Abstract
Pollen tubes are among the fastest tip-growing plant cells and represent an excellent experimental system for studying the dynamics and spatiotemporal control of polarized cell growth. However, investigating pollen tube tip growth in the model plant Arabidopsis remains difficult because in vitro pollen germination and pollen tube growth rates are highly variable and largely different from those observed in pistils, most likely due to growth-promoting properties of the female reproductive tract. We found that in vitro grown Arabidopsis pollen respond to brassinosteroid (BR) in a dose-dependent manner. Pollen germination and pollen tube growth increased nine- and fivefold, respectively, when media were supplemented with 10 µM epibrassinolide (epiBL), resulting in growth kinetics more similar to growth in vivo. Expression analyses show that the promoter of one of the key enzymes in BR biosynthesis, CYP90A1/CPD, is highly active in the cells of the reproductive tract that form the pathway for pollen tubes from the stigma to the ovules. Pollen tubes grew significantly shorter through the reproductive tract of a cyp90a1 mutant compared to the wild type, or to a BR perception mutant. Our results show that epiBL promotes pollen germination and tube growth in vitro and suggest that the cells of the reproductive tract provide BR compounds to stimulate pollen tube growth.





Similar content being viewed by others
Abbreviations
- BRs:
-
Brassinosteroids
- epiBL:
-
Epibrassinolide
- PGM:
-
Pollen germination medium
- CLSM:
-
Confocal laser scanning microscope
- GFP:
-
Green fluorescent protein
- NLS:
-
Nuclear localization signal
- ECM:
-
Extracellular matrix
- IQR:
-
Interquartile range
References
Alonso JM et al (2003) Genome-wide insertional mutagenesis of Arabidopsis thaliana. Science 301:653–657
Bajguz A (2007) Metabolism of brassinosteroids in plants. Plant Physiol Biochem 45(2):95–107
Bajguz A, Tretyn A (2003) The chemical characteristic and distribution of brassinosteroids in plants. Phytochemistry 62:1027–1046
Bancos S, Nomura T, Sato T, Molnár G, Bishop GJ, Koncz C, Yokota T, Nagy F, Szekeres M (2002) Regulation of transcript levels of the Arabidopsis cytochrome P450 genes involved in brassinosteroid biosynthesis. Plant Physiol 130:504–513
Bao F, Shen J, Brady SR, Muday GK, Asami T, Yang Z (2004) Brassinosteroids Interact with auxin to promote lateral root development in Arabidopsis. Plant Physiol 134:1624–1631
Beale KM, Johnson MA (2013) Speed dating, rejection, and finding the perfect mate: advice from flowering plants. Curr Opin Plant Biol 16:590–597
Boavida LC, McCormick S (2007) Temperature as a determinant factor for increased and reproducible in vitro pollen germination in Arabidopsis thaliana. Plant J 52:570–582
Bou Daher F, Chebli Y, Geitmann A (2009) Optimization of conditions for germination of cold-stored Arabidopsis thaliana pollen. Plant Cell Rep 28:347–357
Brewbaker JL, Kwach BH (1963) The essential role of calcium ion in pollen germination and pollen tube growth. Am J Bot 50(9):747–858
Caño-Delgado A, Yin Y, Yu C, Vafeados D, Mora-García S, Cheng J-C, Nam KH, Li J, Chory J (2004) BRL1 and BRL3 are novel brassinosteroid receptors that function in vascular differentiation in Arabidopsis. Development 131:5341–5351
Chae K, Lord EM (2011) Pollen tube growth and guidance: roles of small, secreted proteins. Ann Bot 108:627–636
Chen D, Zhao J (2008) Free IAA in stigmas and styles during pollen germination and pollen tube growth of Nicotiana tabacum. Physiol Plant 134:202–215
Cheung AY, Wu HM (2008) Structural and signaling networks for the polar cell growth machinery in pollen tubes. Annu Rev Plant Biol 59:547–572
Choe S, Dilkes BP, Fujioka S, Takatsuto S, Sakurai A, Feldmann KA (1998) The DWF4 gene of Arabidopsis encodes a cytochrome P450 That mediates multiple 22α-hydroxylation steps in brassinosteroid biosynthesis. Plant Cell 10:231–243
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Clouse SD (1996) Molecular genetic studies confirm the role of brassinosteroids in plant growth and development. Plant J 10:1–8
Clouse SD (2011) Brassinosteroids. Arabidopsis Book Am Soc Plant Biol 9:e0151
Crawford BC, Yanofsky MF (2008) The formation and function of the female reproductive tract in flowering plants. Curr Biol 18:972–978
Crawford BC, Yanofsky MF (2011) HALF FILLED promotes reproductive tract development and fertilization efficiency in Arabidopsis thaliana. Development 138:2999–3009
Crawford BC, Ditta G, Yanofsky MF (2007) The NTT gene is required for transmitting-tract development in carpels of Arabidopsis thaliana. Curr Biol 17:1101–1108
Dresselhaus T, Franklin-Tong N (2013) Male–female crosstalk during pollen germination, tube growth and guidance, and double fertilization. Mol Plant 6:1018–1036
Ephritikhine G, Fellner M, Vannini C, Lapous D, Barbier-Brygoo H (1999) The sax1 dwarf mutant of Arabidopsis thaliana shows altered sensitivity of growth responses to abscisic acid, auxin, gibberellins and ethylene and is partially rescued by exogenous brassinosteroids. Plant J 18:303–314
Fan LM, Wang YF, Wang H, Wu WH (2001) In vitro Arabidopsis pollen germination and characterization of the inward potassium currents in Arabidopsis pollen grain protoplasts. J Exp Bot 52:1603–1614
Friedrichsen DM, Nemhauser J, Muramitsu T, Maloof JN, Alonso J, Ecker JR, Furuya M, Chory J (2002) Three redundant brassinosteroid early response genes encode putative bHLH transcription factors required for normal growth. Genetics 162:1445–1456
Fujioka S, Sakurai A (1997) Brassinosteroids. Nat Prod Rep 14:1–10
Fujioka S, Yokota T (2003) Biosynthesis and metabolism of brassinosteroids. Annu Rev Plant Biol 54:137–164
Gebert M, Dresselhaus T, Sprunck S (2008) F-actin organization and pollen tube tip growth in Arabidopsis are dependent on the gametophyte-specific Armadillo repeat protein ARO1. Plant Cell 20:2798–2814
Grove MD, Spencer GF, Rohwedder WK, Mandava N, Worley JF, Warthen JD, Steffens GL, Flippen-Anderson JL, Cook JC (1979) Brassinolide, a plant growth-promoting steroid isolated from Brassica napus pollen. Nature 281:216–217
Hewitt FR, Hough T, O’Neill P, Sasse JM, Williams EG, Rowan KS (1985) Effect of Brassinolide and other growth regulators on the germination and growth of pollen tubes of Prunus avium using a multiple hanging-drop assay. Aust J Plant Physiol 12(2):201–211
Higashiyama T (2010) Peptide signaling in pollen–pistil interactions. Plant Cell Physiol 51:177–189
Huang H-Y, Jiang W-B, Hu Y-W, Wu P, Zhu J-Y, Liang W-Q, Wang Z-Y, Lin W-H (2012) BR signal influences Arabidopsis Ovule and seed number through regulating related genes expression by BZR1. Mol Plant 6(2):456–469
Johnson MA, Preuss D (2002) Plotting a course: multiple signals guide pollen tubes to their targets. Dev Cell 2:273–281
Johnson-Brousseau SA, McCormick S (2004) A compendium of methods useful for characterizing Arabidopsis pollen mutants and gametophytically-expressed genes. Plant J 39:761–775
Kim TW, Hwang JY, Kim YS, Joo SH, Chang SC, Lee JS, Takatsuto S, Kim SK (2005) Arabidopsis CYP85A2, a cytochrome P450, mediates the Baeyer–Villiger oxidation of castasterone to brassinolide in brassinosteroid biosynthesis. Plant Cell 17:2397–2412
Kwon M, Choe S (2005) Brassinosteroid biosynthesis and dwarf mutants. Journal of Plant Biology 48:1–15
Lennon KA, Roy S, Hepler PK, Lord EM (1998) The structure of the transmitting tissue of Arabidopsis thaliana (L.) and the path of pollen tube growth. Sex Plant Reprod 11:49–59
Li H, Lin Y, Heath RM, Zhu MX, Yang Z (1999) Control of pollen tube tip growth by a Rop GTPase-dependent pathway that leads to tip-localized Calcium influx. Plant Cell 11:1731–1742
Lord EM, Russell SD (2002) The mechanisms of pollination and fertilization in plants. Annu Rev Cell Dev Biol 18:81–105
Mandava N (1988) Plant growth-promoting brassinosteroids. Annu Rev Plant Physiol Plant Mol Biol 39:23–52
Mascarenhas JP (1993) Molecular mechanisms of pollen tube growth and differentiation. Plant Cell 5:1303–1314
Mathur J, Molnár G, Fujioka S, Takatsuto S, Sakurai A, Yokota T, Adam G, Voigt B, Nagy F, Maas C, Schell J, Koncz C, Szekeres M (1998) Transcription of the Arabidopsis CPD gene, encoding a steroidogenic cytochrome P450, is negatively controlled by brassinosteroids. Plant J 14:593–602
Mo Y, Nagel C, Taylor LP (1992) Biochemical complementation of chalcone synthase mutants defines a role for flavonols in functional pollen. Proc Natl Acad Sci USA 89:7213–7217
Mori T, Kuroiwa H, Higashiyama T, Kuroiwa T (2006) GENERATIVE CELL SPECIFIC 1 is essential for angiosperm fertilization. Nat Cell Biol 8(1):64–71
Mouline K, Véry AA, Gaymard F, Boucherez J, Pilot G, Devic M, Bouchez D, Thibaud JB, Sentenac H (2002) Pollen tube development and competitive ability are impaired by disruption of a Shaker K+ channel in Arabidopsis. Genes Dev 16:339–350
Müssig C, Shin G-H, Altmann T (2003) Brassinosteroids Promote Root Growth in Arabidopsis. Plant Physiol 133:1261–1271
Noguchi T, Fujioka S, Choe S, Takatsuto S, Yoshida S, Yuan H, Feldmann KA, Tax FE (1999) Brassinosteroid-insensitive dwarf mutants of Arabidopsis accumulate brassinosteroids. Plant Physiol 121:743–752
Ohnishi T, Godza B, Watanabe B, Fujioka S, Hategan L, Ide K, Shibata K, Yokota T, Szekeres M, Mizutani M (2012) CYP90A1/CPD, a brassinosteroid biosynthetic cytochrome P450 of Arabidopsis, catalyzes C-3 oxidation. J Biol Chem 287:31551–31560
Palanivelu R, Tsukamoto T (2012) Pathfinding in angiosperm reproduction: pollen tube guidance by pistils ensures successful double fertilization. Wiley Interdiscip Rev Dev Biol 1:96–113
Palanivelu R, Brass L, Edlund AF, Preuss D (2003) Pollen tube growth and guidance is regulated by POP2, an Arabidopsis gene that controls GABA levels. Cell 114:47–59
Poppenberger B, Rozhon W, Khan M, Husar S, Adam G, Luschnig C, Fujioka S, Sieberer T (2011) CESTA, a positive regulator of brassinosteroid biosynthesis. EMBO J 30:1149–1161
Qin Y, Yang Z (2011) Rapid tip growth: insights from pollen tubes. Semin Cell Dev Biol 22:816–824
Qin Y, Wysocki RJ, Somogyi A, Feinstein Y, Franco JY, Tsukamoto T, Dunatunga D, Levy C, Smith S, Simpson R, Gang D, Johnson MA, Palanivelu R (2011) Sulfinylated azadecalins act as functional mimics of a pollen germination stimulant in Arabidopsis pistils. Plant J 68:800–815
Rodriguez-Enriquez MJ, Mehdi S, Dickinson HG, Grant-Downton RT (2013) A novel method for efficient in vitro germination and tube growth of Arabidopsis thaliana pollen. New Phytologist 197:668–679
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, Preibisch S, Rueden C, Saalfeld S, Schmid B, Tinevez JY, White DJ, Hartenstein V, Eliceiri K, Tomancak P, Cardona A (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9:676–682
Schmidt J, Altmann T, Adam G (1997) Brassinosteroids from seeds of Arabidopsis thaliana. Phytochemistry 45(7):1325–1327
Sedgley M (1975) Flavanoids in pollen and stigma of Brassica oleracea and their effects on pollen germination in vitro. Ann Bot 39:1091–1095
Shimada Y, Goda H, Nakamura A, Takatsuto S, Fujioka S, Yoshida S (2003) Organ-specific expression of brassinosteroid-biosynthetic genes and distribution of endogenous brassinosteroids in Arabidopsis. Plant Physiol 131:287–297
Singh I, Shono M (2005) Physiological and molecular effects of 24-Epibrassinolide, a brassinosteroid on thermotolerance of tomato. Plant Growth Regul 47:111–119
Singh DP, Jermakow AM, Swain SM (2002) Gibberellins are required for seed development and pollen tube growth in Arabidopsis. Plant Cell 14:3133–3147
Smith AG, Eberle CA, Moss NG, Anderson NO, Clasen BM, Hegeman AD (2013) The transmitting tissue of Nicotiana tabacum is not essential to pollen tube growth, and its ablation can reverse prezygotic interspecific barriers. Plant Reprod 26:339–350
Smyth DR, Bowman JL, Meyerowitz EM (1990) Early flower development in Arabidopsis. Plant Cell 2:755–767
Sotomayor C, Castro J, Velasco N, Toro R (2012) Influence of seven growth regulators on fruit set, pollen germination and pollen tube growth of almonds. J Agric Sci Technol B:1051–1056
Sprunck S (2010) Let’s get physical: gamete interaction in flowering plants. Biochem Soc Trans 38:635–640
Sprunck S, Rademacher S, Vogler F, Gheyselinck J, Grossniklaus U, Dresselhaus T (2012) Egg cell-secreted EC1 triggers sperm cell activation during double fertilization. Science 338:1093–1097
Stanley R, Liskens H (1974) Pollen. Springer-Verlag, New York
Steber CM, McCourt P (2001) A role for brassinosteroids in germination in Arabidopsis. Plant Physiol 125:763–769
Szekeres M, Németh K, Koncz-Kálmán Z, Mathur J, Kauschmann A, Altmann T, Rédei GP, Nagy F, Schell J, Koncz C (1996) Brassinosteroids rescue the deficiency of CYP90, a cytochrome P450, controlling cell elongation and de-etiolation in Arabidopsis. Cell 85:171–182
Takada S, Jürgens G (2007) Transcriptional regulation of epidermal cell fate in the Arabidopsis embryo. Development 134:1141–1150
Takeuchi H, Higashiyama T (2011) Attraction of tip-growing pollen tubes by the female gametophyte. Curr Opin Plant Biol 14:614–621
Taylor LP, Hepler PK (1997) Pollen germination and tube growth. Annu Rev Plant Physiol Plant Mol Biol 48:461–491
Thussagunpanit J, Jutamanee K, Chai-arree W, Kaveeta L (2012) Increasing photosynthetic efficiency and pollen germination with 24-Epibrassinolide in rice (Oryza sativa L.) under heat stress. Thai J Bot 4:135–143
Twell D, Yamaguchi J, McCormick S (1990) Pollen-specific gene expression in transgenic plants: coordinate regulation of two different tomato gene promoters during microsporogenesis. Development 109:705–713
Vriet C, Russinova E, Reuzeau C (2013) From Squalene to Brassinolide: the Steroid Metabolic and Signaling Pathways across the Plant Kingdom. Mol Plant 6:1738–1757
Wu J, Qin Y, Zhao J (2008a) Pollen tube growth is affected by exogenous hormones and correlated with hormone changes in styles in Torenia fournieri L. Plant Growth Regul 55:137–148
Wu J, Lin Y, Zhang XL, Pang DW, Zhao J (2008b) IAA stimulates pollen tube growth and mediates the modification of its wall composition and structure in Torenia fournieri. J Exp Bot 59:2529–2543
Wu J, Shang Z, Wu J, Jiang X, Moschou PN, Sun W, Roubelakis-Angelakis KA, Zhang S (2010) Spermidine oxidase-derived H2O2 regulates pollen plasma membrane hyperpolarization-activated Ca2+-permeable channels and pollen tube growth. Plant J 63:1042–1053
Wu J, Qin X, Tao S, Jiang X, Liang Y-K, Zhang S (2014) Long-chain base phosphates modulate pollen tube growth via channel-mediated influx of calcium. Plant J. doi:10.1111/tpj.12576
Yang Z (2008) Cell polarity signaling in Arabidopsis. Annu Rev Cell Dev Biol 24:551–575
Yang X-H, Xu Z-H, Xue H-W (2005) Arabidopsis Membrane Steroid Binding Protein 1 Is Involved in Inhibition of Cell Elongation. Plant Cell 17:116–131
Ye Q, Zhu W, Li L, Zhang S, Yin Y, Ma H, Wang X (2010) Brassinosteroids control male fertility by regulating the expression of key genes involved in Arabidopsis anther and pollen development. Proc Natl Acad Sci 107:6100–6105
Yetisen AK, Jiang L, Cooper JR, Qin Y, Palanivelu R, Zohar Y (2011) A microsystem-based assay for studying pollen tube guidance in plant reproduction. J Micromech Microeng 21:054018
Ylstra B, Touraev A, Moreno RM, Stöger E, van Tunen AJ, Vicente O, Mol JN, Heberle-Bors E (1992) Flavonols stimulate development, germination, and tube growth of tobacco pollen. Plant Physiol 100:902–907
Ylstra B, Touraev A, Brinkmann AO, Heberle-Bors E, Tunen A (1995) Steroid hormones stimulate germination and tube growth of in vitro matured tobacco pollen. Plant Physiol 107:639–643
Yu G-H, Zou J, Feng J, Peng X-B, Wu J-Y, Wu Y-L, Palanivelu R, Sun M-X (2014) Exogenous γ-aminobutyric acid affects pollen tube growth via modulating putative Ca2+-permeable membrane channels and is coupled to negative regulation on glutamate decarboxylase. J Exp Bot 65(12):3235–3248
Zheng B, Chen X, McCormick S (2011) The anaphase-promoting complex is a dual integrator that regulates both MicroRNA-mediated transcriptional regulation of cyclin B1 and degradation of Cyclin B1 during Arabidopsis male gametophyte development. Plant Cell 23:1033–1046
Zimmermann P, Hirsch-Hoffmann M, Hennig L, Gruissem W (2004) GENEVESTIGATOR. Arabidopsis microarray database and analysis toolbox. Plant Physiol 136:2621–2632
Acknowledgments
We thank Mily Ron for providing the Gateway destination vector pGreenII-GW:NLS:3xEGFP and David Twell for sharing the Lat52p:GFP marker line. We are grateful to Frank Sprenger for assistance with the Spinning Disc Confocal Microscope and Thomas Dresselhaus for critical discussions. We thank Monika Kammerer for technical assistance. This work was funded by the German Research Council (DFG) with grants SFB 924 and SP 686/1-2 to SS.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Tetsuya Higashiyama.
Electronic supplementary material
Below is the link to the electronic supplementary material.
497_2014_247_MOESM1_ESM.pdf
Figure S1 Scheme of the pollen application procedure. To balance out the time lags associated with pollen application and image acquisition, and to take varying pollen qualities into account, pollen was applied to the PGM in a standardized fashion. a By gently dabbing dehiscent anthers flowers on plates, spots of comparable size and pollen density were applied to the germination medium. Scale bar: 100 µm. b Seven pollen germination plates (A to G) containing PGM supplemented with varying epiBL concentrations (0 to 50 µM) were each subdivided in 8 sectors. Pollen was applied to the first sector of plate A (1.; yellow circle) and subsequently to the other 6 plates (2. – 7.). For the next sector, pollen application started with the second plate (B), and ended with plate A (pollination order 8. – 14.). For sector three, pollen application started with plate C (pollination order 15. – 21.), and so on. Images were acquired in the same order as the pollination was performed. (PDF 644 kb)
497_2014_247_MOESM2_ESM.pdf
Figure S2 BRI1-like (BRL) receptor kinases with BR binding activity have low expression values in reproductive tissues. Expression of BRI1-like 1 (BRL1) (light gray) and BRL3 (dark gray) in different reproductive and non-reproducti-ve tissues. Log2-fold expression values and gene expression levels displayed relative to the signal intensity on the 22 k ATH1 array according to the GENEVESTIGATOR microarray database (Zimmermann et al. 2004). Num-ber of samples used for calculating mean values ± 1SE: embryo, 38; endosperm, 11; pollen, 52; pistil, 29; car-pel, 2; stigma, 3; ovary, 4; ovule, 2; silique, 372; seed, 323; seedling, 2,330; shoot, 4,504; leaf, 2,849; roots, 1,063. (PDF 178 kb)
497_2014_247_MOESM3_ESM.pdf
Figure S3 CYP90A1/CPD promoter activity in developing pistils. a – f Pistils of plants expressing the NLS:3xEGFP reporter under control of the CYP90A1/CPD promoter (CYP90A1p:NLS:3xEGFP) were imaged at different developmental stages. Flower stages were defined according to Smyth et al. (1990). In pistils of flower stages 9 (a) and 10 (b), only very weak and ubiquitous promoter activity was detected. In flower stages 11 (c), 12 (d), and 13 (e), strong GFP signals appeared in the nuclei of reproductive tract cells, including the stigma, style and transmitting tract (tt). f After fertilization (flower stage 14), promoter activity in the cells of the reproductive tract started to diminish. Note that in flower stage 9 (a) the septum often ruptures while preparing the tiny pistils. Scale bars: a – d 100 µm, e, f 150 µm. (PDF 11053 kb)
497_2014_247_MOESM4_ESM.pdf
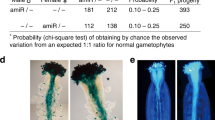
Figure S4 Dwarf phenotypes of homozygous cyp90a1-1 and bri1-10. Compared to the wild type (Col-0), the BR biosynthesis mutant cyp90a1-1 and the BR perception mutant bri1-10 both exhibited severe dwarf phenotypes. (PDF 5709 kb)
Rights and permissions
About this article
Cite this article
Vogler, F., Schmalzl, C., Englhart, M. et al. Brassinosteroids promote Arabidopsis pollen germination and growth. Plant Reprod 27, 153–167 (2014). https://doi.org/10.1007/s00497-014-0247-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00497-014-0247-x