Abstract
Purpose
Some cancer patients in palliative care require intravenous administration of symptom relieving drugs. Peripherally inserted central catheters (PICCs) and midline catheters (MCs) provide easy and accessible intravenous access. However, limited evidence supports the use of these devices in palliative care. The aim was to assess the use, safety, and efficacy of PICC and MC in this patient population.
Methods
A retrospective study of all palliative care cancer patients who received PICC or MC at the Department of Palliative Medicine at Akershus University Hospital between 2020 and 2022.
Results
A total of 374 patients were included; 239 patients received a PICC and 135 an MC with a total catheterization duration of 11,698 days. The catheters remained in place until death in 91% of patients, with a median catheter dwell time of 21 days for PICCs and 2 days for MCs. The complication rate was 3.3 per 1000 catheter days, with minor bleeding and accidental dislocation as the most common. The catheters were utilized primarily for opioids and other symptom directed treatments, and 89% of patients received a patient or nurse-controlled analgesia pump. Patients with PICC or MC discharged to home or nursing homes spent 81% of their time out of hospital.
Conclusion
PICC and MC provide safe parenteral access for palliative care cancer patients where intravenous symptom treatment is indicated. Their use can facilitate intravenous symptom treatment beyond the confines of a hospital and supplement the traditional practice relying on subcutaneous administration.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In palliative care, the traditional route for parenteral drug administration is subcutaneous [1, 2]. However, the intravenous route has several potential advantages in this setting, including a faster onset of drug action, greater flexibility in injection volumes, drug and excipient use, less local infiltrations and irritation, and more predictable pharmacokinetics [3,4,5]. Peripheral venous access can be challenging to obtain and maintain in palliative care cancer patients. The use of peripherally inserted central venous catheters (PICC) and midline catheters (MC) allows for easy administration of intravenous medications and provision of regular care in both hospital and home settings, potentially reducing the burden on the patient and healthcare system compared to peripheral venous access [6, 7].
PICCs are inserted in the basilic, cephalic, or brachial vein without general anesthesia or a surgical incision, and their presence reduces the discomfort of frequent venipuncture and cannulation. The MC is a peripheral intravenous line measuring 3–8 inches and is inserted using a similar procedure, but with its tip not advanced beyond the distal axillary vein. The MC is indicated for short-term use (< 30 days). Removal of PICC or MC is a simple, safe procedure that can, if needed, be carried out at home [8].
There are several studies on PICC placement in cancer patients with the indication to maintain chemotherapy and/or nutrition therapy [9, 10]. These studies suggest PICCs are generally safe and convenient and can substitute other centrally or peripherally inserted venous devices. Based on systematic reviews and expert opinions, The Michigan Appropriateness Guide for Intravenous Catheters [8] states that a PICC could be indicated for palliative treatment during end-of-life care. Tripathi et al. [11] recently conducted a systematic review on the use of MC in a wide patient population. The review demonstrated complication rates for MC comparable to PICC but included only one study conducted in a palliative care unit [7]. In this study, the procedure was well tolerated, and the quality of care measured with the Palliative care Outcome Scale improved after catheter insertion, likely caused by improved symptom treatment.
Guidelines for symptom treatment in palliative care are based on the subcutaneous route [1, 2, 12] but advocate the intravenous route when subcutaneous administration is likely to fail, rapid pain control is needed or the patient already has a central venous catheter (CVC). The growing access to PICC and MC facilitates intravenous treatment in home care settings, making these methods increasingly popular in palliative care. However, there is limited evidence to support this practice. Therefore, this study is aimed at assessing the use, safety, and efficacy of PICC and MC in palliative care cancer patients admitted to a specialist palliative care hospital unit.
Methods
Study design
This is a retrospective observational study.
Study population and setting
This study was conducted at the Department of Palliative Medicine, Akershus University Hospital (Ahus), which has a 12-bed palliative care ward, and serves a catchment area of 594.000. All adult cancer patients who had received a PICC or MC during admission at the Department of Palliative Medicine from January 2020 to December 2021 were included.
PICC and MC were inserted ultrasound guided by specially trained nurse anesthetists to ensure that best insertion practice was followed. The PICCs used were the 1 lm 4Fr PowerPiccSolo. The MCs used were 1 lm 4Fr Vygon and 1 lm 18G PowerGlide catheters.
Data collection and management
Patients were identified from the medical records based on ICD-10 code for cancer (C00-C97) and intervention codes for insertion of a PICC or MC (PHX 17, PHX 25). Based on their first catheter insertion, the study population was stratified into two groups, as either PICC or MC. All subsequent CVC insertions were recorded for each patient, and each insertion was treated as an independent data point for outcome analysis. Patients who had a central venous line prior to admission were excluded due to expected insufficient documentation. Patients still alive at the time of data extraction in October 2022 were excluded.
We described patient demographics in accordance with the European Association for Palliative Care (EAPC) list of characteristics that describes palliative care patients (EAPC basic dataset) [13]. Present anticancer therapy was registered as ongoing if continuation of chemotherapy, immunotherapy, or radiotherapy was planned at time of PICC or MC placement. Only diagnoses and treatment documented in the patient’s medical records could be considered for registration.
Karnofsky performance status (KPS) [14] was registered at time of catheter insertion as either 50 or above or below 50. KPS is registered as less than 50 when a patient is unable to care for self and in need of institutional or equivalent care. When performance status was registered in the patient files, that value was recorded. In patients lacking KPS, a score was estimated based on the description of the patient's performance status.
Indications for PICC and MC placement, oral and parenteral medication before and after PICC/MC placement, data regarding the procedure, the total days of PICC/MC use, the type and time of catheter related complications, and the reasons for PICC removal were collected. Overall survival was measured from the time of PICC/MC placement to the time of death. Catheter complications were registered as described in the electronic records. Any suspected catheter-related infections were registered as a catheter related infection even if further diagnostics were not performed. Catheter occlusion, arm swelling, or pain was registered as a thrombosis/occlusion even if further diagnostics were not performed.
Statistical analyses
Continuous variables were summarized as median and categorical variables were described using frequency distributions. The chi-squared test or Fisher’s exact test when appropriate was employed to assess the association between two categorical variables. The Wilcoxon-Mann–Whitney test (for two groups) was used to compare continuous variables. Survival was presented with Kaplan–Meier curves and compared using the log-rank test.
All analyses were conducted using IBM SPSS Statistics 28.0.1.1 software.
Research ethics
A Remit Assessment Form was submitted to the Regional Ethics Committee, which approved the project as a quality assurance project. The study was approved by the Data Protection Officer at Ahus.
Results
Study population
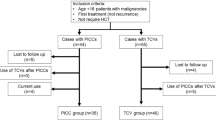
From January 2020 to December 2021, 948 individual patients were admitted to the Department of Palliative Medicine. A PICC or MC intervention code was present in 401 individual medical records. Among these 374 individual patients were included in the study with a total catheterization duration of 11,698 days. Patients incorrectly registered as admitted to the department (n = 5), patients with a CVC in place prior to admission (n = 10), and patients alive at time of data extraction (n = 12) were excluded.
The median (range) age of patients was 70 (28–97) years, and 50% were female (Table 1). The majority of patients (93%) had metastatic disease. The most common cancer diagnoses were malignant neoplasms of digestive organs (35%) and respiratory organs (21%). Most patients (74%) had stopped all anticancer therapy at time of catheter insertion, and 74% of the patients had a Karnofsky status below 50.
Of the 374 patients in the study, 239 (64%) patients received a PICC and 135 (36%) patients received an MC. Patients who received an MC had a lower performance status (p < 0.001) and less ongoing anticancer treatment (p < 0.001) than patients who received a PICC. Catheters were most commonly placed in the right arm (87%) through the basilic (76%) or brachial vein (17%).
Utilization
Referral indications and actual catheter utilization for the 374 patients are shown in Table 2. Regular or as needed intravenous medication for symptom treatment was a referral reason in 98% of patients. The need for an intravenous patient or nurse controlled analgesia/infusion pump (PCA-pump) was an indication in 76% of referrals. The option to do blood sampling from PICC or MC was an added indication in 64% of patients. Hydration or parenteral nutrition was a referral reason for 5.5% of patients, and tumor-targeted treatment was one of the indications in 1.1% of patients.
Before catheter placement, 53% of patients already had one or more PCA-pumps. Following catheter placement, 331 (89%) of patients received PCA-pumps. Of these patients, 48% periodically utilized more than one PCA pump.
Following catheter placement, intravenous opioids were administered to 91% of the 374 patients, most commonly through a PCA-pump (86%) (Table 3). Intravenous sedatives were administered with a PCA-pump in 45% of patients, and another 21% received sedatives as intravenous injections. Intravenous antibiotics including treatment of oral candidiasis was provided for 19% of patients. Between 13 and 19% of patients received intravenous antacids, corticosteroids, and antiemetics. Palliative sedation with propofol infusion was provided for 12 patients (3.2%). After catheter placement, 22% of patients did not receive any oral medication.
Catheter dwell time and complications
Patient survival and catheter dwell time are shown in Fig. 1. The first established catheters were kept until time of death for 339 patients (91%). The median (IQR) survival after PICC placement was 24 (8–63) days and after MC 3 (1–8) days (p < 0.001). The catheter dwell time for PICC and MC was, respectively, 21 (8–53) and 2 (1–8) days (p < 0.001).
In eight patients, the catheters were removed due to no further need. Ten patients with MC kept their catheter for more than the recommended 30 days. No complications were observed in these patients. In the PICC group, eleven patients kept their catheter for more than six months.
The overall complication rate was 3.3 per 1000 catheter days. The rate was higher for the MC group (7.2) compared to PICC (2.9) (p = 0.038) (Table 4). Complications overall occurred at median (IQR) 13 (1–50) days post PICC and 2 (0–19) days post-MC placement.
Catheter = related complications were reported in 39 patients and resulted in catheter removal for 28 of these patients (Table 4). After catheter removal, 25 of these patients received a new PICC, MC, or VAP directly (15 patients) or after a period of subcutaneous or short term intravenous access (10 patients) (Fig. 2).
One of the patients with a second PICC needed a third and fourth catheter due to occlusion and later infection. One patient with PICC died 71 days after catheter placement with symptoms of a catheter related sepsis confirmed by positive blood and catheter tip culture of Staphylococcus aureus. Intensive care unit admission was discussed but not initiated due to terminal stage cancer.
Hemorrhage and accidental dislocation occurred the same day as catheter placement in eight patients. Hemorrhage was controlled with dressings and compression without removing the catheter in all but one patient.
Hospitalization after catheter placement
Of patients who received an MC, 68% were hospitalized until death (Table 5). Patients who received a PICC were more often discharged to home (49%, p < 0.001). Patients with a PICC or MC eventually died in hospital in 57% of cases. Patients with an MC more often died in hospital than patients with a PICC (p < 0.001). Of the 233 patients who were discharged alive after catheter placement, 51% had no readmissions to hospital. Patients discharged alive after PICC or MC placement spent median (IQR) 81% (70%–91%) of remaining lifetime outside the hospital, i.e., at home or in nursing home. Regular injections that had been established during admission were consistently maintained in home care or nursing homes, and 81% of patients discharged alive had a PCA-pump.
Discussion
The PICC and MC catheters were safely inserted and had few complications in palliative care cancer patients, with accidental catheter dislocation and minor bleeding as the most common complications. The patients had advanced disease, low performance status, and short life expectancy reflected in the short catheter dwell time and patient survival after PICC and especially MC placement. Importantly the catheters were in place until death in 91% of patients, indicating that PICC and MC is a feasible solution for parenteral drug administration in palliative care patients with advanced disease.
The present finding of low complication rates aligns with previous studies on palliative care cancer patients earlier in their disease trajectory. [9,10,11]. Former studies have primarily focused on providing intravenous access for life prolonging treatment such as tumor targeted treatment, antibiotics, hydration, and nutrition. These studies have demonstrated that PICC and MC were well tolerated and preferred by patients over repeated peripheral vein punctures, with complications occurring in a minority of cases. The present study suggests that PICC and MC could provide safe and effective intravenous access also for late-stage cancer patients requiring parenteral administration of symptom relieving drugs.
The catheters were used in line with referral reasons, primarily for analgesia and sedatives with a PCA-pump. Intravenous nutrition and hydration were infrequent in our study population indicating that a venous access is not seen as an opportunity to intensify life prolonging and perhaps futile treatment in end-stage palliative care.
Catheter dwell time and patient survival after catheter placement have a large range in the literature. Catheters placed early in the disease trajectory primarily for tumor-targeted treatment are typically kept for several months [10]. Studies on terminally ill cancer patients [15,16,17] report a PICC dwell time in the same range as our study.
The complication rate for PICC catheters in this study (2.9 per 1000 catheter days) was higher than the rate reported in a large retrospective study of PICC catheters in cancer patients receiving chemotherapy [10] (1.09 per 1000 catheter days). However, that study did not include accidental catheter removal as a complication, which may account for the difference in findings. The short dwell time for MC in our study, contributes to a low number of complications but a relatively high complication rate per 1000 catheter days. The proximity of MC insertion to the time of death in the present study reflects a tradition at the Department of Palliative Medicine of requesting MC catheters to avoid repeated venipuncture and secure intravenous access during end of life care.
Estimation of life expectancy is challenging. Even experienced palliative care clinicians tend to overestimate [18]. This could influence the choice of an MC or PICC catheter: The low median dwell time for PICC where 62% of patients kept the catheter for a month or less, indicates that for many of these patients an MC would be adequate. Furthermore, the low median survival after MC placement suggests that a peripheral venous catheter could have been sufficient for some of these patients. This raises concerns about the potential for overtreatment due to the availability of interventions. There were no registered complications for patients with an indwelling MC for longer than the recommended duration of one month. This suggest that patients with short life expectancy and well-functioning MC may not need a renewed PICC or MC, even if the patient lives longer than expected. This further highlights the importance of prudent use of medical interventions. It is crucial to ascertain that treatments are employed judiciously even when they are easily available.
Subcutaneous administration is traditionally accepted as a good enough parenteral route for symptom relief in palliative care cancer patients. Our research findings may reflect a shift in clinical practice towards the intravenous route. The clinicians’ emerging preference for the intravenous route for pain and symptom management in palliative care should be explored further to see if this relates to better symptom treatment or simply represents an unnecessary complication of current practice guidelines. Despite receiving intravenous infusions, patients in our study spent most of their remaining lifetime at home or nursing homes demonstrating that intravenous treatment no longer is limited to hospitalized patients.
A major strength of this study is the inclusion of a complete cohort of patients receiving PICC and MC catheter at a large department of palliative medicine that serves a catchment area of one-tenth of the Norwegian population. Complete hospital records and charts were thoroughly reviewed adding to a high internal validity.
One important limitation in the present study is the single-center study design where hospital traditions including the institutional culture for intravenous symptom relief with MC or PICC in end of life care could affect external validity. Potentially incomplete records and missing data limit the retrospective study design based on medical records. Even though IV PCA-pumps and other established infusions were routinely continued after discharge, there was no direct access to records from nursing home or home care, therefore events occurring after discharge that were not reported to the Department of Palliative Medicine might have been missed. The low median dwell time for MC in this material prohibits drawing solid conclusions regarding the complication profile. Patients in their final days of life are spared examinations and diagnostics not resulting in any meaningful therapy. Consequently, complications like thrombosis and infection might have been missed, even though medical history or findings that indicated such complications, also without diagnostic verification, were counted as a complication in our study.
Despite these limitations, the present findings indicate that PICC and MC are safe and efficient parenteral access devices for palliative care cancer patients where continuity of intravenous symptom treatment is seen as an advantage, and can provide a robust option for intravenous symptom treatment outside the hospital setting where practice traditionally has relied on subcutaneous infusions.
Data availability
The data that support the findings of this study are not openly available due to reasons of sensitivity and are available from the corresponding author upon reasonable request. Data are located in controlled access data storage at Akershus University Hospital.
References
Fallon M, Giusti R, Aielli F, Hoskin P, Rolke R, Sharma M et al (2018) Management of cancer pain in adult patients: ESMO clinical practice guidelines. Ann Oncol 29(Supplement 4):iv166–iv91. https://doi.org/10.1093/annonc/mdy152
National Institute for Health and Care Excellence (2015) Care of dying adults in the last days of life. https://www.nice.org.uk/guidance/ng31. Accessed 15 Sep 2023
Dale O, Thoner J, Nilsen T, Tveita T, Borchgrevink PC, Klepstad P (2007) Serum and cerebrospinal fluid morphine pharmacokinetics after single doses of intravenous and intramuscular morphine after hip replacement surgery. Eur J Clin Pharmacol 63(9):837–842. https://doi.org/10.1007/s00228-007-0329-x
Thomas JR, Wallace MS, Yocum RC, Vaughn DE, Haller MF, Flament J (2009) The INFUSE-Morphine Study: use of recombinant human hyaluronidase (rHuPH20) to enhance the absorption of subcutaneously administered morphine in patients with advanced illness. J Pain Symptom Manage 38(5):663–672. https://doi.org/10.1016/j.jpainsymman.2009.03.009
Tveita T, Thoner J, Klepstad P, Dale O, Jystad Å, Borchgrevink P (2008) A controlled comparison between single doses of intravenous and intramuscular morphine with respect to analgesic effects and patient safety. Acta Anaesthesiol Scand 52(7):920–925
Bortolussi R, Zotti P, Conte M, Marson R, Polesel J, Colussi A et al (2015) Quality of life, pain perception, and distress correlated to ultrasound-guided peripherally inserted central venous catheters in palliative care patients in a home or hospice setting. J Pain Symptom Manage 50(1):118–123. https://doi.org/10.1016/j.jpainsymman.2015.02.027
Magnani C, Calvieri A, Giannarelli D, Espino M, Casale G (2019) Peripherally inserted central catheter, midline, and “short” midline in palliative care: patient-reported outcome measures to assess impact on quality of care. J Vasc Access 20(5):475–481. https://doi.org/10.1177/1129729818814732
Chopra V, Flanders SA, Saint S, Woller SC, O’Grady NP, Safdar N et al (2015) The Michigan Appropriateness Guide for Intravenous Catheters (MAGIC): results from a multispecialty panel using the RAND/UCLA appropriateness method. Ann Intern Med 163(6 Suppl):S1-40. https://doi.org/10.7326/M15-0744
He E, Ye K, Zheng H (2021) Clinical effect and safety of venous access ports and peripherally inserted central catheters in patients receiving tumor chemotherapy: a systematic review and meta-analysis. Ann Palliat Med 10(8):9105–13. https://doi.org/10.21037/apm-21-1926
Campagna S, Gonella S, Berchialla P, Morano G, Rigo C, Zerla PA et al (2019) Can peripherally inserted central catheters be safely placed in patients with cancer receiving chemotherapy? A retrospective study of almost 400,000 catheter-days. Oncologist 24(9):e953–e959. https://doi.org/10.1634/theoncologist.2018-0281
Tripathi S, Kumar S, Kaushik S (2021) The practice and complications of midline catheters: a systematic review. Crit Care Med 49(2):e140–e150. https://doi.org/10.1097/ccm.0000000000004764
Norwegian Directorate of Health. Nasjonalt handlingsprogram med retningslinjer for palliasjon i kreftomsorgen [National action program with guidelines for palliative care for patients with cancer]. Oslo: Norwegian Directorate of Health; 2019. https://www.helsedirektoratet.no/tema/palliasjon. Accessed 15 Sep 2023
Sigurdardottir KR, Kaasa S, Rosland JH, Bausewein C, Radbruch L, Haugen DF (2014) The european association for palliative care basic dataset to describe a palliative care cancer population: results from an international Delphi process. Palliat Med 28(6):463–473. https://doi.org/10.1177/0269216314521264
Karnofsky DA, Abelmann WH, Craver LF, Burchenal JH (1948) The use of the nitrogen mustards in the palliative treatment of carcinoma With particular reference to bronchogenic carcinoma. Cancer 1(4):634–56. https://doi.org/10.1002/1097-0142(194811)1:4%3c634::AID-CNCR2820010410%3e3.0.CO;2-L
Park EJ, Park K, Kim JJ, Oh SB, Jung KS, Oh SY et al (2021) Safety, efficacy, and patient satisfaction with initial peripherally inserted central catheters compared with usual intravenous access in terminally Ill cancer patients: a randomized phase II study. Cancer Res Treat 53(3):881–888. https://doi.org/10.4143/crt.2020.1008
Yamada R, Morita T, Yashiro E, Otani H, Amano K, Tei Y et al (2010) Patient-reported usefulness of peripherally inserted central venous catheters in terminally ill cancer patients. J Pain Symptom Manage 40(1):60–66. https://doi.org/10.1016/j.jpainsymman.2009.11.327
Park K, Jun HJ, Oh SY (2016) Safety, efficacy, and patient-perceived satisfaction of peripherally inserted central catheters in terminally ill cancer patients: a prospective multicenter observational study. Support Care Cancer 24(12):4987–4992. https://doi.org/10.1007/s00520-016-3360-6
Amano K, Maeda I, Shimoyama S, Shinjo T, Shirayama H, Yamada T et al (2015) The accuracy of physicians’ clinical predictions of survival in patients with advanced cancer. J Pain Symptom Manage 50(2):139–46.e1. https://doi.org/10.1016/j.jpainsymman.2015.03.004
Funding
Open access funding provided by Akershus University Hospital (AHUS) This study was funded by South-Eastern Norway Regional Health Authority.
Author information
Authors and Affiliations
Contributions
OMF and SS conceived the idea for the study. OMF and EG planned and designed the study. EG performed the data collection, analyzed the data and prepared the first and final version of the manuscript. OMF, SS and KMA critically revised the manuscript and analyses strategy, and approved the submission of the final version.
Corresponding author
Ethics declarations
Ethics approval
A Remit Assessment Form was submitted to the Regional Ethics Committee, which approved the project as a quality assurance project. The study was approved by the Data Protection Officer at Akershus University Hospital.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gravdahl, E., Steine, S., Augestad, K.M. et al. Use and safety of peripherally inserted central catheters and midline catheters in palliative care cancer patients: a retrospective review. Support Care Cancer 31, 580 (2023). https://doi.org/10.1007/s00520-023-08045-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00520-023-08045-2