Abstract
Patient-reported outcome measures obtained via E-Health tools ease the assessment burden and encourage patient participation in cancer care (PaCC Study)
Background
E-health based patient-reported outcome measures (PROMs) have the potential to automate early identification of both nutrition status and distress status in cancer patients while facilitating treatment and encouraging patient participation. This cross-sectional study assessed the acceptability, accuracy, and clinical utility of PROMs collected via E-Health tools among patients undergoing treatment for stomach, colorectal, and pancreatic tumors.
Results
Eight-nine percent mostly, or completely, agreed that PROMs via tablets should be integrated in routine clinical care. Men were significantly more likely to require help completing the questionnaires than women (inv.OR= 0.51, 95% CI=(0.27, 0.95), p = 0.035). The level of help needed increased by 3% with each 1-year increase in age (inv. OR=1.03, 95% CI=(1.01, 1.06), p = 0.013). On average, a patient tended to declare weight which was 0.84 kg inferior to their true weight (Bland and Altman 95 % CI=(-3.9, 5.6); SD: 2.41) and a height which was 0.95 cm superior to their true height (Bland and Altman 95 % CI=(−5, 3.1); SD 2.08). Patient-reported nutrition status was significantly associated with the professionally generated assessment (95% CI=(2.27, 4.15), p < 0.001). As nutrition status declined, the distress score increased (95%CI=(0.88, 1.68), p < 0.001). Of the patients, 48.8% who were both distressed and malnourished requested supportive care to address their problems.
Conclusion
Patient-reported assessments utilizing E-health tools are an accurate and efficient method to encourage patient participation in cancer care while simultaneously ensuring that regular assessment of psycho-social and nutritional aspects of care are efficiently integrated in the daily clinical routine.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Medical care, including oncology care, is currently shifting from a disease-centered approach to a more personalized approach. As this shift occurs, the importance of integrating patient-reported (PR) assessments into routine care of cancer patients is increasing. Both psycho-social and nutrition interventions rely on validated screening tools designed to identify patients with a need for an intervention. However, until now, the burden of distributing, completing, and inputting data from both psycho-social and nutrition screening assessments with the purpose of triaging patient care is still placed largely on healthcare professionals (HCP) [1]. Both patients, and HCPs, can therefore benefit by harnessing the potential of E-health tools which integrate and automate assessments based on patient-reported outcomes measures (PROMs). This approach can improve efficiency, making it easier to integrate required screening and assessments into the clinical routine. Furthermore, E-health tools potentiate early identification and provide a more dynamic approach to patient-centered care by enabling and encouraging active patient participation [2, 3]. In fact, Chrischiles et al. found that among the general population, high-frequency users had higher odds of recognizing symptoms and adverse effects among users ≥ 65 years of age [4].
Therefore, the objective of this duo-centric study was to assess the acceptability, accuracy, and clinical utility of PROMs collected via E-Health tools among patients undergoing treatment for stomach, colorectal, and pancreatic tumors.
Methods
This cross-sectional study took place in Germany and Switzerland. The primary focus was to assess the accuracy and clinical utility of PROMs collected via E-health tools during routine care.
Participants and data collection
Patients diagnosed with stomach, pancreas, and colorectal cancer undergoing therapy were recruited using convenience sampling at out-patient cancer therapy centers in Germany and in Switzerland. All patients over the age of 18 who provided informed consent were eligible for inclusion in this study. Individuals with significant cognitive or functional health issues were not approached. As the goal was to assess the acceptance and accuracy of PROMs collected via E-Health tools for all patients receiving routine care, previous experience with tablet-based assessments was not considered. Exclusion criteria were limited to lack of consent and linguistic challenges as the questionnaires were provided only in German. All participants were formally asked for permission to use their de-identified data for research purposes and were given the option to opt out at any point. This study was approved by the Institutional Ethics Committee at the Ludwig Maximillian University of Munich (Reference number:19–954), Germany, and the Cantonal Ethics Committee Zurich, Switzerland (Reference number: 2018–01,129).
E-health platform
CANKADO’s E-health platform was utilized for all data collection. CANKADO is approved as an active Class I medical device within the European Union (registration number DE/CA59/11976/2017) and is compliant with the US Food and Drug Administration classification for Mobile Medical Devices (2015 Appendix B). CANKADO provides full patient privacy protection and data handling compliant with ICH GCP E6(R2).
Questionnaires
After obtaining consent, patients were given a tablet asked to complete a total of 79 tablet-based questions consisting of questions pertaining to the acceptability of tablet-based questionnaires and the following 2 validated questionnaires: (1) the Patient Generated Subjective Global Assessment Short Form (PG-SGA SF) which is designed for nutrition screening and assessment and (2) the distress thermometer (DT) which is designed for psycho-social screening and assessment [5,6,7]. If technical problems or any difficulties arose, patients were instructed to request assistance. Upon completion of the tablet-based questions, patients returned the tablet and it was disinfected. Dietitians then completed a nutrition risk assessment using the Nutrition Risk Score (NRS-2002) and weighed and measured the patients. Same day requests for further supportive care, any difficulty completing the questionnaires, as well as any need for assistance were tracked.
Patient Generated Subjective Global Assessment Short Form (German 18–006 v05.10.18.)
The PG-SGA SF was modified from the Subjective Global Assessment (SGA) for the oncology population by Ottery et al. It is recommended by the Oncology Nutrition Dietetic Practice Group of the Academy of Nutrition and Dietetics and has been translated and linguistically validated in many countries [8,9,10,11]. The German language validation was published in 2019, shortly before this study was initiated [12]. The PG-SGA SF comprises of 4 components: weight history, food intake, nutrition impact symptoms, and activities and function. The PG-SGA SF has demonstrated comparable sensitivity and specificity to that of the full-length scored tool [13]. Numerical scores range from 0 to 36 and a score ≥ 9 indicates a high risk for malnutrition [14].
Nutrition Risk Score (NRS-20002)
Also, recommended by ESPEN, the NRS-2002 is a nutrition risk screening tool for hospitalized patients and is designed to be completed by the HCP. The NRS-2002 consists of three components: the severity of disease, nutritional status, and age. A score ≥ 3 indicates risk of, or existing malnutrition [15].
Distress thermometer (DT)
Recommended by the National Comprehensive Cancer Network (NCCN), as well as national guidelines for psycho-oncological assessment, counseling, and treatment of adult cancer patients, the DT is a psycho-social subjective test based on patient-reported (PR) data aimed at measuring the level of distress the patient is currently experiencing. It consists of a scale from 0 to 10, with 0 indicating no distress and 10 indicating extreme distress [6, 16]. While some literature recommends a cutoff score of ≥ 4 as an indication of clinically elevated distress, the German language version has been validated at a cut off score of ≥ 5 [17].
Statistical analyses
All data was analyzed using only completers (patients without missing values). Questions related to acceptability and difficulty level of the E-health platform were formulated analogue to previous studies with each item scored on a 5-point Likert scale [18, 19]. Linear models and cumulative link models when the response distribution was discrete were used to analyze the significance of the results. When so, inverse cumulative odds ratio (inv. OR) with the corresponding 95% confidence interval (95% CI) were computed. Categorical data were presented as frequency (number) and percentage (%) and as their respective categories according to established cut off points. Proportions were compared with Chi-Squared Tests of Independence. Bland–Altman plots for weight, height, and body mass index (BMI) were used to analyze the agreement between PR and professionally measured data [20]. The Wilcoxon-Test was used to compare results between male and female patients. A linear mixed effect model using the patient as random effect and cancer type, time, and the interaction term as fixed effects was fitted to assess the weight evolution over time for each cancer type. Data analysis and presentation was done with the R system for statistical computing (version 3.6.1). All tests were two-sided and the significance level was set to 0.05.
Results
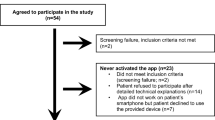
In total,188 patients were asked to participate. Nineteen percent (n = 36) chose not to participate or withdrew consent. One hundred fifty-two patients (median age 62 years; range 22–86 years) completed the questionnaires. Sixty-five patients had colorectal cancer (43%); 50 patients had pancreatic cancer (33%); 25 patients had stomach cancer (16%); and the remaining 12 patients (7%) listed their diagnosis as “other.” All reported results are based on completed information only.
A high proportion (89%) mostly, or completely, agreed that PROMs via tablets should be integrated in routine clinical care. While there was no evidence that gender affected this response (inv. OR = 0.62, 95% CI = (0.29,1.28), p = 0.207), younger patients agreed significantly more than older patients with the statement (inv. OR = 0.96, 95% CI = (0.93,0.99), p = 0.017). Similarly, older patients tended to find the tablet-based format more difficult to handle when compared to younger patients inv. OR = 1.03 95% CI = (1.0, 1.06), p = 0.052) (Fig. 1). While age was similarly distributed between the sexes, men were significantly more likely to require help completing the questionnaires than women (inv. OR = 0.51, 95% CI = (0.27, 0.95), p = 0.035). Additionally, the level of help needed increased by 3% with each 1-year increase in age (Inv. OR = 1.03, 95% CI = (1.01, 1.06), p = 0.013). Both effects remain statistically significant once adjusted for other variables.
PR-height and PR-weight were closely associated with the patients’ true height and weight. In fact, 83% of the patients declared a weight which was within 2 kilos (kg) of their true weight. Likewise, 86% of patients declared a height within 2 cm (cm) of their true height. On average a patient tended to declare weight which was 0.84 kg inferior to their true weight (Bland and Altman CI = 95%; SD: 2.41) (Fig. 2a), whereas patients tended to declare a height which was on average 0.95 cm superior to their true height (Bland and Altman CI = 95%; SD 2.08) (Fig. 2b). The average BMI was 23.92 kg/m2, which falls within the normal range proposed by the World Health Organization. Only ten patients had a BMI below 18.5 kg/m2 and thus would be considered at nutrition risk based on BMI alone, while 51 patients (34%) had a BMI above the normal range.
The PR-nutrition status was significantly associated with the HCP generated assessment. In fact, a 1-unit increase in NRS-2002 was associated with an average increase in PG-SGA SF of 3.21 (95% CI = (2.27, 4.15), p < 0.001). These results remained significant when adjusting the data for a more normal response distribution, both when using the square root transformation and the Asinh transformation (p < 0.001). When the continuous scales were transformed using their respective categorical cut off scores, analogue to Zhu et a., 70% of the PR- nutrition status correlated with the HCP assessed nutrition status (Table 1) [21].
Pancreatic and stomach cancer patients reported a mean weight loss of more than 6% (6.8 and 6.5% respectively) of their body weight when compared with their current PR-weight over the previous six months (Fig. 3). Colon cancer patients reported the least weight loss over the previous 6 months (3.9%). The PR evolution of weight over time showed a continuous weight loss trend among all three cancer types over the previous 6-month period. The average weight loss per month was 0.84 kg among stomach cancer patients (95% CI = (− 1.14, − 0.54), p < 0.001), followed by an average 0.78 kg loss among pancreatic cancer patients ((95% CI = (− 1.14, − 0.54), p < 0.001). Colon cancer patients reported a 0.5 kg loss per month ((95% CI = (− l0.68, − 0.31), p < 0.001).
As nutrition status declined, the distress score increased, while the activity levels declined. In fact, a 1-unit increase in PG-SGA SF score was associated with an average increase on the DT of 1.28 (95%CI = (0.88, 1.68), p < 0.001) and a simultaneous decrease in activity level (1 unit decrease in the activity level results in an increase on the PG-SGA-SF score of 4.75, (95% CI = (3.86, 5.64) p < 0.001). Similarly, there was a significant association between a rise in the PR-distress status and an increased NRS-2002 score (inv. OR = 1.33, 95%CI = (1.17, 1.52), p < 0.001). When the continuous scales were transformed using their respective categorical cut off scores, the significance remained (Table 1). No significant differences were found between males and females with respect to the level of distress (Wilcoxon p = 0.77) or the association between distress and nutrition status (p = 0.19).
A total of 38.2% (58/152 patients) of all patients requested supportive care in the form of nutrition interventions on the same day after completing the questionnaires. Similarly, 28.2% (43/152 patients) presented with both a declined nutrition status a distress level ≥ 5. Among these 43 patients, almost half (48.8%) requested same day supportive care. Patients who requested nutrition counseling had, on average, a 2.05 point higher PG-SGA SF score than patients who did not request counseling (95% CI = (− 0.2, 4.31), p = 0.074). Furthermore, patients seemed to have a good understanding of their nutrition risk. In fact, the PG-SGA score among patients who reported feeling well-nourished was, on average, 7.09 points lower than patients who did not believe that they were currently well nourished (95% CI = (4.52, 9.66), p < 0.001).
Discussion
E-health tools could potentially contribute to an easy and efficient identification and treatment of patients’ care needs while simultaneously encouraging active participation on behalf of the patient [22]. However, before these integration of E-health platforms into the clinical care settings, it is important to analyze how, and if, age and gender may affect the overall acceptance of E-health applications. Similar to previous studies conducted among oncology patients regarding the collection of PROMs via E-health tools, the majority of patients (89%) agreed that such methods should be integrated into clinical care [23,24,25,26]. This result may be biased due to the fact that those patients who agreed to participate in our study and similar studies were more likely to be open to the idea of E-health tools and have less difficulty using them. However, in our study, 188 patients were asked to participate. Nineteen percent (n = 36) chose not to participate or withdrew consent. Only half of the patients (18/36) cited reasons specifically related to the use of E-health tools, while others cited reasons not related to E-health tools such as being too tired or not having their reading glasses on hand. Therefore, we were not able to confirm or deny this potential bias. Our results also showed a linear relationship of age to the need for assistance while utilizing our E-health application. This result confirms previous research which found that elderly people have less technological self-efficacy and a higher levels of anxiety while using innovative technology in healthcare settings and therefore may require more assistance [27]. Gender additionally affected the likelihood that patients required assistance completing the questionnaires via E-health tools with women requiring significantly less help than men. This information may be useful in identifying the best collective for future researchers planning similar interventions with E-health tools and assessing their resources available for providing assistance when required.
As PROMs become more integrated into clinical care, it is also important that their clinical accuracy and utility is reported in various populations and settings. Therefore, we analyzed the differences between PR-data regarding height and weight as well as nutrition status and compared PR-data to data obtained by HCPs via established clinical assessments methods. We further analyzed what percentage of patients requested same day supportive care after completion of the PROMs questionnaires.
While previous studies have reported biases between self-reported and measured anthropometrics, these results are influenced by settings, social aspects, as well as the method of data collection and the population being studied [28,29,30,31,32]. While subjects included in population studies tend to underreport their weight and overestimate their height, other population-specific studies showed that underweight participants tend to over-report their weight, whereas overweight participants tend to underreport their weight [33, 34]. In contrast, our study found that 83% of patients declared a weight within 2 kg of their true weight and a height within 2 cm of their true height. This may have been due to the fact that our patients were undergoing therapy and therefore were more inclined to be aware of their height, weight, and weight change. These results are also comparable with the NutriNet-Santé study (n = 2514) which concluded that the PR-anthropometric data is valid enough to be used when studying associations of nutritional factors with anthropometrics and other health outcomes [30]. It is important to note, however, that although our data revealed only few outliers, our largest outliers were 12.8 kg (underestimated) and 14.9 kg (overestimated). The patient who underestimated his/her weight by 12 kg also reported a stable weight for the previous 3 months and had a measured BMI of 28.4 kg/m2. Therefore, this outlier may be explained by previous studies that report the tendency of patients who are overweight to under-sreport their weight [28]. The patient who overestimated his/her weight had lost 16 kg since receiving their diagnosis, indicating that perhaps this subject was unaware of the extent of the weight loss. It is therefore important to be aware that significant weight changes, as well as patient’s perception of their BMI may influence the accuracy of PR-weight. PR outliers for height consisted of only 3 overestimations by 8, 10, and 11 cm. While studies have shown that over-reporting height of 2.5–4.5 cm is common, this only partly explains the discrepancy [28, 35]. The accuracy of PR-height in the literature is more consistent than weight. In fact, several studies show that PR-height tend to be within 2.5 cm estimate true height and can be considered an accurate and valid method for collecting this data, even among cancer patients [28, 32, 35, 36].
Height and weight make up only a small part of nutrition assessments. Thus, it is also important to assess the accuracy of other PR-nutrition assessments in comparison to HCP generated assessments. Therefore, we chose to compare the results of the NRS-2002 HCP generated nutrition assessment to that of the PR-version assessed using the PG-SGA SF. Our results were consistent with previous studies, which found significant associations between the two assessments analyzing both the continuous and categorical scales [5, 37]. In fact, other validation studies conducted among cancer patients conclude that the PG-SGA SF shows a higher specificity and sensitivity compared to various HCP based assessments like the NRS-2002 [38,39,40]. As the PG-SGA SF shows consistent results for accuracy and comparative, if not better identification of malnutrition, we therefore agree that it is an appropriated method to assess PR-nutrition status, as it allows quick identification and prioritization of patients and takes the assessment burden off of the HCPs.
Our PR-data revealed patients reported a significant weight loss for all three cancer types going back six months. According to international consensus criteria, weight loss > 5% over the last 6 months is classified as cancer cachexia [41]. Both pancreatic and stomach cancer patients lost on average more than 6% of their weight over a 6-month period when compared with their current weight indicating a need for optimum oncological and general medical management. It should be noted that weight loss etiology is very complex, correlated with tumor location, size and depth, and the type and length multimodal therapy provided which could not be analyzed within the framework of this study. Additionally, the patients in our study were offered regular nutrition and psychological interventions which may indicate that patients who are not offered such support could fare worse [42,43,44]. Scientific data relating the accuracy of weight history recall is scarce. What little data exists suggest that patients can be unclear about the magnitude of their weight change. Unfortunately, we were not able to collect HCP height and weight measurements going back in time in order to validate or compare our findings. However, as weight loss is considered to be an independent prognostic factor for decreased survival in cancer patients, more data regarding the accuracy of PR-weight recall could potentially help physicians more confidently identify patients at risk [42, 45, 46].
We were further able to track to number of study participants who requested supportive care on the same day of the study. In total, 58/152 patients (38.2%) requested supportive care in the form of nutrition therapy. The patients who requested nutrition care had PG-SGA score that was on average 2.05 points higher than patients who did not request supportive care (95% CI = (− 0.2, 4.31); p = 0.074). Among the patients who were both distressed and malnourished (n = 43), almost half (48.8%) requested supportive care. These results may indicate that integrating assessment questionnaires based on PROMs into clinical routine assessments may empower patients to actively participate in their care.
Our study had several strengths and limitations. Firstly, we were able to include centers in two different countries indicating that the results are not limited to the infrastructures, resources, and patient population at a single center. This also indicates that the E-health platform is appropriate for use in multicenter and/or international studies. As all data in this study were generated at a single-point observational basis, we could not validate the results regarding the accuracy of weight history which could be considered a limitation. While cross-sectional studies such as ours are useful for gaining insights into a population and establishing evaluation parameters and processes for future studies, our design was not as stringent as in randomized controlled trials and should thus be regarded as such. Furthermore, the inclusion criteria did not limit patients in regard to cancer stage or type of therapy and could thus not account for such potentially confounding factors. As we did not collect any data regarding social status, we cannot determine if this may have played a role in our results. The fact that we excluded individuals with linguistic challenges may have resulted in the exclusion of individuals with a migrant background and could also be considered a limitation. Additionally, patients who agreed to participate in our study were more likely to be open to the idea of E-health tools which could have created an inherent bias in the data collection. As cancer patients undergoing treatment can also experience neuropathy, finger dexterity may have played a role in their need for assistance and could have affected our results. Lastly 12 patients (7%) listed their diagnosis as “other” yet were considered eligible for the study, although the primary tumor could not be retrospectively identified we can say with confidence that it must have been one of the three types listed in our inclusion criteria.
Conclusion
Nutrition status, distress status, symptoms, and weight history are inter-related and are all known to affect clinical outcomes. The use of PROMs obtained via E-health platforms to identify these factors could not only ease the burden of HCPs who carry out these assessments but potentially lead to early identification and treatment while simultaneously encouraging patient participation. These results support the accuracy and clinical utility of PROMs when compared to HCP generated data. Randomized controlled trials using digital platforms to obtain PROMs followed up with provision of early professional psycho-social and nutrition interventions for cancer patients at risk for malnutrition and/or distress should be conducted.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
CANKADO is approved as an active Class I medical device within the European Union (registration number DE / CA59 / 11,976/2017) and is compliant with the FDA classification for Mobile Medical Devices (2015 Appendix B).
Abbreviations
- BMI:
-
Body mass index
- CI:
-
Confidence interval
- CM:
-
Centimeters
- DT:
-
Distress thermometer
- ESPEN:
-
European Society for Clinical Nutrition and Metabolism
- HCP:
-
Healthcare professional
- Inv. OR:
-
Inverse odds ratio
- NCCN:
-
National Comprehensive Cancer Network
- NRS-2002:
-
Nutrition Risk Score
- KG:
-
Kilos
- PG-SGA SF:
-
Patient Generated Subjective Global Assessment Short Form
- PR:
-
Patient-reported
- PROMs:
-
Patient-reported outcome measures
- QoL:
-
Quality of life
- SGA:
-
Subjective global assessment
References
van Bokhorst-de van der Schueren MAE, Guaitoli PR, Jansma EP, de Vet HCW (2014) Nutrition screening tools: Does one size fit all? A systematic review of screening tools for the hospital setting. Clin Nutr 33(1):39–58. https://doi.org/10.1016/j.clnu.2013.04.008
Faithfull S, Turner L, Poole K, Joy M, Manders R, Weprin J et al (2019) Prehabilitation for adults diagnosed with cancer: A systematic review of long-term physical function, nutrition and patient-reported outcomes. Eur J Cancer Care (Engl) 28(4):e13023. https://doi.org/10.1111/ecc.13023
Tran K, Zomer S, Chadder J, Earle C, Fung S, Liu J et al (2018) Measuring patient-reported outcomes to improve cancer care in Canada: an analysis of provincial survey data. Curr Oncol 25(2):176–179. https://doi.org/10.3747/co.25.3995
Chrischilles EA, Hourcade JP, Doucette W, Eichmann D, Gryzlak B, Lorentzen R et al (2014) Personal health records: a randomized trial of effects on elder medication safety. J Am Med Inform Assoc 21(4):679–686. https://doi.org/10.1136/amiajnl-2013-002284
Abbott J, Teleni L, McKavanagh D, Watson J, McCarthy AL, Isenring E (2016) Patient-Generated Subjective Global Assessment Short Form (PG-SGA SF) is a valid screening tool in chemotherapy outpatients. Support Care Cancer 24(9):3883–3887. https://doi.org/10.1007/s00520-016-3196-0
Riba MB, Donovan KA, Andersen B, Braun I, Breitbart WS, Brewer BW et al (2019) Distress Management, Version 3.2019, NCCN Clinical Practice Guidelines in Oncology. J Natl Compre Cancer Netw 17(10):1229–49. https://doi.org/10.6004/jnccn.2019.0048
Erickson N, Vogt L, Kolm A, Norman K, Fey T, Schiffler V et al (2018) Linguistic and content validation of the german scored patient-generated subjective global assessment for the austrian, german and swiss setting. Clin Nutr 37:S125. https://doi.org/10.1016/j.clnu.2018.06.1470
Leser MLN, Bergerson S, Trujillo E (2018) Oncology Nutrition for Clinical Practice, 1st edn. Academy of Nutrition & Dietetics, Chicago
Sealy MJ, Hass U, Ottery FD, van der Schans CP, Roodenburg JLN, Jager-Wittenaar H (2018) Translation and cultural adaptation of the scored patient-generated subjective global assessment: an interdisciplinary nutritional instrument appropriate for Dutch cancer patients. Cancer Nurs 41(6):450–462. https://doi.org/10.1097/NCC.0000000000000505
Nitichai N, Angkatavanich J, Somlaw N, Voravud N, Lertbutsayanukul C (2019) Validation of the Scored Patient-Generated Subjective Global Assessment (PG-SGA) in Thai setting and association with nutritional parameters in cancer patients. Asian Pac J Cancer Prev 20(4):1249–1255. https://doi.org/10.31557/APJCP.2019.20.4.1249
De Groot LM, Lee G, Ackerie A, van der Meij BS (2020) Malnutrition Screening and Assessment in the Cancer Care Ambulatory Setting: Mortality Predictability and Validity of the Patient-Generated Subjective Global Assessment Short form (PG-SGA SF) and the GLIM Criteria. Nutrients 12(8). https://doi.org/10.3390/nu12082287
Erickson N, Storck LJ, Kolm A, Norman K, Fey T, Schiffler V et al (2019) Tri-country translation, cultural adaptation, and validity confirmation of the Scored Patient-Generated Subjective Global Assessment. Support Care Cancer 27(9):3499–3507. https://doi.org/10.1007/s00520-019-4637-3
Abbott J, Teleni L, McKavanagh D, Watson J, McCarthy AL, Isenring E (2016) Patient-Generated Subjective Global Assessment Short Form (PG-SGA SF) is a valid screening tool in chemotherapy outpatients. Support Care Cancer 24(9):3883–3887. https://doi.org/10.1007/s00520-016-3196-0
Vigano AL, di Tomasso J, Kilgour RD, Trutschnigg B, Lucar E, Morais JA et al (2014) The abridged patient-generated subjective global assessment is a useful tool for early detection and characterization of cancer cachexia. J Acad Nutr Diet 114(7):1088–1098. https://doi.org/10.1016/j.jand.2013.09.027
Kondrup J, Allison SP, Elia M, Vellas B, Plauth M (2003) ESPEN guidelines for nutrition screening 2002. Clin Nutr 22(4):415–421. https://doi.org/10.1016/s0261-5614(03)00098-0
Cutillo A, O’Hea E, Person S, Lessard D, Harralson T, Boudreaux E (2017) The distress thermometer: cutoff points and clinical use. Oncol Nurs Forum 44(3):329–336. https://doi.org/10.1188/17.ONF.329-336
Wüller J, Küttner S, Foldenauer AC, Rolke R, Pastrana T (2016) Accuracy of the distress thermometer for home care patients with palliative care needs in Germany. Palliat Support Care 15(3):288–294. https://doi.org/10.1017/S1478951516000699
Richtering SS, Morris R, Soh SE, Barker A, Bampi F, Neubeck L et al (2017) Examination of an eHealth literacy scale and a health literacy scale in a population with moderate to high cardiovascular risk: Rasch analyses. PLoS ONE 12(4):e0175372. https://doi.org/10.1371/journal.pone.0175372
Hennemann S, Beutel ME, Zwerenz R (2017) Ready for eHealth? Health professionals’ acceptance and adoption of ehealth interventions in inpatient routine care. J Health Commun 22(3):274–284. https://doi.org/10.1080/10810730.2017.1284286
Bland JM, Altman DG (1986) Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1(8476):307–310
Zhu C, Wang B, Gao Y, Ma X (2018) Prevalence and relationship of malnutrition and distress in patients with Cancer using questionnaires. BMC Cancer 18(1):1272. https://doi.org/10.1186/s12885-018-5176-x
Bjoernes CD, Laursen BS, Delmar C, Cummings E, Nohr C (2012) A dialogue-based Web application enhances personalized access to healthcare professionals–an intervention study. BMC Med Inform Decis Mak 12:96. https://doi.org/10.1186/1472-6947-12-96
Girgis A, Durcinoska I, Levesque JV, Gerges M, Sandell T, Arnold A et al (2017) eHealth System for collecting and utilizing Patient Reported Outcome Measures for Personalized Treatment and Care (PROMPT-Care) among cancer patients: mixed methods approach to evaluate feasibility and acceptability. J Med Internet Res 19(10):e330. https://doi.org/10.2196/jmir.8360
Abernethy AP, Herndon JE, Wheeler JL, Day JM, Hood L, Patwardhan M et al (2009) Feasibility and acceptability to patients of a longitudinal system for evaluating cancer-related symptoms and quality of life: pilot study of an e/tablet data-collection system in academic oncology. J Pain Symptom Manage 37(6):1027–1038. https://doi.org/10.1016/j.jpainsymman.2008.07.011
Mullen KH, Berry DL, Zierler BK (2004) Computerized symptom and quality-of-life assessment for patients with cancer part II: acceptability and usability. Oncol Nurs Forum 31(5):E84–E89. https://doi.org/10.1188/04.onf.e84-e89
Jansen F, van Uden-Kraan CF, van Zwieten V, Witte BI, Verdonck-de Leeuw IM (2015) Cancer survivors’ perceived need for supportive care and their attitude towards self-management and eHealth. Support Care Cancer 23(6):1679–1688. https://doi.org/10.1007/s00520-014-2514-7
Steele R, Lo A, Secombe C, Wong YK (2009) Elderly persons’ perception and acceptance of using wireless sensor networks to assist healthcare. Int J Med Informatics 78(12):788–801. https://doi.org/10.1016/j.ijmedinf.2009.08.001
Gorber SC, Tremblay M, Moher D, Gorber B (2007) A comparison of direct vs. self-report measures for assessing height, weight and body mass index: a systematic review. Obes Rev 8(4):307–26. https://doi.org/10.1111/j.1467-789X.2007.00347.x
Spencer EA, Appleby PN, Davey GK, Key TJ (2002) Validity of self-reported height and weight in 4808 EPIC-Oxford participants. Public Health Nutr 5(4):561–565. https://doi.org/10.1079/phn2001322
Touvier M, Méjean C, Kesse-Guyot E, Pollet C, Malon A, Castetbon K et al (2010) Comparison between web-based and paper versions of a self-administered anthropometric questionnaire. Eur J Epidemiol 25(5):287–296. https://doi.org/10.1007/s10654-010-9433-9
McAdams MA, Van Dam RM, Hu FB (2007) Comparison of self-reported and measured BMI as correlates of disease markers in US adults. Obesity (Silver Spring, Md) 15(1):188–196. https://doi.org/10.1038/oby.2007.504
Villarini M, Acito M, Gianfredi V, Berrino F, Gargano G, Somaini M et al (2019) Validation of self-reported anthropometric measures and body mass index in a subcohort of the DianaWeb population study. Clin Breast Cancer 19(4):e511–e518. https://doi.org/10.1016/j.clbc.2019.04.008
Dauphinot V, Wolff H, Naudin F, Guéguen R, Sermet C, Gaspoz JM et al (2009) New obesity body mass index threshold for self-reported data. J Epidemiol Community Health 63(2):128–132. https://doi.org/10.1136/jech.2008.077800
Hercberg S, Castetbon K, Czernichow S, Malon A, Mejean C, Kesse E et al (2010) The Nutrinet-Santé Study: a web-based prospective study on the relationship between nutrition and health and determinants of dietary patterns and nutritional status. BMC Public Health 10:242. https://doi.org/10.1186/1471-2458-10-242
Briot K, Legrand E, Pouchain D, Monnier S, Roux C (2010) Accuracy of patient-reported height loss and risk factors for height loss among postmenopausal women. CMAJ 182(6):558–562. https://doi.org/10.1503/cmaj.090710
Stehman CR, Buckley RG, Dos Santos FL, Riffenburgh RH, Swenson A, Mulligan S et al (2011) Bedside Estimation of Patient Height for Calculating Ideal Body Weight in the Emergency Department. J Emerg Med 41(1):97–101. https://doi.org/10.1016/j.jemermed.2009.12.016
Du H, Liu B, Xie Y, Liu J, Wei Y, Hu H et al (2017) Comparison of different methods for nutrition assessment in patients with tumors. Oncol Lett 14(1):165–170. https://doi.org/10.3892/ol.2017.6154
Håkonsen SJ, Pedersen PU, Bath-Hextall F, Kirkpatrick P (2015) Diagnostic test accuracy of nutritional tools used to identify undernutrition in patients with colorectal cancer: a systematic review. JBI Database Syst Rev Implement Rep 13(4):141–187. https://doi.org/10.11124/jbisrir-2015-1673
Mendes NP, Barros TA, Rosa COB, Franceschini S (2019) Nutritional Screening Tools Used and Validated for Cancer Patients: A Systematic Review. Nutr Cancer 71(6):898–907. https://doi.org/10.1080/01635581.2019.1595045
Zhang YH, Xie FY, Chen YW, Wang HX, Tian WX, Sun WG et al (2018) Evaluating the nutritional status of oncology patients and its association with quality of life. Biomed Environ Sci 31(9):637–644. https://doi.org/10.3967/bes2018.088
Fearon K, Strasser F, Anker SD, Bosaeus I, Bruera E, Fainsinger RL et al (2011) Definition and classification of cancer cachexia: an international consensus. Lancet Oncol 12(5):489–495. https://doi.org/10.1016/s1470-2045(10)70218-7
Pressoir M, Desne S, Berchery D, Rossignol G, Poiree B, Meslier M et al (2010) Prevalence, risk factors and clinical implications of malnutrition in French comprehensive cancer centres. Br J Cancer 102(6):966–971. https://doi.org/10.1038/sj.bjc.6605578
Nemer L, Krishna SG, Shah ZK, Conwell DL, Cruz-Monserrate Z, Dillhoff M et al (2017) Predictors of pancreatic cancer-associated weight loss and nutritional interventions. Pancreas 46(9):1152–1157. https://doi.org/10.1097/MPA.0000000000000898
Yoon SL, Kim JA, Kelly DL, Lyon D, George TJ Jr (2019) Predicting unintentional weight loss in patients with gastrointestinal cancer. J Cachexia Sarcopenia Muscle 10(3):526–535. https://doi.org/10.1002/jcsm.12398
Ross PJ, Ashley S, Norton A, Priest K, Waters JS, Eisen T et al (2004) Do patients with weight loss have a worse outcome when undergoing chemotherapy for lung cancers? Br J Cancer 90(10):1905–1911. https://doi.org/10.1038/sj.bjc.6601781
Martin L, Senesse P, Gioulbasanis I, Antoun S, Bozzetti F, Deans C et al (2015) Diagnostic criteria for the classification of cancer-associated weight loss. J Clin Oncol 33(1):90–99. https://doi.org/10.1200/jco.2014.56.1894
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
N. Erickson: conceptualization, methodology, investigation, resources, data analysis and curation, entire writing process, visualization, and project administration. T. Schinkoethe: software, data curation, visualization, resources. C. Eckhardt: conceptualization, methodology, investigation, resources, data analysis, validation, review, and editing. L. Storck: project administration, investigation, review, and editing. A. Joos: investigation, review, and editing. L. Liu: resources, review, and editing. P.E. Ballmer: supervision, review, and editing. F. Mumm: resources, review, and Editing. T. Fey: conceptualization, review and editing, resources, project administration, and supervision. V. Heinemann: review and editing conceptualization, resources, visualization, validation, and supervision.
Corresponding author
Ethics declarations
Ethics approval
The methodology and all questionnaires utilized in this study were approved by the Human Research Ethics committee of the Ludwig Maximillian University of Munich in Germany and the Cantonal Ethics Committee Zurich, Switzerland (Reference number LMU19-954).
Informed consent to participate and/or consent to publish
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
L. Liu acknowledges the support of China Scholarship Council (No. 201908080031). T. Schinköthe is owner and Managing Director of CANKADO and owns stock in the company. N. Erickson has received honoraria for participating in symposia for CSL Behring, Fresenius, Baxter, Havas Lynx Group, Nutricia and GHD. V. Heinemann has received honoraria for participating in symposia and advisory boards for Merck, Amgen, Roche, Sanofi, Boehringer Ingelheim, Celgene, Sirtex Medical, Baxalta, SERVIER, Halozyme, MSD, Bristol-Myers Squibb, MSD Oncology, Lilly and Taiho Pharmaceutical. He has also received research funding from Amgen, Roche, Merck, Celgene, Boehringer Ingelheim, Sirtex Medical, Shire and Servier and travel support from Merck, Roche, Sirtex Medical, Amgen, SERVIER, Shire, MSD and Bristol-Myers Squibb. None of these activities were related to the content of this article. All remaining authors declared no conflicts of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Erickson, N., Schinkoethe, T., Eckhardt, C. et al. Patient-reported outcome measures obtained via E-Health tools ease the assessment burden and encourage patient participation in cancer care (PaCC Study). Support Care Cancer 29, 7715–7724 (2021). https://doi.org/10.1007/s00520-021-06351-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-021-06351-1