Abstract
Purpose
The opioid crisis resulting from its use disorder and overdose poses additional challenges for cancer pain management. The American Society of Clinical Oncology Practice Guideline recommends acupuncture therapy for the management of adult cancer-related pain (CRP), but the effectiveness of transcutaneous electrical acupoint stimulation (TEAS) on CRP remains uncertain.
Methods
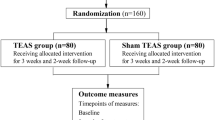
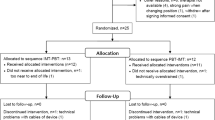
This 5-week prospective randomized clinical trial was conducted at 2 hospitals in China, and participants with CRP receiving chronic opioid therapy were randomized 1:1 into two groups between December 2014 and June 2018. The true TEAS group underwent 15 sessions of TEAS treatments over 3 consecutive weeks, while the control group received sham stimulation. The primary outcome was the numerical rating scale (NRS) score in the past 24h at week 3. The secondary outcomes included morphine equivalent daily dose, quality of life and adverse events.
Results
A total of 159 participants were included in the modified intention-to-treat population. The baseline characteristics were similar in both groups. The mean NRS scores were 0.98 points at week 3 in the true TEAS group and 1.41 points in the sham group, with the mean difference between groups of -0.43 points (P < 0.001; OR = 0.68, P < 0.05). The proportion of patients with NRS reduction more than thirty percentage at week 3 was 50.00% in the true TEAS group and 35.44% in the sham group (RD = 0.15, P > 0.05; RR = 1.41, P > 0.05). No significant difference in pain intensity between the two groups was observed during the follow-up period without TEAS intervention (week 4, OR = 0.83, P > 0.05; week 5, OR = 0.83, P > 0.05). The Karnofsky Performance Status value suggested that patients in the true TEAS group experienced an improved quality of life (Between-group differences: week 3, 3.5%, P < 0.05; week 4, 4.6%, P < 0.001; week 5, 5.6%, P < 0.001).
Conclusions
The 3-week application of TEAS in patients with CRP receiving chronic opioid therapy resulted in a statistically significant reduction in pain scores, but the observed reduction was of uncertain clinical significance. The prolonged analgesic effect of TEAS was not confirmed in this trial.
ClinicalTrial.gov
ChiCTR-TRC-13003803.

Similar content being viewed by others
Data availability
The data generated in this study, including deidentified participant data and the data dictionary, are available upon reasonable request. Researchers interested in accessing the data for valid research purposes can contact the corresponding author, Yi Liang, at liangyiwww@126.com, with a methodologically sound proposal. Requests will be subject to review and approval.
Abbreviations
- CRP:
-
Cancer-related pain
- EQ-5D-3L:
-
Three-level version of EuroQol five-dimensional questionnaire
- KPS:
-
The Karnofsky Performance Status
- MEDD:
-
Morphine equivalent daily dose
- mITT:
-
Modified intention-to-treat
- NRS:
-
Numerical rating scale
- QoL:
-
Quality of life
- TEAS:
-
Transcutaneous electrical acupoint stimulation
- TENS:
-
Transcutaneous electrical nerve stimulation
References
Neufeld NJ, Elnahal SM, Alvarez RH (2017) Cancer pain: a review of epidemiology, clinical quality and value impact. Futur Oncol (London, England) 13(9):833–841. https://doi.org/10.2217/fon-2016-0423
Ng JY, Sharma AE (2021) Guidelines for cancer-related pain: a systematic review of complementary and alternative medicine recommendations. Pain Pract: Off J World Inst Pain 21(4):454–467. https://doi.org/10.1111/papr.12964
van den Beuken-van Everdingen MH, Hochstenbach LM, Joosten EA, Tjan-Heijnen VC, Janssen DJ (2016) Update on prevalence of pain in patients with cancer: systematic review and meta-analysis. J Pain Symptom Manage 51(6):1070-1090.e1079. https://doi.org/10.1016/j.jpainsymman.2015.12.340
Anekar AA, Hendrix JM, Cascella M (2023) WHO Analgesic ladder. In: StatPearls [Internet]. StatPearls Publishing, Treasure Island, FL. https://www.ncbi.nlm.nih.gov/books/NBK554435/
Plante GE, VanItallie TB (2010) Opioids for cancer pain: the challenge of optimizing treatment. Metab: Clin Exp 59(Suppl 1):S47-52. https://doi.org/10.1016/j.metabol.2010.07.010
Corli O, Floriani I, Roberto A, Montanari M, Galli F, Greco MT, Caraceni A, Kaasa S, Dragani TA, Azzarello G, Luzzani M, Cavanna L, Bandieri E, Gamucci T, Lipari G, Di Gregorio R, Valenti D, Reale C, Pavesi L, Iorno V, Crispino C, Pacchioni M, Apolone G, On behalf of the CERP STUDY OF PAIN GROUP (2016) Are strong opioids equally effective and safe in the treatment of chronic cancer pain? A multicenter randomized phase 4 ‘real life’ trial on the variability of response to opioids. Annals of Oncology 27:1107–1115
Pargeon KL, Hailey BJ (1999) Barriers to effective cancer pain management: a review of the literature. J Pain Symptom Manage 18(5):358–368. https://doi.org/10.1016/s0885-3924(99)00097-4
Liou KT, Korenstein D, Mao JJ (2021) Medicare coverage of acupuncture for chronic low back pain: does it move the needle on the opioid crisis? J Gen Intern Med 36(2):527–529. https://doi.org/10.1007/s11606-020-05871-6
Dowell D, Haegerich TM, Chou R (2016) CDC Guideline for Prescribing Opioids for Chronic Pain. MMWR Recomm Rep 65:1–49. https://doi.org/10.15585/mmwr.rr6501e1external
Mosley M, Gurman AW (2014) The Opioid epidemic: current challenges for pain management. MD Conf Express 14(2):6–7. https://doi.org/10.1177/155989771402001
Gomes T, Tadrous M, Mamdani MM, Paterson JM, Juurlink DN (2018) The burden of opioid-related mortality in the United States. JAMA Netw Open 1(2):e180217. https://doi.org/10.1001/jamanetworkopen.2018.0217
Wiffen PJ, Wee B, Derry S, Bell RF, Moore RA (2017) Opioids for cancer pain ‐ an overview of Cochrane reviews. Cochrane Database Syst Rev (7). https://doi.org/10.1002/14651858.CD012592.pub2
Yang J, Bauer BA, Wahner-Roedler DL, Chon TY, Xiao L (2020) The modified WHO analgesic ladder: is it appropriate for chronic non-cancer pain? J Pain Res 13:411–417. https://doi.org/10.2147/jpr.S244173
Liou KT, Trevino KM, Meghani SH, Li QS, Deng G, Korenstein D et al (2021) Fear of analgesic side effects predicts preference for acupuncture: a cross-sectional study of cancer patients with pain in the USA. Support Care Cancer : Off J Multinatl Assoc Support Care Cancer 29(1):427–435. https://doi.org/10.1007/s00520-020-05504-y
Lu W, Rosenthal DS (2010) Recent advances in oncology acupuncture and safety considerations in practice. Curr Treat Options Oncol 11:141–146. https://doi.org/10.1007/s11864-010-0126-0
Lu W, Rosenthal DS (2018) Oncology Acupuncture for chronic pain in cancer survivors: a reflection on the American Society of Clinical Oncology Chronic Pain Guideline. Hematol Oncol Clin North Am 32(3):519–533. https://doi.org/10.1016/j.hoc.2018.01.009
Ge L, Wang Q, He Y, Wu D, Zhou Q, Xu N et al (2022) Acupuncture for cancer pain: an evidence-based clinical practice guideline. Chin Med 17(1):8. https://doi.org/10.1186/s13020-021-00558-4
Swarm RA, Paice JA, Anghelescu DL, Are M, Bruce JY, Buga S et al (2019) Adult Cancer Pain, Version 3.2019, NCCN Clinical Practice Guidelines in Oncology. J Nat Compr Cancer Netw: JNCCN 17(8):977–1007. https://doi.org/10.6004/jnccn.2019.0038
Song B, Chang Y, Li Y, Zhu J (2020) Effects of transcutaneous electrical acupoint stimulation on the postoperative sleep quality and pain of patients after video-assisted thoracoscopic surgery: a prospective, randomized controlled trial. Nat Sci Sleep 12:809–819. https://doi.org/10.2147/nss.S270739
Zhou X, Cao SG, Tan XJ, Liu XD, Li ZQ, Kong LX et al (2021) Effects of Transcutaneous electrical acupoint stimulation (TEAS) on Postoperative recovery in patients with gastric cancer: a randomized controlled trial. Cancer Manag Res 13:1449–1458. https://doi.org/10.2147/cmar.S292325
Meng D, Mao Y, Song Q, Yan C, Zhao Q, Yang M et al (2021) The efficacy and safety of transcutaneous electrical acupoint stimulation (TEAS) for postoperative pain in laparoscopy: A protocol for systematic review and meta-analysis of randomized controlled trials. Medicine 100(25):e26348. https://doi.org/10.1097/md.0000000000026348
Ao L, Shi J, Bai Y, Zhang S, Gan J (2021) Effects of transcutaneous electrical acupoint stimulation on perioperative immune function and postoperative analgesia in patients undergoing radical mastectomy: A randomized controlled trial. Exp Ther Med 21(3):184. https://doi.org/10.3892/etm.2021.9615
Fang J, Zhang Y, Xuan L, Liu K, Chen L (2006) Observation on clinical therapeutic effect of transcutaneous point electric stimulation on periarthritis of shoulder at different stages. Chin Acupunct Moxibustion 26:11–14
Gavronsky S, Koeniger-Donohue R, Steller J, Hawkins JW (2012) Postoperative pain: acupuncture versus percutaneous electrical nerve stimulation. Pain Manag Nurs: Off J Am Soc Pain Manag Nurses 13(3):150–156. https://doi.org/10.1016/j.pmn.2009.08.001
Han JS, Ho YS (2011) Global trends and performances of acupuncture research. Neurosci Biobehav Rev 35(3):680–687. https://doi.org/10.1016/j.neubiorev.2010.08.006
Qu F, Li R, Sun W, Lin G, Zhang R, Yang J et al (2017) Use of electroacupuncture and transcutaneous electrical acupoint stimulation in reproductive medicine: a group consensus. J Zhejiang Univ Sci B 18(3):186–193. https://doi.org/10.1631/jzus.B1600437
Turan Z, Topaloğlu M, Özyemişçi-Taşkıran Ö (2020) What is the effectiveness and adverse event data of transcutaneous electrical nerve stimulation (TENS) in reducing pain in adults with chronic pain? An overview of Cochrane Reviews summary with commentary. Turk J Phys Med Rehab 66(2):210–213. https://doi.org/10.5606/tftrd.2020.6484
Hurlow A, Bennett MI, Robb KA, Johnson MI, Simpson KH, Oxberry SG (2012) Transcutaneous electric nerve stimulation (TENS) for cancer pain in adults. Cochrane Database Syst Rev (3). https://doi.org/10.1002/14651858.CD006276.pub3
Gibson W, Wand BM, Meads C, Catley MJ, O’Connell NE (2019) Transcutaneous electrical nerve stimulation (TENS) for chronic pain - an overview of Cochrane Reviews. Cochrane Database Syst Revs 2(2):Cd011890. https://doi.org/10.1002/14651858.CD011890.pub2
Liang Y, Bao G, Gong L, Zhou J, Kong X, Ran R et al (2019) Evaluating the analgesic effect and advantage of transcutaneous electrical acupoint stimulation combined with opioid drugs for moderate to severe cancer-related pain: a study protocol for a randomized controlled trial. Trials 20(1):40. https://doi.org/10.1186/s13063-018-3145-y
Dworkin RH, Turk DC, Wyrwich KW, Beaton D, Cleeland CS, Farrar JT et al (2008) Interpreting the clinical importance of treatment outcomes in chronic pain clinical trials: IMMPACT recommendations. J Pain 9(2):105–121. https://doi.org/10.1016/j.jpain.2007.09.005
Dworkin RH, Turk DC, Peirce-Sandner S, Burke LB, Farrar JT, Gilron I et al (2012) Considerations for improving assay sensitivity in chronic pain clinical trials: IMMPACT recommendations. Pain 153(6):1148–1158. https://doi.org/10.1016/j.pain.2012.03.003
Liu GG, Wu H, Li M, Gao C, Luo N (2014) Chinese time trade-off values for EQ-5D health states. Value Health : J Int Soc Pharmacoeconomics Outcome Res 17(5):597–604. https://doi.org/10.1016/j.jval.2014.05.007
Williamson A, Hoggart B (2005) Pain: a review of three commonly used pain rating scales. J Clin Nurs 14(7):798–804. https://doi.org/10.1111/j.1365-2702.2005.01121.x
Grieco A, Long CJ (1984) Investigation of the Karnofsky Performance Status as a measure of quality of life. Health Psychol: Off J Div Health Psychol, Am Psychol Assoc 3(2):129–142. https://doi.org/10.1037//0278-6133.3.2.129
Bennett MI, Johnson MI, Brown SR, Radford H, Brown JM, Searle RD (2010) Feasibility study of transcutaneous electrical nerve stimulation (TENS) for cancer bone pain. J Pain 11(4):351–359. https://doi.org/10.1016/j.jpain.2009.08.002
Robb KA, Newham DJ, Williams JE (2007) Transcutaneous electrical nerve stimulation vs. transcutaneous spinal electroanalgesia for chronic pain associated with breast cancer treatments. J Pain Symptom Manag 33(4):410–419. https://doi.org/10.1016/j.jpainsymman.2006.09.020
Gadsby JG, Franks A, Jarvis P, Dewhurst F (1997) Acupuncture-like transcutaneous electrical nerve stimulation within palliative care: a pilot study. Complement Ther Med 5(1):13–18. https://doi.org/10.1016/S0965-2299(97)80084-2
Hershman DL, Unger JM, Greenlee H, Capodice JL, Lew DL, Darke AK et al (2018) Effect of acupuncture vs sham acupuncture or waitlist control on joint pain related to aromatase inhibitors among women with early-stage breast cancer: a randomized clinical Trial. JAMA 320(2):167–176. https://doi.org/10.1001/jama.2018.8907
Caraceni A, Hanks G, Kaasa S, Bennett MI, Brunelli C, Cherny N et al (2012) Use of opioid analgesics in the treatment of cancer pain: evidence-based recommendations from the EAPC. Lancet Oncol 13(2):e58-68. https://doi.org/10.1016/s1470-2045(12)70040-2
Lu Z, Wang Q, Sun X, Zhang W, Min S, Zhang J et al (2021) Transcutaneous electrical acupoint stimulation before surgery reduces chronic pain after mastectomy: A randomized clinical trial. J Clin Anesth 74:110453. https://doi.org/10.1016/j.jclinane.2021.110453
Gewandter JS, Smith SM, Dworkin RH, Turk DC, Gan TJ, Gilron I et al (2021) Research approaches for evaluating opioid sparing in clinical trials of acute and chronic pain treatments: initiative on methods, measurement, and pain assessment in clinical trials recommendations. Pain 162(11):2669–2681. https://doi.org/10.1097/j.pain.0000000000002283
He YGX, May BH, Zhang AL, Liu Y, Lu C, Mao JJ, Xue CC, Zhang H (2020) Clinical evidence for association of acupuncture and acupressure with improved cancer pain: a systematic review and meta-analysis. JAMA Oncol 6:271–278. https://doi.org/10.1001/jamaoncol.2019.5233
Nakano J, Ishii K, Fukushima T, Ishii S, Ueno K, Matsuura E et al (2020) Effects of transcutaneous electrical nerve stimulation on physical symptoms in advanced cancer patients receiving palliative care. Int J Rehab Res Internationale Zeitschrift fur Rehabilitationsforschung Revue internationale de recherches de readaptation 43(1):62–68. https://doi.org/10.1097/mrr.0000000000000386
Tao WW, Jiang H, Tao XM, Jiang P, Sha LY, Sun XC (2016) Effects of Acupuncture, Tuina, Tai Chi, Qigong, and traditional Chinese medicine five-element music therapy on symptom management and quality of life for cancer patients: a meta-analysis. J Pain Symptom Manage 51(4):728–747. https://doi.org/10.1016/j.jpainsymman.2015.11.027
Szmit M, Agrawal S, Gozdzik W, Kubler A, Agrawal A, Pruchnicki P et al (2021) Transcutaneous Electrical acupoint stimulation reduces postoperative analgesic requirement in patients undergoing inguinal hernia repair: a randomized, placebo-controlled study. J Clin Med 10(1). https://doi.org/10.3390/jcm10010146
Hale M, Wild J, Reddy J, Yamada T, Arjona Ferreira JC (2017) Naldemedine versus placebo for opioid-induced constipation (COMPOSE-1 and COMPOSE-2): two multicentre, phase 3, double-blind, randomised, parallel-group trials. Lancet Gastroenterol Hepatol 2(8):555–564. https://doi.org/10.1016/s2468-1253(17)30105-x
Wu GJ, Xu F, Sun XM, Chen JDZ (2020) Transcutaneous neuromodulation at ST36 (Zusanli) is more effective than transcutaneous tibial nerve stimulation in treating constipation. J Clin Gastroenterol 54(6):536–544. https://doi.org/10.1097/mcg.0000000000001184
Takahashi T (2006) Acupuncture for functional gastrointestinal disorders. J Gastroenterol 41(5):408–417. https://doi.org/10.1007/s00535-006-1773-6
Xiao Y, Xu F, Lin L, Chen J (2022) Transcutaneous electrical acustimulation improves constipation by enhancing rectal sensation in patients with functional constipation and lack of rectal sensation. Clin Transl Gastroenterol 13(5):e00485. https://doi.org/10.14309/ctg.0000000000000485
Zhou J, Wang J, Ning B, Hu Y, Zhao Q, Tan W et al (2022) Sustained ameliorating effects and autonomic mechanisms of transcutaneous electrical acustimulation at ST36 in patients with chronic constipation. Front Neurosci 16:1038922. https://doi.org/10.3389/fnins.2022.1038922
Acknowledgements
We extend our gratitude to Professor Mao and her team from the School of Public Health at Zhejiang Chinese Medicine University for their valuable statistical support. We would like to thank Erasmus Shannah Rose for the language review of this paper. Additionally, we appreciate the dedication of the medical and nursing staff at the trial sites, as well as the patients who participated in the study.
Funding
This work received financial support from the Key Project of Health Science and Technology Program of National Health Commission and Zheiang Province (WKJ-ZI-2436 to YLiang), the Key Foundation of Chinese Medicine Research Program of Zhejiang Province (2021ZZ017 to Yi Liang), and the General Medical Science and Technology Project of Zhejiang Province (2014KYA162 to Yi Liang), and the Graduate Student Scientific Research Fund Project of Zhejiang Chinese Medicine University (2023YKJ01 to Yi Liang). The funding source played no role in the study design, data collection and analyses, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Yi Liang and Jianqiao Fang had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Zhengyi Lyu, Shuxin Tian and Guanai Bao contributed equally as first authors to this work. Concept and design: Yi Liang, Jianqiao Fang, Liyan Gong, Xiangming Kong, Weiping Zhang. Acquisition, analysis, or interpretation of data: Guanai Bao, Zhengyi Lyu, Yang Liu, Shan Liu, Jie Zhou, Conghua Ji, Ran Ran, Nie Na. Drafting of the manuscript: Zhengyi Lyu, Shuxin Tian, Rui Huang. Revising of the manuscript: Yi Liang, Guoyin Kai, Jianqiao Fang. Statistical analysis: Shuxin Tian, Shan Liu, Conghua Ji. Obtained funding: Yi Liang, Jianqiao Fang. Administrative, technical, or material support: Xianming Lin, Xiaomei Shao, Weiping Zhang. Supervision: Yi Liang, Weiping Zhang, Liyan Gong. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Ethical approval for this trial was granted by the medical ethics committee of the Third Affiliated Hospital of Zhejiang Chinese Medical University [2014–09-86 (Ke)].
Consent to participate
All study participants signed informed consent forms.
Consent for publication
All authors consent to the publication of this manuscript in Supportive Care in Cancer.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Zhengyi Lyu, Shuxin Tian, and Guanai Bao contributed equally as first authors to this work.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lyu, Z., Tian, S., Bao, G. et al. Transcutaneous electrical acupoint stimulation for cancer-related pain management in patients receiving chronic opioid therapy: a randomized clinical trial. Support Care Cancer 32, 16 (2024). https://doi.org/10.1007/s00520-023-08240-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00520-023-08240-1