associated with increased risk of toxicities [37]. The presence of folate stablises the binding of the active fluorouracil metabolite of capecitabine, fluorodeoxyuridine monophosphate, to thymidylate synthase, forming a covalently bound ternary complex [38]. A higher level of exogenous folate may enhance the inhibition of thymidylate synthase, thereby contributing to toxicities [35].
Pyridoxine has been suggested to potentially prevent HFS [17, 39]. About 10% of our patients were prescribed vitamin B complex containing pyridoxine around the initiation of capecitabine treatment. However, we did not observe any effect on HFS in the present study.
Furthermore, the use of CCB was associated with a reduced risk of all-grade and grade ≥2 HFS in the present study, which was not previously reported. One potential explanation is that CCB may exhibit anti-inflammatory effects, as shown in in-vitro studies, which could mitigate HFS induced by capecitabine [40, 41]. While Kanbayashi et al found that concomitant use of renin-angiotensin system inhibitors was associated with a higher risk of HFS [42], we did not find such association in our study. The exact mechanism by which antihypertensive agents affects HFS is still unclear. In addition, others studies found that concomitant use of proton pump inhibitors may ameliorate capecitabine-induced HFS [43]; however, it is suggested that proton pump inhibitor use may reduce capecitabine bioavailability through increased gastric pH, potentially leading to decreased efficacy [44]. Future studies are needed to confirm the association between the use of CCB and HFS, understand the mechanisms on HFS, and explore possible interactions with capecitabine.
Besides that, we identified a negative association between baseline neutrophil count and the occurrence of HFS. While low baseline neutrophil counts and HFS have been associated with better outcomes after fluoropyrimidine-based chemotherapy, individually [45, 46], the relationship between neutrophil count and HFS remains unclear. This observation is counterintuitive, considering that HFS is thought to be related to inflammatory changes mediated by cyclooxygenase-2 [47]. However, the low baseline neutrophil level may be confounded by previous chemotherapy exposure, despite we had adjusted for this in the multivariable analysis. We noted that patients with prior chemotherapy treatment were more susceptible to HFS. Given the observational nature of our study, we cannot ascertain whether this is a delayed effect of previous chemotherapy. Furthermore, we did not gather information on the duration of previous treatment and the time gap to the initiation of capecitabine treatment.
This study has several limitations. Firstly, it was conducted in a single centre. However, Sarawak General Hospital serves the majority of cancer patients in Sarawak, making our study population representative of a large segment of cancer patients. Secondly, the retrospective design and use of secondary data from paper notes made our study prone to missing data and entry errors. Despite our efforts to capture capecitabine patients within the cross-sectional study period, we were unable to trace all patients' clinic documentations. Thirdly, we acknowledge the potential for inter-assessor variability in HFS assessment, despite the clear definitions provided in CTCAE version 5 [5]. The assessment of HFS was conducted by trained medical doctors in our centre.
In summary, our study provides real-world data on capecitabine-associated HFS among Malaysian patients. We also identified several risk factors associated with the occurrence and worsening of HFS with capecitabine use in cancer therapy. We showed that HFS is dose-limiting and associated with prolonged capecitabine exposure. Older age, Chinese race, and female sex were the significant demographic risk factors associated with HFS. We also found that folic acid intake may exacerbate the risk of HFS. Furthermore, our findings suggest a potential association between the use of CCB and a reduced risk of HFS and its worsening. We also observed a potential relationship between low neutrophil count and HFS. These findings enable us to assess the prevalence and impact of HFS in Malaysian patient population, while the identification of associated risk factors contributes to the understanding of HFS development that may inform strategies for its effective management.
Data availability
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
References
Chintala L, Vaka S, Baranda J, Williamson SK (2011) Capecitabine versus 5-fluorouracil in colorectal cancer: where are we now? Oncol Rev 5(2):129–140. https://doi.org/10.1007/s12156-011-0074-3
Ter Veer E, Ngai LL, Valkenhoef GV, Mohammad NH, Anderegg MCJ, van Oijen MGH, van Laarhoven HWM (2017) Capecitabine, 5-fluorouracil and S-1 based regimens for previously untreated advanced oesophagogastric cancer: A network meta-analysis. Sci Rep 7(1):7142. https://doi.org/10.1038/s41598-017-07750-3
Kwakman JJM, Elshot YS, Punt CJA, Koopman M (2020) Management of cytotoxic chemotherapy-induced hand-foot syndrome. Oncol Rev 14(1):442. https://doi.org/10.4081/oncol.2020.442
Gressett SM, Stanford BL, Hardwicke F (2006) Management of hand-foot syndrome induced by capecitabine. J Oncol Pharm Pract 12(3):131–141. https://doi.org/10.1177/1078155206069242
U.S. Department of Health and Human Services (2017) Common Terminology Criteria for Adverse Events (CTCAE) Version 5.0. United States. Available from: https://ctep.cancer.gov/protocoldevelopment/electronic_applications/ctc.htm#ctc_50. Accessed 12 February 2024
Hsu YH, Shen WC, Wang CH, Lin YF, Chen SC (2019) Hand-foot syndrome and its impact on daily activities in breast cancer patients receiving docetaxel-based chemotherapy. Eur J Oncol Nurs 43:101670. https://doi.org/10.1016/j.ejon.2019.09.011
Urakawa R, Tarutani M, Kubota K, Uejima E (2019) Hand Foot Syndrome Has the Strongest Impact on QOL in Skin Toxicities of Chemotherapy. J Cancer 10(20):4846–4851. https://doi.org/10.7150/jca.31059
Genetech USA (2015) XELODA (capecitabine) tablets, for oral use: Highlights for prescribing information. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2015/020896s037lbl.pdf. Accessed 31 October 2023
Leicher LW, de Graaf JC, Coers W, Tascilar M, de Groot JW (2017) Tolerability of capecitabine monotherapy in metastatic colorectal cancer: a real-world study. Drugs R D 17(1):117–124. https://doi.org/10.1007/s40268-016-0154-8
Blum JL, Barrios CH, Feldman N, Verma S, McKenna EF, Lee LF, Scotto N, Gralow J (2012) Pooled analysis of individual patient data from capecitabine monotherapy clinical trials in locally advanced or metastatic breast cancer. Breast Cancer Res Treat 136(3):777–788. https://doi.org/10.1007/s10549-012-2288-x
Huang J, Ye S, Feng S, Zheng M, Zhong M (2023) Prevalence of hand-foot syndrome following chemotherapy for colorectal cancer: a systematic review and meta-analysis. Int J Colorectal Dis 38(1):61. https://doi.org/10.1007/s00384-023-04345-5
CodeBlue (2021) MOH Mulls Building New Cancer Centre In Samarahan. CodeBlue. Available from:, Malaysia https://codeblue.galencentre.org/2021/11/17/moh-mulls-building-new-cancer-centre-in-samarahan. Accessed 31 May 2023
Mohamad N, Othman NA, Yusup NL (2017) Cancer Drug Counselling: A Guide for Pharmacists. Pharmaceutical Services Division, Ministry of Health Malaysia, Malaysia Available from: https://pharmacy.moh.gov.my/sites/default/files/document-upload/cancer-drug-counselling-guide-pharmacists-2016.pdf. Accessed 18 March 2024
Cockcroft DW, Gault MH (1976) Prediction of creatinine clearance from serum creatinine. Nephron 16(1):31–41. https://doi.org/10.1159/000180580
Hofheinz RD, Gencer D, Schulz H, Stahl M, Hegewisch-Becker S, Loeffler LM, Kronawitter U, Bolz G, Potenberg J et al (2015) Mapisal Versus Urea Cream as Prophylaxis for Capecitabine-Associated Hand-Foot Syndrome: A Randomized Phase III Trial of the AIO Quality of Life Working Group. J Clin Oncol 33(22):2444–2449. https://doi.org/10.1200/JCO.2014.60.4587
Wolf SL, Qin R, Menon SP, Rowland KM Jr, Thomas S, Delaune R, Christian D, Pajon ER Jr, Satele DV et al (2010) Placebo-controlled trial to determine the effectiveness of a urea/lactic acid-based topical keratolytic agent for prevention of capecitabine-induced hand-foot syndrome: North Central Cancer Treatment Group Study N05C5. J Clin Oncol 28(35):5182–5187. https://doi.org/10.1200/JCO.2010.31.1431
Corrie PG, Bulusu R, Wilson CB, Armstrong G, Bond S, Hardy R, Lao-Sirieix S, Parashar D, Ahmad A et al (2012) A randomised study evaluating the use of pyridoxine to avoid capecitabine dose modifications. Br J Cancer 107(4):585–587. https://doi.org/10.1038/bjc.2012.318
Zhang RX, Wu XJ, Wan DS, Lu ZH, Kong LH, Pan ZZ, Chen G (2012) Celecoxib can prevent capecitabine-related hand-foot syndrome in stage II and III colorectal cancer patients: result of a single-center, prospective randomized phase III trial. Ann Oncol 23(5):1348–1353. https://doi.org/10.1093/annonc/mdr400
Yamamoto D, Yamamoto C, Iwase S, Kuroda Y, Odagiri H, Nagumo Y (2010) Efficacy of Vitamin E Treatment for Hand-Foot Syndrome in Patients Receiving Capecitabine. Breast Care (Basel) 5(6):415–416. https://doi.org/10.1159/000322660
Stein A, Quidde J, Schroder JK, Gohler T, Tschechne B, Valdix AR, Hoffkes HG, Schirrmacher-Memmel S, Wohlfarth T et al (2016) Capecitabine in the routine first-line treatment of elderly patients with advanced colorectal cancer--results from a non-interventional observation study. BMC Cancer 16:82. https://doi.org/10.1186/s12885-016-2113-8
Tanyi JL, Smith JA, Ramos L, Parker CL, Munsell MF, Wolf JK (2009) Predisposing risk factors for palmar-plantar erythrodysesthesia when using liposomal doxorubicin to treat recurrent ovarian cancer. Gynecol Oncol 114(2):219–224. https://doi.org/10.1016/j.ygyno.2009.04.007
Jiang Y, Mason M, Cho Y, Chittiprolu A, Zhang X, Harden K, Gong Y, Harris MR, Barton DL (2022) Tolerance to oral anticancer agent treatment in older adults with cancer: a secondary analysis of data from electronic health records and a pilot study of patient-reported outcomes. BMC Cancer 22(1):950. https://doi.org/10.1186/s12885-022-10026-3
Meulendijks D, van Hasselt JGC, Huitema ADR, van Tinteren H, Deenen MJ, Beijnen JH, Cats A, Schellens JHM (2016) Renal function, body surface area, and age are associated with risk of early-onset fluoropyrimidine-associated toxicity in patients treated with capecitabine-based anticancer regimens in daily clinical care. Eur J Cancer 54:120–130. https://doi.org/10.1016/j.ejca.2015.10.013
van Beek MWH, Roukens M, Jacobs WCH, Timmer-Bonte JNH, Kramers C (2018) Real-World Adverse Effects of Capecitabine Toxicity in an Elderly Population. Drugs Real World Outcomes 5(3):161–167. https://doi.org/10.1007/s40801-018-0138-9
Louie SG, Ely B, Lenz HJ, Albain KS, Gotay C, Coleman D, Raghavan D, Shields AF, Gold PJ et al (2013) Higher capecitabine AUC in elderly patients with advanced colorectal cancer (SWOGS0030). Br J Cancer 109(7):1744–1749. https://doi.org/10.1038/bjc.2013.517
Ilich AI, Danilak M, Kim CA, Mulder KE, Spratlin JL, Ghosh S, Chambers CR, Sawyer MB (2016) Effects of gender on capecitabine toxicity in colorectal cancer. J Oncol Pharm Pract 22(3):454–460. https://doi.org/10.1177/1078155215587345
Ozdemir BC, Gerard CL, Espinosa da Silva C (2022) Sex and Gender Differences in Anticancer Treatment Toxicity: A Call for Revisiting Drug Dosing in Oncology. Endocrinology 163(6). https://doi.org/10.1210/endocr/bqac058
Wagner AD (2020) Sex differences in cancer chemotherapy effects, and why we need to reconsider BSA-based dosing of chemotherapy. ESMO Open 5(5):e000770. https://doi.org/10.1136/esmoopen-2020-000770
Ozdemir BC, Oertelt-Prigione S, Adjei AA, Borchmann S, Haanen JB, Letsch A, Mir O, Quaas A, Verhoeven RHA et al (2022) Investigation of sex and gender differences in oncology gains momentum: ESMO announces the launch of a Gender Medicine Task Force. Ann Oncol 33(2):126–128. https://doi.org/10.1016/j.annonc.2021.11.011
Brazelton A, Yande S, Pope R, Johnson ML, Musher B, Trivedi MV (2022) Racial and ethnic differences in capecitabine toxicity in patients with gastrointestinal tract cancers. Ann Gastroenterol 35(2):182–186. https://doi.org/10.20524/aog.2022.0688
Mizusawa J, Sato H, Rubinstein LV, Fujiwara T, Yonemori K, Hirakawa A (2023) Racial differences in longitudinal toxicities of anticancer agents in early phase cancer clinical trials. Cancer Med 12(17):18098–18109. https://doi.org/10.1002/cam4.6370
Rosmarin D, Palles C, Church D, Domingo E, Jones A, Johnstone E, Wang H, Love S, Julier P et al (2014) Genetic markers of toxicity from capecitabine and other fluorouracil-based regimens: investigation in the QUASAR2 study, systematic review, and meta-analysis. J Clin Oncol 32(10):1031–1039. https://doi.org/10.1200/JCO.2013.51.1857
Lin S, Yue J, Guan X, Yuan P, Wang J, Luo Y, Fan Y, Cai R, Li Q et al (2019) Polymorphisms of MTHFR and TYMS predict capecitabine-induced hand-foot syndrome in patients with metastatic breast cancer. Cancer Commun (Lond) 39(1):57. https://doi.org/10.1186/s40880-019-0399-z
Ma Y, Tang L, Wang HX, Xu YC, Ma Y, Zhang FC (2012) Capecitabine for the treatment for advanced gastric cancer: efficacy, safety and ethnicity. J Clin Pharm Ther 37(3):266–275. https://doi.org/10.1111/j.1365-2710.2011.01289.x
Yap YS, Kwok LL, Syn N, Chay WY, Chia JWK, Tham CK, Wong NS, Lo SK, Dent RA et al (2017) Predictors of Hand-Foot Syndrome and Pyridoxine for Prevention of Capecitabine-Induced Hand-Foot Syndrome: A Randomized Clinical Trial. JAMA Oncol 3(11):1538–1545. https://doi.org/10.1001/jamaoncol.2017.1269
Zahrina AK, Norsa'adah B, Hassan NB, Norazwany Y, Norhayati I, Roslan MH, Wan Nazuha WR (2014) Adherence to capecitabine treatment and contributing factors among cancer patients in Malaysia. Asian Pac J Cancer Prev 15(21):9225–9232. https://doi.org/10.7314/apjcp.2014.15.21.9225
Kok DE, van Duijnhoven FJ, Lubberman FJ, McKay JA, Lanen AV, Winkels RM, Wesselink E, van Halteren HK, de Wilt JH et al (2024) Intake and biomarkers of folate and folic acid as determinants of chemotherapy-induced toxicities in patients with colorectal cancer: a cohort study. Am J Clin Nutr 119(2):294–301. https://doi.org/10.1016/j.ajcnut.2023.11.023
Longley DB, Harkin DP, Johnston PG (2003) 5-fluorouracil: mechanisms of action and clinical strategies. Nat Rev Cancer 3(5):330–338. https://doi.org/10.1038/nrc1074
Lian S, Zhang X, Zhang Y, Zhao Q (2021) Pyridoxine for prevention of hand–foot syndrome caused by chemotherapy agents: a meta-analysis. Clin Exp Dermatol 46(4):629–635. https://doi.org/10.1111/ced.14486
Das R, Burke T, Van Wagoner DR, Plow EF (2009) L-type calcium channel blockers exert an antiinflammatory effect by suppressing expression of plasminogen receptors on macrophages. Circ Res 105(2):167–175. https://doi.org/10.1161/CIRCRESAHA.109.200311
Shima E, Katsube M, Kato T, Kitagawa M, Hato F, Hino M, Takahashi T, Fujita H, Kitagawa S (2008) Calcium channel blockers suppress cytokine-induced activation of human neutrophils. Am J Hypertens 21(1):78–84. https://doi.org/10.1038/ajh.2007.13
Kanbayashi Y, Taguchi T, Ishikawa T, Otsuji E, Takayama K (2023) Risk factors of capecitabine-induced hand-foot syndrome: a single-institution, retrospective study. Oncology 101(7):407–414. https://doi.org/10.1159/000529851
Takemura M, Ikemura K, Yoshinami T, Toyozumi Y, Shintani T, Ueda M, Shimazu K, Okuda M (2022) Proton pump inhibitors ameliorate capecitabine-induced hand-foot syndrome in patients with breast cancer: a retrospective study. Anticancer Res 42(5):2591–2598. https://doi.org/10.21873/anticanres.15737
Chu MP, Hecht JR, Slamon D, Wainberg ZA, Bang YJ, Hoff PM, Sobrero A, Qin S, Afenjar K et al (2017) association of proton pump inhibitors and capecitabine efficacy in advanced gastroesophageal cancer: secondary analysis of the TRIO-013/LOGiC randomized clinical trial. JAMA Oncol 3(6):767–773. https://doi.org/10.1001/jamaoncol.2016.3358
Grothey A, Yoshino T, Bodoky G, Ciuleanu T, Garcia-Carbonero R, Garcia-Alfonso P, Van Cutsem E, Muro K, Mytelka DS et al (2018) Association of baseline absolute neutrophil counts and survival in patients with metastatic colorectal cancer treated with second-line antiangiogenic therapies: exploratory analyses of the RAISE trial and validation in an electronic medical record data set. ESMO Open 3(3):e000347. https://doi.org/10.1136/esmoopen-2018-000347
Zielinski C, Lang I, Beslija S, Kahan Z, Inbar MJ, Stemmer SM, Anghel R, Vrbanec D, Messinger D et al (2016) Predictive role of hand-foot syndrome in patients receiving first-line capecitabine plus bevacizumab for HER2-negative metastatic breast cancer. Br J Cancer 114(2):163–170. https://doi.org/10.1038/bjc.2015.419
Lou Y, Wang Q, Zheng J, Hu H, Liu L, Hong D, Zeng S (2016) Possible Pathways of Capecitabine-Induced Hand-Foot Syndrome. Chem Res Toxicol 29(10):1591–1601. https://doi.org/10.1021/acs.chemrestox.6b00215
Subcommittee of Malaysian Guidelines for Good Clinical Practice (2018) Malaysian Guideline for Good Clinical Practice. National Pharmaceutical Regulatory Agency, Malaysia Available from: http://www.nccr.gov.my/view_file.cfm?fileid=31. Accessed 31 May 2023
Acknowledgements
We want to thank the Director General of Health of Malaysia for the permission to publish this article. We also thank Li Fang Lim, Hanisah Hossain and Roisin Lim for their contributions in data collection.
Funding
The authors received no financial support for the research and publication of this paper.
Author information
Authors and Affiliations
Contributions
Teck Long King conceived and designed the study. Teck Long King collected and analysed the data, and drafted the manuscript. Pei Jye Voon provided access to the patient data. Pei Jye Voon, Kah Hay Yuen, and Dzul Azri Mohamed Noor reviewed and revised the manuscript. All authors approved the manuscript.
Corresponding author
Ethics declarations
Ethic approval
The study protocol received approval from Medical Research and Ethics Committee of the Ministry of Health, Malaysia (NMRR ID-22-01737-RYT). The study was conducted in compliance with Malaysian Guideline for Good Clinical Practice [48] and Declaration of Helsinki.
Consent to participate
Given the retrospective nature of this study, the informed consent process was waived.
Competing interests
The authors have no relevant financial or non-financial interest to disclose.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 24 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
King, T.L., Voon, P.J., Yuen, K.H. et al. Hand-foot syndrome in cancer patients on capecitabine: examining prevalence, impacts, and associated risk factors at a cancer centre in Malaysia. Support Care Cancer 32, 345 (2024). https://doi.org/10.1007/s00520-024-08490-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00520-024-08490-7