Abstract
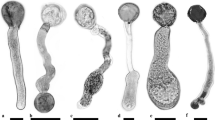
Expansive growth in plant cells is a formidable problem for biophysical studies, and the mechanical principles governing the generation of complex cellular geometries are still poorly understood. Pollen, the male gametophyte stage of the flowering plants, is an excellent model system for the investigation of the mechanics of complex growth processes. The initiation of pollen tube growth requires first of all, the spatially confined formation of a protuberance. This process must be controlled by the mechanical properties of the cell wall, since turgor is a non-vectorial force. In the elongating tube, cell wall expansion is confined to the apex of the cell, requiring the tubular region to be stabilized against turgor-induced tensile stress. Tip focused surface expansion must be coordinated with the supply of cell wall material to this region requiring the precise, logistical control of intracellular transport processes. The advantage of such a demanding mechanism is the high efficiency it confers on the pollen tube in leading an invasive way of life.




Similar content being viewed by others
References
Anderson JR, Barnes WS, Bedinger P (2002) 2, 6-Dichlorbenzonitrile, a cellulose biosynthesis inhibitor, affects morphology and structural integrity of petunia and lily pollen tubes. J Plant Physiol 159:61–67
Aouar L, Chebli Y, Geitmann A (2009) Morphogenesis of complex plant cell shapes—the mechanical role of crystalline cellulose in growing pollen tubes. Sex Plant Reprod (in press)
Baluška F, Salaj J, Mathur J, Braun M, Jasper F, Šamaj J, Chua NH, Barlow PW, Volkmann D (2000) Root hair formation: F-actin-dependant tip growth is initiated by local assembly of profilin-supported F-actin meshworks accumulated within expansin-enriched bulges. Dev Biol 227:618–632
Baskin T (2005) Anisotropic expansion of the plant cell wall. Annu Rev Cell Dev Biol 21:203–222
Benkert R, Obermeyer G, Bentrup FW (1997) The turgor pressure of growing lily pollen tubes. Protoplasma 198:1–8
Bibikova TN, Jacob T, Dahse I, Gilroy S (1998) Localized changes in apoplastic and cytoplasmic pH are associated with root hair development in Arabidopsis thaliana. Development 125:2925–2934
Bolick MR (1981) Mechanics as an aid to interpreting pollen structure and function. Rev Paleobot Palynol 35:61–80
Bolick MR, Vogel S (1992) Breaking strengths of pollen grain walls. Plant Syst Evol 181:171–178
Bosch M, Cheung AY, Hepler PK (2005) Pectin methylesterase, a regulator of pollen tube growth. Plant Physiol 138:1334–1346
Bove J, Vaillancourt B, Kroeger J, Hepler PK, Wiseman PW, Geitmann A (2008) Magnitude and direction of vesicle dynamics in growing pollen tubes using spatiotemporal image correlation spectroscopy (STICS). Plant Physiol 147:1646–1658
Carpita NC, Gibeaut DM (1993) Structural models of primary cell walls in flowering plants: consistency of molecular structure with the physical properties of the walls during growth. Plant J 3:1–30
Castells T, Seoane-Camba JA, Suàrez-Cervera M (2003) Intine wall modifications during germination of Zygophyllum fabago (Zygophyllaceae) pollen grains. Can J Bot 81:1267–1277
Castle E (1958) The topography of tip growth in a plant cell. J Gen Physiol 41:913–926
Chebli Y, Bou Daher F, Sanyal M, Aouar L, Geitmann A (2008) Microwave assisted processing of plant cells for optical and electron microscopy. Bull Micr Soc Can 36:15–19
Cho H-T, Cosgrove DJ (2002) Regulation of root hair initiation and expansin gene expression in Arabidopsis. Plant Cell 14:3237–3253
Cresti M, Pacini E, Ciampolini F, Sarfatti G (1977) Germination and early tube development in vitro of Lycopersicum peruvianum pollen: ultrastructural features. Planta 136:239–247
Cresti M, Ciampolini F, Mulcahy DLM, Mulcahy G (1985) Ultrastructure of Nicotiana alata pollen, its germination and early tube formation. Amer J Bot 72:719–727
Dai S, Chen T, Chong K, Xue Y, Liu S, Wang T (2007) Proteomics identification of differentially expressed proteins associated with pollen germination and tube growth reveals characteristics of germinated Oryza sativa pollen. Mol Cell Proteomics 6:207–230
Derksen J, Rutten T, Lichtscheidl IK, DeWin AHN, Pierson ES, Rongen G (1995) Quantitative analysis of the distribution of organelles in tobacco pollen tubes: implications for exocytosis and endocytosis. Protoplasma 188:267–276
Derksen J, Li Y-Q, Knuiman B, Geurts H (1999) The wall of Pinus sylvestris L. pollen tubes. Protoplasma 208:26–36
Dumais J, Long SR, Shaw SL (2004) The mechanics of surface expansion anisotropy in Medicago truncatula root hairs. Plant Physiol 136:3266–3275
Dumais J, Shaw SL, Steele CR, Long SR, Ray PM (2006) An anisotropic-viscoplastic model of plant cell morphogenesis by tip growth. Int J Dev Biol 50:209–222
Edlund AF, Swanson R, Preuss D (2004) Pollen and stigma structure and function: the role of diversity in pollination. Plant Cell 16(Suppl):S84–S97
Edlund AF, Zheng Q, Lyles R, Mosley R, Sibener SJ, Preuss D (2009) Breaching the exine wall: pollen tube germination in Arabidopsis thaliana (Brassicaceae). Ann Bot (in press)
Geitmann A, Dumais J (2009) Not-so-tip-growth. Plant Signal Behav 4:136–138
Geitmann A, Ortega JKE (2009) Mechanics and modeling of plant cell growth. Trends Plant Sci 14:467–478
Geitmann A, Parre E (2004) The local cytomechanical properties of growing pollen tubes correspond to the axial distribution of structural cellular elements. Sex Plant Reprod 17:9–16
Geitmann A, Steer MW (2006) The architecture and properties of the pollen tube cell wall. In: Malhó R (ed) The pollen tube: a cellular and molecular perspective, plant cell Monographs, vol 3. Springer, Berlin, pp 177–200
Gossot O, Geitmann A (2007) Pollen tube growth—coping with mechanical obstacles involves the cytoskeleton. Planta 226:405–416
Harold FM (2002) Force and compliance: rethinking morphogenesis in walled cells. Fungal Genet Biol 37:271–282
Heath IB (1987) Preservation of a labile cortical array of actin filaments in growing hyphal tips of the fungus Saprolegnia ferax. Eur J Cell Biol 44:10–16
Heslop-Harrison J (1975) The adaptive significance of the exine. In: Ferguson IK, Muller J (eds) The evolutionary significance of the exine. Academic Press, London, pp 27–38
Heslop-Harrison J (1979a) Aspects of the structure, cytochemistry and germination of the pollen of rye (Secale cereale L.). Ann Bot 44(Suppl):1–47
Heslop-Harrison J (1979b) An interpretation of the hydrodynamics of pollen. Am J Bot 50:831–842
Heslop-Harrison J (1979c) Pollen walls as adaptive systems. Ann Mo Bot Gard 66:813–829
Heslop-Harrison J (1987) Pollen germination and pollen-tube growth. Int Rev Cytol 107:1–78
Heslop-Harrison Y, Heslop-Harrison J (1992) Germination of monocolpate angiosperm pollen: evolution of actin cytoskeleton and wall during hydration, activation and tube emergence. Ann Bot 69:385–394
Higashiyama T, Hamamura Y (2008) Gametophytic pollen tube guidance. Sex Plant Reprod 21:17–26
Holmes-Davis R, Tanaka CK, Vensel WH, Hurkman WJ, McCormick S (2005) Proteome mapping of mature pollen of Arabidopsis thaliana. Proteomics 5:4864–4884
Hughes J, McCully ME (1975) The use of an optical brightener in the study of plant structure. Stain Technol 50:319–329
Knox RB, Heslop-Harrison J (1970) Pollen-wall proteins: localization and enzymic activity. J Cell Sci 6:1–27
Koehl MAR, Quillin KJ, Pell A (2000) Mechanical design of fiber-wound hydraulic skeletons: the stiffening and straightening of embryonic notochords. Am Zool 40:28–41
Kroeger JH, Bou Daher F, Grant M, Geitmann A (2009) Microfilament orientation constrains vesicle flow and spatial distribution in growing pollen tubes. Biophys J 97:1822–1831
Lazzaro MD, Donohue JM, Soodavar FM (2003) Disruption of cellulose synthesis by isoxaben causes tip swelling and disorganizes cortical microtubules in elongating conifer pollen tubes. Protoplasma 220:201–207
Lenartowska M, Michalska A (2008) Actin filament organization and polarity in pollen tubes revealed by myosin II subfragment 1 decoration. Planta 228:891–896
Li Y-Q, Zhang HQ, Pierson ES, Huang FY, Linskens HF, Hepler PK, Cresti M (1996) Enforced growth-rate fluctuation causes pectin ring formation in the cell wall of Lilium longiflorum pollen tubes. Planta 200:41–49
Lovy-Wheeler A, Wilsen KL, Baskin TI, Hepler PK (2005) Enhanced fixation reveals the apical cortical fringe of actin filaments as a consistent feature of the pollen tube. Planta 221:95–104
Márquez J, Seoane-Camba JA, Suárez-Cervera M (1997) Allergenic and antigenic proteins released in the apertural sporoderm during the activation process in grass pollen grains. Sex Plant Reprod 10:269–278
Money NP (1997) Wishful thinking of turgor revisited: the mechanics of fungal growth. Fungal Genet Biol 21:173–187
Money NP, Harold FM (1992) Extension growth of the water mold Achlya: interplay of turgor and wall strength. Cell Biol 89:4245–4249
Money NP, Harold FM (1993) Two water molds can grow without measurable turgor pressure. Planta 190:426–430
Muller J (1979) Form and function in angiosperm pollen. Ann Mo Bot Gard 66:593–632
Nepi M, Pacini E (1999) What could be the significance of polysiphony in Lavatera arborea? In: Clément C, Pacini E, Audran J-C (eds) Anther and pollen: from biology to biotechnology. Springer, Berlin, pp 13–20
Noir S, Bräutigam A, Colby T, Schmidt J, Panstruga R (2005) A reference map of the Arabidopsis thaliana mature pollen proteome. Biochem Biophys Res Commun 337:1257–1266
Pacini E (1990) Harmomegathic characters of Pteridophyta spores and Spermatophyta pollen. Plant Syst Evol Suppl 5:53–69
Parre E, Geitmann A (2005a) More than a leak sealant—the physical properties of callose in pollen tubes. Plant Physiol 137:274–286
Parre E, Geitmann A (2005b) Pectin and the role of the physical properties of the cell wall in pollen tube growth of Solanum chacoense. Planta 220:582–592
Payne WW (1972) Observations of harmomegathy in pollen of Anthophyta. Grana 12:93–98
Payne WW (1981) Structure and function in angiosperm pollen wall evolution. Rev Palaebot Palynol 35:39–60
Pickett-Heaps JD, Klein AG (1998) Tip growth in plant cells may be amoeboid and not generated by turgor pressure. Proc R Soc London 265:1453–1459
Pina C, Pinto F, Feijó JA, Becker JD (2005) Gene family analysis of the Arabidopsis pollen transcriptome reveals biological implications for cell growth, division control, an gene expression regulation. Plant Physiol 138:744–756
Proseus T, Boyer J (2006) Calcium pectate chemistry controls growth rate of Chara corallina. J Exp Bot 57:3989–4002
Proseus T, Boyer J (2007) Tension required for pectate chemistry to control growth in Chara corallina. J Exp Bot 58:4283–4292
Rae AL, Harris PJ, Bacic A, Clarke AE (1985) Composition of the cell walls of Nicotiana alata Link et Otto pollen tubes. Planta 166:128–133
Röckel N, Wolf S, Kost B, Rausch T, Greiner S (2008) Elaborate spatial patterning of cell-wall PME and PMEI at the pollen tube tip involves PMEI endocytosis, and reflects the distribution of esterified and de-esterified pectins. Plant J 53:133–143
Rowley JR, Skvarla JJ (2000) The elasticity of the exine. Grana 39:1–7
Russell SD, Bhalla PL, Singh MB (2008) Transcriptome-based examination of putative pollen allergens of rice (Oryza sativa ssp. japonica). Mol Plant 1:751–759
Sassen MMA (1964) Fine structure of Petunia pollen grain and pollen tube. Acta Bot Neerl 13:175–181
Scotland RW, Barnes SH, Blackmore S (1990) Harmomegathy in the Acanthaceae. Grana 29:37–45
Sheoran IS, Pedersen EJ, Ross ARS, Sawhney VK (2009) Dynamics of protein expression during pollen germination in canola (Brassica napus). Planta 230:779–793
Suarez-Cervera M, Arcalis E, Le Thomas A, Seoane-Camba J (2002) Pectin distribution pattern in the apertural intine of Euphorbia peplus L. (Euphorbiaceae) pollen. Sex Plant Reprod 14:291–298
Suen DF, Huang AH (2007) Maize pollen coat xylanase facilitates pollen tube penetration into silk. J Biol Chem 282:625–636
Tian G-H, Chen M-H, Zaltsman A, Citovsky V (2006) Pollen-specific pectin methylesterase involved in pollen tube growth. Dev Biol 294:83–91
Tiwari SC, Polito VS (1988) Spatial and temporal organisation of actin during hydration, activation and germination of pollen in Pyrus communis L.: a population study. Protoplasma 147:5–15
Valdivia ER, Stephenson AG, Durachko DM, Cosgrove D (2009) Class B b-expansins are needed for pollen separation and stigma penetration. Sex Plant Reprod 22:141–152
Vidali L, McKenna ST, Hepler PK (2001) Actin polymerization is essential for pollen tube growth. Mol Biol Cell 12:2534–2545
Vissenberg K, Fry SC, Verbelen JP (2001) Root hair initiation is coupled to a highly localized increase of xyloglucan endotransglycosylase action in Arabidopsis roots. Plant Physiol 127:1125–1135
Wodehouse RP (1935) Pollen grains. McGraw-Hill, New York
Zerzour R, Kroeger JH, Geitmann A (2009) Polar growth in pollen tubes is associated with spatially confined dynamic changes in cell mechanical properties. Dev Biol 334:437–446
Acknowledgments
Research in the Geitmann lab is funded by the Natural Sciences and Engineering Research Council of Canada (NSERC), the Fonds Québécois de la Recherche sur la Nature et les Technologies (FQRNT), and the Human Frontier Science Program (HFSP). Thanks to Youssef Chebli and Louise Pelletier for preparing the scanning electron micrographs. I am grateful to Phil Lintilhac for critically reading the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Scott Russell.
Rights and permissions
About this article
Cite this article
Geitmann, A. How to shape a cylinder: pollen tube as a model system for the generation of complex cellular geometry. Sex Plant Reprod 23, 63–71 (2010). https://doi.org/10.1007/s00497-009-0121-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00497-009-0121-4