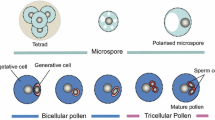
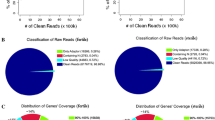
Key message
Bioinformatics for Pollen.
Abstract
Pollen plays a key role in crop production, and its development is the most delicate phase in reproduction. Different metabolic pathways are involved in pollen development, and changes in the level of some metabolites, as well as responses to stress, are correlated with the reduction in pollen viability, leading consequently to a decrease in the fruit production. However, studies on pollen may be hard because gamete development and fertilization are complex processes that occur during a short window of time. The rise of the so-called -omics sciences provided key strategies to promote molecular research in pollen tissues, starting from model organisms and moving to increasing number of species. An integrated multi-level approach based on investigations from genomics, transcriptomics, proteomics and metabolomics appears now feasible to clarify key molecular processes in pollen development and viability. To this aim, bioinformatics has a fundamental role for data production and analysis, contributing varied and ad hoc methodologies, endowed with different sensitivity and specificity, necessary for extracting added-value information from the large amount of molecular data achievable. Bioinformatics is also essential for data management, organization, distribution and integration in suitable resources. This is necessary to catch the biological features of the pollen tissues and to design effective approaches to identifying structural or functional properties, enabling the modeling of the major involved processes in normal or in stress conditions. In this review, we provide an overview of the available bioinformatics resources for pollen, ranging from raw data collections to complete databases or platforms, when available, which include data and/or results from -omics efforts on the male gametophyte. Perspectives in the fields will also be described.


Similar content being viewed by others
References
Abdul-Baki AA, Stommel JR (1995) Pollen viability and fruit set of tomato genotypes under optimum and high-temperature regimes. Hortscience 30:115–117
Alexandersson M, Cawley S, Pachter L (2003) SLAM: cross-species gene finding and alignment with a generalized pair hidden Markov model. Genome Res 13:496–502
Anders S, Huber W (2010) Differential expression analysis for sequence count data. Genome Biol 11:R106. doi:10.1186/gb-2010-11-10-r106
Arnold K, Bordoli L, Kopp J, Schwede T (2006) The SWISS-MODEL workspace: a web-based environment for protein structure homology modelling. Bioinformatics 22:195–201
Bandyopadhyay S, Pal SK (2007) Classification and learning using genetic algorithms: applications in bioinformatics and web intelligence. Springer Science & Business Media, Berlin
Becker JD, Boavida LC, Carneiro J, Haury M, Feijó JA (2003) Transcriptional profiling of arabidopsis tissues reveals the unique characteristics of the pollen transcriptome. Plant Physiol 133:713–725. doi:10.1104/pp.103.028241
Bita CE, Zenoni S, Vriezen WH, Mariani C, Pezzotti M, Gerats T (2011) Temperature stress differentially modulates transcription in meiotic anthers of heat-tolerant and heat-sensitive tomato plants. BMC Genom 12:384. doi:10.1186/1471-2164-12-384
Blunsom P (2004) Hidden markov models Lecture notes, August 15, p 18–19
Boguski MS, Lowe TM, Tolstoshev CM (1993) dbEST–database for “expressed sequence tags”. Nat Genet 4:332–333. doi:10.1038/ng0893-332
Bokszczanin KL, Fragkostefanakis S (2013) Perspectives on deciphering mechanisms underlying plant heat stress response and thermotolerance. Front Plant Sci 4:315. doi:10.3389/fpls.2013.00315
Bokszczanin K et al (2015) Identification of novel small ncRNAs in pollen of tomato. BMC Genom 16:714
Bokvaj P, Hafidh S, Honys D (2015) Transcriptome profiling of male gametophyte development in Nicotiana tabacum. Genom Data 3:106–111. doi:10.1016/j.gdata.2014.12.002
Bolser D, Staines DM, Pritchard E, Kersey P (2016) Ensembl plants: integrating tools for visualizing, mining, and analyzing plant genomics data. Methods Mol Biol (Clifton, NJ) 1374:115–140. doi:10.1007/978-1-4939-3167-5_6
Borges F, Pereira PA, Slotkin RK, Martienssen RA, Becker JD (2011) MicroRNA activity in the Arabidopsis male germline. J Exp Bot 62:1611–1620. doi:10.1093/jxb/erq452
Bostan H, Chiusano ML (2015) NexGenEx-Tom: a gene expression platform to investigate the functionalities of the tomato genome. BMC Plant Biol 15:48. doi:10.1186/s12870-014-0412-2
Brown GR et al (2015) Gene: a gene-centered information resource at NCBI. Nucleic Acids Res 43:D36–D42. doi:10.1093/nar/gku1055
Burge C, Karlin S (1997) Prediction of complete gene structures in human genomic DNA. J Mol Biol 268:78–94
Camacho C, Coulouris G, Avagyan V, Ma N, Papadopoulos J, Bealer K, Madden TL (2009) BLAST+: architecture and applications. BMC Bioinform 10:421. doi:10.1186/1471-2105-10-421
Caspi R et al (2014) The MetaCyc database of metabolic pathways and enzymes and the BioCyc collection of Pathway/Genome Databases. Nucleic Acids Res 42:D459–D471. doi:10.1093/nar/gkt1103
Chaturvedi P, Ischebeck T, Egelhofer V, Lichtscheidl I, Weckwerth W (2013) Cell-specific analysis of the tomato pollen proteome from pollen mother cell to mature pollen provides evidence for developmental priming. J Proteome Res 12:4892–4903. doi:10.1021/pr400197p
Chen F, Mackey AJ, Vermunt JK, Roos DS (2007) Assessing performance of orthology detection strategies applied to eukaryotigenomes. PLoS One 2:e383. doi:10.1371/journal.pone.0000383
Chettoor AM et al (2014) Discovery of novel transcripts and gametophytic functions via RNA-seq analysis of maize gametophytic transcriptomes. Genome Biol 15:414. doi:10.1186/s13059-014-0414-2
Cheung AY, Wu HM (2008) Structural and signaling networks for the polar cell growth machinery in pollen tubes. Annu Rev Plant Biol 59:547–572. doi:10.1146/annurev.arplant.59.032607.092921
Childs KL et al (2007) The TIGR plant transcript assemblies database. Nucleic Acids Res 35:D846–D851. doi:10.1093/nar/gkl785
Chiusano ML (2015) On the multifaceted aspects of bioinformatics in the next generation era: the run that must keep the quality. Next Gener Seq Appl 2:e106
Chiusano ML et al (2008) ISOL@: an Italian SOL Anaceae genomics resource. BMC Bioinform 9 Suppl(2):S7. doi:10.1186/1471-2105-9-s2-s7
Chou K-C, Cai Y-D (2002) Using functional domain composition and support vector machines for prediction of protein subcellular location. J Biol Chem 277:45765–45769
Colaco R, Moreno N, Feijo JA (2012) On the fast lane: mitochondria structure, dynamics and function in growing pollen tubes. J Microsc 247:106–118. doi:10.1111/j.1365-2818.2012.03628.x
Cole C, Barber JD, Barton GJ (2008) The Jpred 3 secondary structure prediction server. Nucleic Acids Res 36:W197–W201
Conesa A, Gotz S (2008) Blast2GO: a comprehensive suite for functional analysis in plant genomics. Int J Plant Genom 2008:619832. doi:10.1155/2008/619832
Cortes C, Vapnik V (1995) Support-vector networks. Mach Learn 20:273–297
D’Agostino N, Aversano M, Chiusano ML (2005) ParPEST: a pipeline for EST data analysis based on parallel computing. BMC Bioinform 6 Suppl(4):S9. doi:10.1186/1471-2105-6-s4-s9
D’Agostino N, Aversano M, Frusciante L, Chiusano ML (2007a) TomatEST database: in silico exploitation of EST data to explore expression patterns in tomato species. Nucleic Acids Res 35:D901–D905. doi:10.1093/nar/gkl921
D’Agostino N, Pizzichini D, Chiusano ML, Giuliano G (2007b) An EST database from saffron stigmas. BMC Plant Biol 7:53. doi:10.1186/1471-2229-7-53
D’Agostino N, Traini A, Frusciante L, Chiusano ML (2009) SolEST database: a “one-stop shop” approach to the study of Solanaceae transcriptomes. BMC Plant Biol 9:142. doi:10.1186/1471-2229-9-142
Dai S, Li L, Chen T, Chong K, Xue Y, Wang T (2006) Proteomic analyses of Oryza sativa mature pollen reveal novel proteins associated with pollen germination and tube growth. Proteomics 6:2504–2529. doi:10.1002/pmic.200401351
Davidson RM et al (2011) Utility of RNA sequencing for analysis of maize reproductive transcriptomes. Plant Genome 4:191–203. doi:10.3835/plantgenome2011.05.0015
Di Salle P, Incerti G, Colantuono C, Chiusano ML (2016) Gene co-expression analyses: an overview from microarray collections in Arabidopsis thaliana. Briefings Bioinform. doi:10.1093/bib/bbw002
Du Z, Zhou X, Ling Y, Zhang Z, Su Z (2010) agriGO: a GO analysis toolkit for the agricultural community. Nucleic Acids Res 38:W64–W70. doi:10.1093/nar/gkq310
Duvick J et al (2008) PlantGDB: a resource for comparative plant genomics. Nucleic Acids Res 36:D959–D965. doi:10.1093/nar/gkm1041
Edgar R, Domrachev M, Lash AE (2002) Gene Expression Omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res 30:207–210
English AC, Patel KS, Loraine AE (2010) Prevalence of alternative splicing choices in Arabidopsis thaliana. BMC Plant Biol 10:102. doi:10.1186/1471-2229-10-102
Fasoli M et al (2012) The grapevine expression atlas reveals a deep transcriptome shift driving the entire plant into a maturation program. Plant Cell 24:3489–3505. doi:10.1105/tpc.112.100230
Feng X, Zilberman D, Dickinson H (2013) A conversation across generations: soma-germ cell crosstalk in plants. Dev Cell 24:215–225. doi:10.1016/j.devcel.2013.01.014
Fernandez-Pozo N et al (2015) The Sol Genomics Network (SGN)–from genotype to phenotype to breeding. Nucleic Acids Res 43:D1036–D1041. doi:10.1093/nar/gku1195
Finn RD et al (2006) Pfam: clans, web tools and services. Nucleic Acids Res 34:D247–D251
Firon N et al (2006) Pollen grains of heat tolerant tomato cultivars retain higher carbohydrate concentration under heat stress conditions. Sci Hortic 109:212–217. doi:10.1016/j.scienta.2006.03.007
Foll M, Gaggiotti O (2008) A genome-scan method to identify selected loci appropriate for both dominant and codominant markers: a Bayesian perspective. Genetics 180:977–993
Fondevilla S, Krezdorn N, Rotter B, Kahl G, Winter P (2015) In planta identification of putative pathogenicity factors from the chickpea pathogen Ascochyta rabiei by de novo transcriptome sequencing using RNA-seq and massive analysis of cDNA ends. Front Microbiol 6:1329. doi:10.3389/fmicb.2015.01329
Fu WJ, Carroll RJ, Wang S (2005) Estimating misclassification error with small samples via bootstrap cross-validation. Bioinformatics (Oxford, England) 21:1979–1986
Fukami-Kobayashi K, Nakamura Y, Tamura T, Kobayashi M (2014) SABRE2: a database connecting plant EST/full-length cDNA clones with Arabidopsis information. Plant Cell Physiol 55:e5. doi:10.1093/pcp/pct177
Fyfe R et al (2009) The European pollen database: past efforts and current activities. Veget Hist Archaeobot 18:417–424. doi:10.1007/s00334-009-0215-9
Giorno F, Wolters-Arts M, Mariani C, Rieu I (2013) Ensuring reproduction at high temperatures: the heat stress response during anther and pollen. Dev Plants 2:489
Goff SA et al (2002) A draft sequence of the rice genome (Oryza sativa L. ssp. japonica). Science 296:92–100. doi:10.1126/science.1068275
Goldberg DE, Holland JH (1988) Genetic algorithms and machine learning. Mach Learn 3:95–99
Goodstein DM et al (2012) Phytozome: a comparative platform for green plant genomics. Nucleic Acids Res 40:D1178–D1186. doi:10.1093/nar/gkr944
Grabherr MG et al (2011) Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat Biotechnol 29:644–652. doi:10.1038/nbt.1883
Grant-Downton R, Hafidh S, Twell D, Dickinson HG (2009) Small RNA pathways are present and functional in the angiosperm male gametophyte. Mol Plant 2:500–512. doi:10.1093/mp/ssp003
Grobei MA et al (2009) Deterministic protein inference for shotgun proteomics data provides new insights into Arabidopsis pollen development and function. Genome Res 19:1786–1800. doi:10.1101/gr.089060.108
Haerizadeh F, Wong CE, Bhalla PL, Gresshoff PM, Singh MB (2009) Genomic expression profiling of mature soybean (Glycine max) pollen. BMC Plant Biol 9:25. doi:10.1186/1471-2229-9-25
Hafidh S, Breznenova K, Ruzicka P, Fecikova J, Capkova V, Honys D (2012) Comprehensive analysis of tobacco pollen transcriptome unveils common pathways in polar cell expansion and underlying heterochronic shift during spermatogenesis. BMC Plant Biol 12:24. doi:10.1186/1471-2229-12-24
Herrero MP, Johnson RR (1980) High temperature stress and pollen viability of maize. Crop Sci 20:796–800. doi:10.2135/cropsci1980.0011183X002000060030x
Hirano H-Y, Sano Y (1991) Molecular characterization of the waxy locus of rice (Oryza sativa). Plant Cell Physiol 32:989–997
Hobo T et al (2008) Various spatiotemporal expression profiles of anther-expressed genes in rice. Plant Cell Physiol 49:1417–1428. doi:10.1093/pcp/pcn128
Hollender CA, Kang C (2014) Floral transcriptomes in woodland strawberry uncover developing receptacle and anther gene networks. Plant Physiol 165:1062–1075
Holmes-Davis R, Tanaka CK, Vensel WH, Hurkman WJ, McCormick S (2005) Proteome mapping of mature pollen of Arabidopsis thaliana. Proteomics 5:4864–4884. doi:10.1002/pmic.200402011
Honys D, Twell D (2003) Comparative analysis of the Arabidopsis pollen transcriptome. Plant Physiol 132:640–652. doi:10.1104/pp.103.020925
Honys D, Twell D (2004) Transcriptome analysis of haploid male gametophyte development in Arabidopsis. Genome Biol 5:R85. doi:10.1186/gb-2004-5-11-r85
Huang X, Madan A (1999) CAP3: A DNA sequence assembly program. Genome Res 9:868–877
Jaillon O et al (2007) The grapevine genome sequence suggests ancestral hexaploidization in major angiosperm phyla. Nature 449:463–467. doi:10.1038/nature06148
Ke T, Yu J, Dong C, Mao H, Hua W, Liu S (2015) ocsESTdb: a database of oil crop seed EST sequences for comparative analysis and investigation of a global metabolic network and oil accumulation metabolism. BMC Plant Biol 15:19. doi:10.1186/s12870-014-0399-8
Kerim T, Imin N, Weinman JJ, Rolfe BG (2003) Proteome analysis of male gametophyte development in rice anthers. Proteomics 3:738–751. doi:10.1002/pmic.200300424
Kim SY, Hong CB, Lee I (2001) Heat shock stress causes stage-specific male sterility in Arabidopsis thaliana. J Plant Res 114:301–307. doi:10.1007/PL00013991
Kim HJ et al (2008) Pepper EST database: comprehensive in silico tool for analyzing the chili pepper (Capsicum annuum) transcriptome. BMC Plant Biol 8:101. doi:10.1186/1471-2229-8-101
Kodama Y, Shumway M, Leinonen R (2012) The sequence read archive: explosive growth of sequencing data. Nucleic Acids Res 40:D54–D56. doi:10.1093/nar/gkr854
Kolesnikov N et al (2015) ArrayExpress update–simplifying data submissions. Nucleic Acids Res 43:D1113–D1116. doi:10.1093/nar/gku1057
Kozomara A, Griffiths-Jones S (2014) miRBase: annotating high confidence microRNAs using deep sequencing data. Nucleic Acids Res 42:D68–D73. doi:10.1093/nar/gkt1181
Lamesch P et al (2012) The arabidopsis information resource (TAIR): improved gene annotation and new tools. Nucleic Acids Res 40:D1202–D1210. doi:10.1093/nar/gkr1090
Lang V, Usadel B, Obermeyer G (2015) De novo sequencing and analysis of the lily pollen transcriptome: an open access data source for an orphan plant species. Plant Mol Biol 87:69–80. doi:10.1007/s11103-014-0261-2
Langfelder P, Horvath S (2008) WGCNA: an R package for weighted correlation network analysis. BMC Bioinform 9:559. doi:10.1186/1471-2105-9-559
Langmead B, Salzberg SL (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9:357–359. doi:10.1038/nmeth.1923
Larranaga P et al (2006) Machine learning in bioinformatics. Briefings Bioinform 7:86–112
Lee JY, Lee DH (2003) Use of serial analysis of gene expression technology to reveal changes in gene expression in Arabidopsis pollen undergoing cold stress. Plant Physiol 132:517–529. doi:10.1104/pp.103.020511
Li C (2008) Automating dChip: toward reproducible sharing of microarray data analysis. BMC Bioinform 9:231. doi:10.1186/1471-2105-9-231
Li R, Li Y, Kristiansen K, Wang J (2008) SOAP: short oligonucleotide alignment program. Bioinformatics (Oxford, England) 24:713–714. doi:10.1093/bioinformatics/btn025
Li H et al (2009) The sequence alignment/map format and SAMtools. Bioinformatics (Oxford, England) 25:2078–2079. doi:10.1093/bioinformatics/btp352
Li XM, Sang YL, Zhao XY, Zhang XS (2013) High-throughput sequencing of small RNAs from pollen and silk and characterization of miRNAs as candidate factors involved in pollen-silk interactions in maize. PLoS One 8:e72852. doi:10.1371/journal.pone.0072852
Liu Y, Zhou J, White KP (2014) RNA-seq differential expression studies: more sequence or more replication? Bioinformatics (Oxford, England) 30:301–304. doi:10.1093/bioinformatics/btt688
Lohse M, Bolger AM, Nagel A, Fernie AR, Lunn JE, Stitt M, Usadel B (2012) RobiNA: a user-friendly, integrated software solution for RNA-Seq-based transcriptomics. Nucleic Acids Res 40:W622–W627. doi:10.1093/nar/gks540
Lohse M et al (2014) Mercator: a fast and simple web server for genome scale functional annotation of plant sequence data. Plant, Cell Environ 37:1250–1258. doi:10.1111/pce.12231
Lopez-Casado G et al (2012) Enabling proteomic studies with RNA-Seq: The proteome of tomato pollen as a test case. Proteomics 12:761–774. doi:10.1002/pmic.201100164
Loraine AE, McCormick S, Estrada A, Patel K, Qin P (2013) RNA-seq of Arabidopsis pollen uncovers novel transcription and alternative splicing. Plant Physiol 162:1092–1109. doi:10.1104/pp.112.211441
Loraine AE, Blakley IC, Jagadeesan S, Harper J, Miller G, Firon N (2015) Analysis and visualization of RNA-Seq expression data using RStudio, Bioconductor, and Integrated Genome Browser Methods in molecular biology (Clifton, NJ) 1284:481-501 doi:10.1007/978-1-4939-2444-8_24
Lu Y, Wei L, Wang T (2015) Methods to isolate a large amount of generative cells, sperm cells and vegetative nuclei from tomato pollen for “omics” analysis Frontiers in plant science 6:391 doi:10.3389/fpls.2015.00391
Lukashin AV, Borodovsky M (1998) GeneMark. hmm: new solutions for gene finding. Nucleic Acids Res 26:1107–1115
Ma J, Skibbe DS, Fernandes J, Walbot V (2008) Male reproductive development: gene expression profiling of maize anther and pollen ontogeny. Genome Biol 9:R181. doi:10.1186/gb-2008-9-12-r181
Martin D, Brun C, Remy E, Mouren P, Thieffry D, Jacq B (2004) GOToolBox: functional analysis of gene datasets based on Gene Ontology. Genome Biol 5:R101. doi:10.1186/gb-2004-5-12-r101
Matsui T, Omasa K (2002) Rice (Oryza sativa L.) Cultivars Tolerant to High Temperature at Flowering: Anther Characteristics. Ann Bot 89:683–687. doi:10.1093/aob/mcf112
McCarthy FM et al (2011) AgBase: supporting functional modeling in agricultural organisms. Nucleic Acids Res 39:D497–D506. doi:10.1093/nar/gkq1115
Misra BB, Assmann SM, Chen S (2014) Plant single-cell and single-cell-type metabolomics. Trends Plant Sci 19:637–646. doi:10.1016/j.tplants.2014.05.005
Morgenstern B (1999) DIALIGN 2: improvement of the segment-to-segment approach to multiple sequence alignment Bioinformatics (Oxford, England) 15:211-218
Moriya Y, Itoh M, Okuda S, Yoshizawa AC, Kanehisa M (2007) KAAS: an automatic genome annotation and pathway reconstruction server. Nucleic Acids Res 35:W182–W185. doi:10.1093/nar/gkm321
NCBI_Resource_Coordinators (2013) Database resources of the National Center for Biotechnology Information Nucleic acids research doi:10.1093/nar/gkt1146 www.ncbi.nlm.nih.gov
Noir S, Brautigam A, Colby T, Schmidt J, Panstruga R (2005) A reference map of the Arabidopsis thaliana mature pollen proteome. Biochem Biophys Res Commun 337:1257–1266. doi:10.1016/j.bbrc.2005.09.185
O’Leary NA et al. (2015) Reference sequence (RefSeq) database at NCBI: current status, taxonomic expansion, and functional annotation Nucleic acids research doi:10.1093/nar/gkv1189 http://www.ncbi.nlm.nih.gov/refseq
Obermeyer G, Fragner L, Lang V, Weckwerth W (2013) Dynamic adaption of metabolic pathways during germination and growth of lily pollen tubes after inhibition of the electron transport chain. Plant Physiol 162:1822–1833. doi:10.1104/pp.113.219857
Oo MM et al (2014) Evaluation of rice promoters conferring pollen-specific expression in a heterologous system, Arabidopsis. Plant Reprod 27:47–58. doi:10.1007/s00497-014-0239-x
Ottaviano E, Mulcahy DL (1989) Genetics of angiosperm pollen. In: John G. Scandalios TRFW, John GS (eds) Advances in genetics, vol 26. Academic Press, Cambridge, pp 1–64. doi:10.1016/S0065-2660(08)60222-9
Palanivelu R, Tsukamoto T (2012) Pathfinding in angiosperm reproduction: pollen tube guidance by pistils ensures successful double fertilization Wiley interdisciplinary reviews. Dev Biol 1:96–113. doi:10.1002/wdev.6
Paterson AH et al (2009) The Sorghum bicolor genome and the diversification of grasses. Nature 457:551–556. doi:10.1038/nature07723
Paupiere MJ, van Heusden AW, Bovy AG (2014) The metabolic basis of pollen thermo-tolerance: perspectives for breeding. Metabolites 4:889–920. doi:10.3390/metabo4040889
Peet MM, Sato S, Gardner RG (1998) Comparing heat stress effects on male-fertile and male-sterile tomatoes. Plant Cell Environ 21:225–231. doi:10.1046/j.1365-3040.1998.00281.x
Pertl H, Schulze WX, Obermeyer G (2009) The pollen organelle membrane proteome reveals highly spatial-temporal dynamics during germination and tube growth of lily pollen. J Proteome Res 8:5142–5152. doi:10.1021/pr900503f
Pina C, Pinto F, Feijó JA, Becker JD (2005) Gene family analysis of the arabidopsis pollen transcriptome reveals biological implications for cell growth, division control, and gene expression regulation. Plant Physiol 138:744–756. doi:10.1104/pp.104.057935
Ploeg A, Heuvelink E (2005) Influence of sub-optimal temperature on tomato growth and yield: a review. J Horticu Sci Biotechnol 80:652–659
Prasad PVV, Boote KJ, Allen LH Jr, Sheehy JE, Thomas JMG (2006) Species, ecotype and cultivar differences in spikelet fertility and harvest index of rice in response to high temperature stress. Field Crops Res 95:398–411. doi:10.1016/j.fcr.2005.04.008
Pressman E, Peet MM, Pharr DM (2002) The effect of heat stress on tomato pollen characteristics is associated with changes in carbohydrate concentration in the developing anthers. Ann Bot 90:631–636
Proost S, Van Bel M, Sterck L, Billiau K, Van Parys T, Van de Peer Y, Vandepoele K (2009) PLAZA: a comparative genomics resource to study gene and genome evolution in plants. Plant Cell 21:3718–3731. doi:10.1105/tpc.109.071506
Qin Y et al (2009) Penetration of the stigma and style elicits a novel transcriptome in pollen tubes, pointing to genes critical for growth in a pistil. PLoS Genet 5:e1000621. doi:10.1371/journal.pgen.1000621
R_Development_Core_Team (2011) R: a language and environment for statistical computing. R Foundation for Statistical Computing,
Redman JC, Haas BJ, Tanimoto G, Town CD (2004) Development and evaluation of an Arabidopsis whole genome Affymetrix probe array. Plant J Cell Mol Biol 38:545–561. doi:10.1111/j.1365-313X.2004.02061.x
Reimand J, Arak T, Vilo J (2011) g:Profiler–a web server for functional interpretation of gene lists (2011 update). Nucleic Acids Res 39:W307–W315. doi:10.1093/nar/gkr378
Richmond T, Somerville S (2000) Chasing the dream: plant EST microarrays. Curr Opin Plant Biol 3:108–116
Romualdi C, Bortoluzzi S, D’Alessi F, Danieli GA (2003) IDEG6: a web tool for detection of differentially expressed genes in multiple tag sampling experiments. Physiol Genom 12:159–162. doi:10.1152/physiolgenomics.00096.2002
Rost B, Yachdav G, Liu J (2004) The predictprotein server. Nucleic Acids Res 32:W321–W326
Rounds CM, Winship LJ, Hepler PK (2011) Pollen tube energetics: respiration, fermentation and the race to the ovule. AoB Plants 2011:plr019. doi:10.1093/aobpla/plr019
Rutley N, Twell D (2015) A decade of pollen transcriptomics. Plant Reprod 28:73–89. doi:10.1007/s00497-015-0261-7
Saeed AI et al (2003) TM4: a free, open-source system for microarray data management and analysis. Biotechniques 34:374–378
Saini H, Sedgley M, Aspinall D (1984) Development anatomy in wheat of male sterility induced by heat stress, water deficit or abscisic acid. Funct Plant Biol 11:243–253. doi:10.1071/PP9840243
Sanetomo R, Hosaka K (2013) Pollen transcriptome analysis of Solanum tuberosum (2n = 4x = 48), S. demissum (2n = 6x = 72), and their reciprocal F1 hybrids. Plant Cell Rep 32:623–636. doi:10.1007/s00299-013-1395-4
SAS_Institute (2003) The SAS system for windows version 9.1
Sato S, Kamiyama M, Iwata T, Makita N, Furukawa H, Ikeda H (2006) Moderate increase of mean daily temperature adversely affects fruit set of Lycopersicon esculentum by disrupting specific physiological processes in male reproductive development. Ann Bot 97:731–738. doi:10.1093/aob/mcl037
Schmid M et al (2005) A gene expression map of Arabidopsis thaliana development. Nat Genet 37:501–506. doi:10.1038/ng1543
Schnable PS et al (2009) The B73 maize genome: complexity, diversity, and dynamics. Science 326:1112–1115. doi:10.1126/science.1178534
Schrauwen JAM, de Groot PFM, van Herpen MMA, van der Lee T, Reynen WH, Weterings KAP, Wullems GJ (1990) Stage-related expression of mRNAs during pollen development in lily and tobacco. Planta 182:298–304. doi:10.1007/BF00197125
Schwab R, Palatnik JF, Riester M, Schommer C, Schmid M, Weigel D (2005) Specific effects of microRNAs on the plant transcriptome. Dev Cell 8:517–527. doi:10.1016/j.devcel.2005.01.018
Sheoran I, Sproule K, Olson DH, Ross AS, Sawhney V (2006) Proteome profile and functional classification of proteins in Arabidopsis thaliana (Landsberg erecta) mature pollen. Sex Plant Reprod 19:185–196. doi:10.1007/s00497-006-0035-3
Sheoran IS, Ross AR, Olson DJ, Sawhney VK (2007) Proteomic analysis of tomato (Lycopersicon esculentum) pollen. J Exp Bot 58:3525–3535. doi:10.1093/jxb/erm199
Sheoran IS, Pedersen EJ, Ross ARS, Sawhney VK (2009a) Dynamics of protein expression during pollen germination in canola (Brassica napus). Planta 230:779–793. doi:10.1007/s00425-009-0983-6
Sheoran IS, Ross AR, Olson DJ, Sawhney VK (2009b) Differential expression of proteins in the wild type and 7B-1 male-sterile mutant anthers of tomato (Solanum lycopersicum): a proteomic analysis. J Proteom 71:624–636. doi:10.1016/j.jprot.2008.10.006
Sierro N et al (2014) The tobacco genome sequence and its comparison with those of tomato and potato. Nat Commun. doi:10.1038/ncomms4833
Simel EJ, Saidak LR, Tuskan GA (1997) Method of extracting genomic DNA from non-germinated gymnosperm and angiosperm pollen. Biotechniques 22(390–392):394
Slotkin RK, Vaughn M, Borges F, Tanurdzic M, Becker JD, Feijo JA, Martienssen RA (2009) Epigenetic reprogramming and small RNA silencing of transposable elements in pollen. Cell 136:461–472. doi:10.1016/j.cell.2008.12.038
Sonnhammer EL, Eddy SR, Birney E, Bateman A, Durbin R (1998) Pfam: multiple sequence alignments and HMM-profiles of protein domains. Nucleic Acids Res 26:320–322
Soriano M, Li H, Boutilier K (2013) Microspore embryogenesis: establishment of embryo identity and pattern in culture. Plant Reprod 26:181–196. doi:10.1007/s00497-013-0226-7
Suwabe K et al (2008) Separated transcriptomes of male gametophyte and tapetum in rice: validity of a laser microdissection (LM) microarray. Plant Cell Physiol 49:1407–1416. doi:10.1093/pcp/pcn124
The_Arabidopsis_Genome_Initiative (2000) Analysis of the genome sequence of the flowering plant Arabidopsis thaliana. Nature 408:796–815. doi:10.1038/35048692
The_Tomato_Genome_Consortium (2012) The tomato genome sequence provides insights into fleshy fruit evolution. Nature 485:635–641
Thimm O et al (2004) MAPMAN: a user-driven tool to display genomics data sets onto diagrams of metabolic pathways and other biological processes. Plant J Cell Mol Biol 37:914–939
Thompson JD, Gibson T, Higgins DG (2002) Multiple sequence alignment using ClustalW and ClustalX. Curr Protocols Bioinform 2.3. 1–2.3. 22
Trapnell C, Pachter L, Salzberg SL (2009) TopHat: discovering splice junctions with RNA-Seq. Bioinformatics (Oxford, England) 25:1105–1111. doi:10.1093/bioinformatics/btp120
Trapnell C et al (2012) Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat Protoc 7:562–578. doi:10.1038/nprot.2012.016
Twell D (1994) The diversity and regulation of gene expression in the pathway of male gametophyte development—molecular and cellular aspects of plant reproduction. Cambridge University Press, Cambridge
Twell D (2011) Male gametogenesis and germline specification in flowering plants. Sex Plant Reprod 24:149–160. doi:10.1007/s00497-010-0157-5
Wang Y, Zhang W-Z, Song L-F, Zou J-J, Su Z, Wu W-H (2008) Transcriptome analyses show changes in gene expression to accompany pollen germination and tube growth in Arabidopsis. Plant Physiol 148:1201–1211. doi:10.1104/pp.108.126375
Wang Z, Gerstein M, Snyder M (2009) RNA-Seq: a revolutionary tool for transcriptomics. Nat Rev Genet 10:57–63. doi:10.1038/nrg2484
Wei LQ, Xu WY, Deng ZY, Su Z, Xue Y, Wang T (2010) Genome-scale analysis and comparison of gene expression profiles in developing and germinated pollen in Oryza sativa. BMC Genom 11:338. doi:10.1186/1471-2164-11-338
Wei LQ, Yan LF, Wang T (2011) Deep sequencing on genome-wide scale reveals the unique composition and expression patterns of microRNAs in developing pollen of Oryza sativa. Genome Biol 12:R53. doi:10.1186/gb-2011-12-6-r53
Whittle CA, Malik MR, Li R, Krochko JE (2010) Comparative transcript analyses of the ovule, microspore, and mature pollen in Brassica napus. Plant Mol Biol 72:279–299. doi:10.1007/s11103-009-9567-x
Wienkoop S, Staudinger C, Hoehenwarter W, Weckwerth W, Egelhofer V (2012) ProMEX—–a mass spectral reference database for plant proteomics. Front Plant Sci 3:125. doi:10.3389/fpls.2012.00125
Xie F, Sun G, Stiller JW, Zhang B (2011) Genome-wide functional analysis of the cotton transcriptome by creating an integrated EST database. PLoS One 6:e26980. doi:10.1371/journal.pone.0026980
Xu X et al (2011) Genome sequence and analysis of the tuber crop potato. Nature 475:189–195. doi:10.1038/nature10158
Yang H, Kaur N, Kiriakopolos S, McCormick S (2006) EST generation and analyses towards identifying female gametophyte-specific genes in Zea mays L. Planta 224:1004–1014. doi:10.1007/s00425-006-0283-3
Zajac BK, Amendt J, Horres R, Verhoff MA, Zehner R (2015) De novo transcriptome analysis and highly sensitive digital gene expression profiling of Calliphora vicina (Diptera: Calliphoridae) pupae using MACE (Massive Analysis of cDNA Ends) Forensic science international. Genetics 15:137–146. doi:10.1016/j.fsigen.2014.11.013
Zawada AM, Rogacev KS, Muller S, Rotter B, Winter P, Fliser D, Heine GH (2014) Massive analysis of cDNA Ends (MACE) and miRNA expression profiling identifies proatherogenic pathways in chronic kidney disease. Epigenetics 9:161–172. doi:10.4161/epi.26931
Zhang Y, Sun Y (2011) HMM-FRAME: accurate protein domain classification for metagenomic sequences containing frameshift errors. BMC Bioinform 12:198
Zhang L et al (2009) A genome-wide characterization of microRNA genes in maize. PLoS Genet 5:e1000716. doi:10.1371/journal.pgen.1000716
Zhou X, Su Z (2007) EasyGO: gene ontology-based annotation and functional enrichment analysis tool for agronomical species. BMC Genom 8:246. doi:10.1186/1471-2164-8-246
Zinn KE, Tunc-Ozdemir M, Harper JF (2010) Temperature stress and plant sexual reproduction: uncovering the weakest links. J Exp Bot 61:1959–1968. doi:10.1093/jxb/erq053
Zou J, Song L, Zhang W, Wang Y, Ruan S, Wu WH (2009) Comparative proteomic analysis of Arabidopsis mature pollen and germinated pollen. J Integr Plant Biol 51:438–455. doi:10.1111/j.1744-7909.2009.00823.x
Funding
This work is supported by the Solanaceae Pollen Thermotolerance—Marie Curie Initial Training Network project (Grant Agreement No. 289220). Luca Ambrosino is supported by the Genopom Pro and HORT Projects (Ministero dell’Istruzione, dell’Università e della Ricerca, Italy).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Enrico Schleiff.
Luca Ambrosino and Hamed Bostan have contributed equally to this work.
A contribution to the special issue ‘Pollen development and stress response’.
Rights and permissions
About this article
Cite this article
Ambrosino, L., Bostan, H., Ruggieri, V. et al. Bioinformatics resources for pollen. Plant Reprod 29, 133–147 (2016). https://doi.org/10.1007/s00497-016-0284-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00497-016-0284-8