Abstract
Several old cultivars, and breeding clones of European pear Pyrus communis L. originating from Belgium, England, Sweden, and Switzerland were evaluated for their resistance/susceptibility to fire blight. Studies were carried out during three consecutive years 2007–2009 in the greenhouse of Warsaw University of Life Sciences, Poland. Strain 691 of Erwinia amylovora was used for artificial infection of plants. Genotypes included in this study considerably varied in their resistance to fire blight. The most resistant was the old English cultivar ‘Hessle’. The other two genotypes, i.e., ‘Gränna Rödpäron’ originating from Sweden, and Pyrus communis FG 1606 from Switzerland were included in a group of low susceptible ones. The most susceptible were Cra Py H 18, Cra Py V 22 and Cra Py W 14 from Belgium.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The genus Pyrus, containing at least 22 species, is a highly diverse source of pome fruit cultivated throughout the temperate climate regions of the world. The major edible species in Europe, North America, and temperate regions of the southern hemisphere is the European pear (Pyrus communis L.), commercially cultivated for fruit production. It encompasses approximately 5,000 cultivars (Monte-Corvo et al. 2001), only a small percentage of which are cultivated commercially (Bell et al. 1996). A major factor that significantly limits the cultivation of European pear is fire blight caused by the bacterium Erwinia amylovora (Burrill) Winslow et al. This disease can be especially problematic in regions where environmental conditions are favorable for the disease development, especially where springtime weather is warm and wet (van der Zwet and Keil 1979). Nearly all cultivars of P. communis are susceptible to this disease (van der Zwet et al. 1974). Breeding of new cultivars resistant to fire blight is one of the most important goals to overcome the problem. Genetic diversity that exists within genus Pyrus has been used for this purpose (Bell and Janick 1977).
Susceptibility of pear or apple genotypes can be assessed through different methods of measurements after the inoculation of blossoms or shoots. The extent of lesion development on the shoot appeared to be most useful for that purpose. Measurements of that type were shown to be strongly correlated with the field susceptibility of apple cultivars in several independent observations (Lespinasse and Aldwinckle 2000). Blossom susceptibility of apple and pear cultivars received less attention than the susceptibility of vegetative tissues. However, the susceptibility to blossom infection may be important in determining how readily infections are initiated in the orchard Aldwinckle and Norelli (1981). The aim of this study was to determine the level of fire blight resistance among different old cultivars and clones originating from breeding programs of several European countries using artificial inoculation. Artificial inoculation of actively growing shoots with virulent strain of Erwinia amylovora provide an effective and reliable means for evaluating fire blight resistance Aldwinckle and Preczewski (1976).
Since susceptibility of shoots is strongly influenced by their physiological state, three independent assessments for fire blight tests, with ten trees as replicates in each, were applied. In each year of the replicated experiment all trees were of the same age.
The other objective of this study was to predict response of pear genotypes to pathogen infection across four measurement times.
Materials and methods
Three independent greenhouse-resistance assessments (summer 2007, 2008, and 2009) were performed at the Warsaw University of Life Sciences, Department of Pomology, in cooperation with Department of Plant Pathology. Plant material for studies originated from Belgium, England, Sweden, Switzerland (Table 1) and was delivered by three breeding centers, i.e., University of Agricultural Sciences, Department of Plant Breeding and Biotechnology, Balsgård, Sweden (H. Nybom); Waloon Agricultural Research Centre, Gembloux, Belgium (M. Lateur); and Research Station Agroscope Changins-Wädenswil (ACW), Switzerland (M. Kellerhals). Commercially cultivated European pear cv ‘Doyenné du Comice’ served as a control.
Every year (2007, 2008, and 2009) in winter time scions of each genotype were grafted on the potted seedlings of Pyrus caucasica in ten replicates. Grafted plants were grown in a greenhouse. At the time of vegetation two shoots were developed from each scion. At the end of June 2007, 2008, and 2009, according to the method used by Bell et al. (2004), two youngest leaves of 50-cm-tall plants were clipped with scissors immersed in inoculum of the E. amylovora aggressive strain 691, containing 108 cfu ml−1. The strain was isolated from ‘Šampion’ apple cv in 1998 at the Research Institute of Pomology and Floriculture, Skierniewice, Poland. In order to provide high humidity conditions after inoculation, plants were covered with plastic bags for 24 h. During 4 weeks of evaluation the following mean values were maintained: 27°C (day), 21°C (night) and 55% air humidity.
After inoculation the disease lesion length and the total length of the current season’s growth of the inoculated shoots were measured. The level of infection was expressed as the percentage of the fire blight lesion lengths in relation to the overall shoot length. The progression of symptoms was monitored for four consecutive weeks after the inoculation and evaluated according to the Gardner scale (Gardner et al. 1980): 0–10% very resistant, 11–30% resistant, 31–50% moderately susceptible, 51–90% susceptible, 91–100% very susceptible.
Statistical analysis
Data for fire blight infection recorded on ten trees (means of two shoots per tree) of each tested genotype across four measurement times and 3 years were analyzed using two-stage approach. At the first stage means of ten trees for each combination of genotype-measurement time–year were calculated. At the second stage, the three-way analysis of variance (ANOVA) based on a mixed model for means of ten trees was carried out for the means designed in an incomplete genotype-measurement time–year classification. Means of ten tree samples were analyzed by ANOVA as the percentage of fire blight infection within each genotype-measurement time–year combination appeared to be not normally distributed. It is known that for not normally distributed variable, when the sample size is large, the distribution of sample means calculated on the basis of the sample taken from a population is asymptotically normal. The incompleteness of the classification is due to the fact that not all genotypes were evaluated in each of the 3 years. Genotype and measurement time were assumed to be fixed factors whereas year a random factor. In this study the most important evaluated factors were genotype and measurement time. Their main and interaction effects of the fire blight infection were tested statistically with F test using respective interactions of these assessed fixed factor effects with year as the error term (McIntosh 1983; Steel et al. 1997). Multiple comparisons of means for genotypes were performed using the Tukey’s method at 0.05 probability level.
As a result of ANOVA, also the adjusted means for a complete classification of genotype × measurement time were obtained. These means are average values of the infection of three test years for each combination of genotype-measurement time. They facilitate to predict response of each tested genotype for the fire blight infection across measurement times. These responses were diverse for the genotypes due to the significance of both main genotype effects and genotype × measurement time interaction. Therefore, classification of the genotype responses into homogenous groups was carried out using Ward’s method of cluster analysis for the adjusted genotype × measurement time means. The squared Euclidean distance was used as the dissimilarity measure between genotypes. The analyses were conducted using Statistica 7.1 package StatSoft (2005).
Results
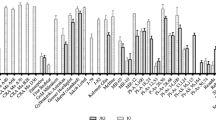
In the analysis of variance (Table 2), based on the mixed model the only interesting effects for genotypes and measurement times were tested. Main effects of genotypes and measurement times as well as effects of genotype × measurement time interaction were significant (P < 0.001 for all these effects). It means that there were significant differences between the infection level of genotypes and between the measurement times, and also the infection response patterns of the genotypes across measurement times were significantly different for the examined genotypes. In the first measurement time most genotypes had quite low infection level; the differences between resistant and susceptible genotypes increased in further measurements. For some genotypes the infection level was very stable during the subsequent measurements (e.g., ‘Hessle’) and for some of them infection level was increasing very quickly (especially for susceptible genotypes, e.g., Cra Py W 14). The genotype × measurement interaction found by the analysis of variance is illustrated by the not parallel response-infection lines for groups of genotypes (Fig. 2). Because of the significant effect of genotype x measurement times interaction in ANOVA, grouping of similar genotypes based on the means of subsequent measurements would be reasonable.
Cluster analysis was performed based on the adjusted means of the fire blight infection (Table 3) for the studied genotypes and measurement times. The squared Euclidean distances were calculated between values in rows of this table for pairs of genotypes. The obtained dendrogram was cut at five-group level (Fig. 1). Variation between these distinguished groups captured about 90% of the total variations among the genotypes. It means that genotypes in each of five groups are considerably homogenous for the infection response across the measurement times.
The most resistant cultivar was ‘Hessle’ which was distinguished in a separate homogenous group 1, based both on multiple comparisons of genotypic means and on disease infection response across the measurement times (Table 3, Fig. 2). This cultivar had very low percentage of infection at all measurements, and was classified as very resistant according to Gardner scale (Gardner et al. 1980). Low susceptibility was observed for two other genotypes, i.e., Pyrus communis FG 1606 and ‘Gränna Rödpäron’ classified as resistant. They belonged to cluster 2 and one genotype mean homogenous group denoted by letter b (Table 3). Genotypes in group 3 (7 genotypes) are moderately susceptible to this disease. The infection response of the genotypes in the group was similar in subsequent measurements. The susceptible genotypes were in groups 4 (5 genotypes) and 5 (3 genotypes). ‘Doyenné du Comice’, which served as a control was ranked according to the Gardner scale to the third class as a moderately susceptible. Data for this cv were published (Bokszczanin et al. 2010) and originate from an experiment conducted parallelly to the present one.
Discussion
Fire blight caused by the bacterium Erwinia amylovora (Burill) Winslow et al. spreads from North America to New Zealand and in the early 1950s first to the British Isles (Billing 2011) and subsequently to the continental Europe. Over 100 years ago Waite (1896) expressed the following opinion on the management of pear fire blight: “No disease has so completely baffled all attempts to find satisfactory remedy.” This remark of Waite is still true and till today fire blight is a great problem for pear, apple and other members of the family Rosaceae. There is no adequate chemical or other treatment for the elimination of the pathogen from plant material without destroying the plant tissues. Also, the treatment with antibiotic streptomycin is not a permanent solution and is forbidden in the EU.
There is a need for solutions which are environmentally friendly, durable and safe for consumers. One of the solutions is the breeding of highly resistant pear cultivars and rootstocks by exploiting genetic variation in the germplasm also with wild pear species (Bokszczanin et al. 2009). Because of that we aimed at determining the levels of resistance of old pear cultivars and breeding clones from different countries that can be used in future as the donors of genes, conferring resistance. Given a long generation time of pear and apple, the breeding progress may in some cases be enhanced by selecting parents with less than the highest level of resistance available but which retain more nearly acceptable horticultural characteristics. Out of 287 cultivars named prior to 1920, only 11% are resistant or highly resistant; out of 113 cultivars released between 1920 and 1978, about one-third were reported to be predominantly resistant (Lespinasse and Aldwinckle 2000). Currently in the World Source of fire blight resistance there are 78 cultivars of European pear resistant to fire blight (Postman 2008).
Resistance to fire blight is an important criterion in the resistant pear and apple breeding programs. Such programs were first developed in the USA and at present they are conducted in other countries, particularly in Europe, utilizing conventional and molecular methods (Fisher and Richter 1999; Lespinasse and Aldwinckle 2000; Sobiczewski et al. 2005; Toth 2005; Toth et al. 2006; Bokszczanin and Przybyla 2007, 2009, 2011; Bokszczanin et al. 2007; Peil et al. 2007, 2008; Kellerhals et al. 2008; Bokszczanin et al. 2009; Lagonenko et al. 2011; Persen et al. 2011). In the INRA pear breeding program carried out at Angers, France, Thibault (1981) developed an initial half-diallel program, including resistant American selections as parents and old European cultivars. Data from pear breeding program conducted at the Istituto Sperimentale per la Frutticoltura, Rome and Forli, Italy, indicate, that in order to combine fire blight resistance and fruit quality, selection for resistance within high-quality susceptible cultivars may be the most effective (Bagnara et al. 1996).
Various methods for evaluating fire blight infection severity were developed, e.g., the method by Gardner et al. (1980) or Le Lezec et al. (1997). Although Gardner scale was originally developed for Malus, it was also successfully applied in the pear breeding programs focused on the fire blight severity assessment of pear mapping populations (‘Doyenné du Comice’ and Asiatic species) and QTLs identification, determining fire blight resistance (Bokszczanin et al. 2009).
Our approach to evaluate fire blight severity is very useful and simply comparable to the ISV index developed by Le Lezec et al. (1997) since the index is based on necrosis percentage and subsequently transformed to the index regarding both the number of shoots of each genotype tested and classes of severity (first 0–20%, second 20–40%, third 40–60%, fourth 60–80%, and fifth 80–100%). Our statistical approach makes it possible to trace very precisely the plant response to pathogen infection determined by the genotype and time after infection and to establish progress of the disease in plants of different genetic background.
According to Le Lezec et al. (1997), ‘Doyenné du Comice’ is considered to be highly susceptible ranking to the fifth class (80–100% of necrosis). In our studies, it was classified as moderately susceptible with mean necrosis of 49.5%, and ranking together with ‘Abbé Fetel’ and ‘Bartlett’ to the third class after Le Lezec et al. (1997).
Our results showed a considerable variation in resistance to fire blight among the studied old cultivars and breeding clones. ‘Hessle’ appeared to be the most promising, a very resistant cultivar ranking to the first class of resistance according to both Gardner et al. (1980) and Le Lezec et al. (1997) scale similar to other main pear cultivars cultivated in the world: ‘Harrow Sweet’, ‘Beurré Bosc’, and ‘Coscia’.
The results of Pyrus–Erwinia amylovora inoculations suggest an incompatible interaction in the case of ‘Hessle’ and in the case of all other tested pear cvs and clones—a compatible host plant–pathogen interaction. Incompatibility is considered as a gene for gene mediated and determined by complementary dominant genes. This model predicts that plant resistance will occur only when a plant possesses a dominant ‘resistance gene’ (R) and the pathogen expresses the complementary dominant ‘avirulence gene’ (Avr). In our studies it was clearly shown that resistance/susceptibility depends on the genotype and as a response to pathogen it develops in time after the infection. Most of the genotypes showed quite low infection level 1 week after the inoculation. In regard to four phased ‘zigzag’ model proposed by Jones and Dangl (2006) it indicates that plant in the phase 1 of the model recognizes pathogen-associated molecular patterns (PAMPs) by the transmembrane pattern recognition receptors (PRRs), resulting in PAMP-triggered immunity (PTI) that can halt further colonization. In phase 2, successful pathogens deploy effectors that contribute to pathogen virulence as it happened in the case of susceptible plant genotypes. Effectors can interfere with PTI. This results in the effector-triggered susceptibility (ETS).
Only in the case of ‘Hessle’ it was possible to observe phase 3, in which a given effector is ‘specifically recognized’ by one of the NB-LRR proteins, resulting in the effector-triggered immunity (ETI). Recognition can be either indirect or through the direct NB-LRR recognition of an effector. ETI is an accelerated and amplified PTI response, resulting in disease resistance. Mechanism of plant resistance against necrotrophic bacteria, that kill host tissue during colonization, has not been elucidated till now.
Interestingly, only the ‘Hessle’ response to pathogen infection was stable across four consecutive weeks after inoculation. It indicates that resistance was triggered very quickly after immediate pathogen recognition, successfully maintained and resulting in overcoming pathogen attack. It is also worth to notice that besides well-known variability in plant response occurring after Erwinia amylovora infection depending, among others, on plant physiological state, in the case of resistant genotypes the plant response is sufficient, effective and stable.
In our studies the level of response to pathogen infection depends on the genotype and changes in time after inoculation also independently for each genotype. It indicates that different genotypes develop different resistance response.
References
Aldwinckle HS, Norelli JL (1981) Varietal differences in incidence of infection of apple blossoms and shoots by Erwinia amylovora (fire blight). Acta Hortic 117:71
Aldwinckle HS, Preczewski JL (1976) Susceptibility of vegetative tissues of apple cultivars to invasion by Erwinia amylovora. Phytopathology 66:1439–1444
Bagnara GL, Rivalta L, Laghi M, Quarta R (1996) Evaluation of fire blight resistance in pear: combining ability and breeding strategy. Acta Hortic 41:383–392
Bell RL, Janick J (1977) Estimation of heritability and combining ability for fire blight resistance in pear. J Amer Soc Hortic Sci 102(2):133–138
Bell RL, Quamme HA, Layne REC, Skirvin RM (1996) Pears. In: Janick J, Moore JN (eds) Fruit breeding, vol 1: tree and tropical fruits. Wiley, New York, pp 441–514
Bell AC, Ranney TG, Eaker TA (2004) Resistance to fire blight among flowering pears and quince. HortScience 40(2):413–415
Billing E (2011) Reflections on fire blight and questions. Acta Hortic 896:33–38
Bokszczanin K, Przybyla AA (2007) Application of AFLP-RGA method in molecular analysis of pear resistance to fire blight. In: Stich K, Halbwirth H (eds) COST Action 864. Pome fruit health research in Europe. Reports from the work group meetings 2007, pp 54–55
Bokszczanin K, Przybyla AA (2009) Preliminary molecular marker maps constructed by using an interspecific cross between Pyrus communis and P. ussuriensis. Acta Hortic 839:487–490
Bokszczanin KL, Przybyla AA (2011) QTLs for fire blight (Erwinia amylovora) resistance in Pyrus ussuriensis. Acta Hortic 896:371–373
Bokszczanin K, Krajewski P, Schollenberger M, Przybyla AA, Odziemkowski S (2007) Susceptibility of pear hybrids to fire blight (in Polish, English summary). Progress in Plant Protection/Postępy w Ochronie Roślin 47(2):48–54
Bokszczanin K, Dondini L, Przybyla AA (2009) First report on the presence of fire blight resistance in linkage group 11 of Pyrus ussuriensis Maxim. J Appl Genet 50(2):99–104
Bokszczanin K, Przybyla AA, Schollenberger M, Odziemkowski S (2010) Far-East species of pear—a potential source of fire blight resistance genes (in Polish). In: 53rd All-Poland conference of fruit trees protection. 25–26 Feb. 2010, Research Institute of Pomology and Floriculture, Skierniewice, Poland (ed.):59–62
Fisher C, Richter K (1999) Results on fire blight resistance in the Pillnitz apple breeding programme. Acta Hortic 489:279–285
Gardner RG, Cummins JN, Aldwinckle HS (1980) Inheritance of fire blight resistance in Malus in relation to rootstock breeding. J Amer Soc Hortic Sci 105(6):912–916
Jones JDG, Dangl JL (2006) The plant immune system. Nature 444:323–329
Kellerhals M, Szalatnay D, Hunziker K, Koller T, Frey J, Duffy B (2008) Selection for fire blight tolerance at Wadenswil. COST 864 Meet, Dresden, Germany, 5 Sept 2008
Lagonenko AL, Kudzina IV, Evtushenkov AN, Krugleevskiy, Kastitskaya MS, Kukharchyk NV (2011) Susceptibility of Belarusian apple and pear cultivars to fire blight. Acta Hortic 896:393–396
Le Lezec M, Lecomte P, Laurens F, Michelesi JC (1997) Sensibilité variétale au feu bactérien [3 parts]. Arb Fruit 503:57–61 504:33–37, 505:31–40
Lespinasse Y, Aldwinckle HS (2000) Breeding for resistance to fire blight. In: Vanneste JL (ed) Fire blight: the disease and its causative agent, Erwinia amylovora.. CABI Publishing, Wallingford, pp 253–273
McIntosh MS (1983) Analysis of combined experiments. Agron J 75:153–155
Monte-Corvo L, Goulão L, Oliveira C (2001) ISSR analysis of cultivars of pear and suitability of molecular markers for clone discrimination. J Amer Soc Hortic Sci 126(5):517–522
Peil A, Garcia-Libreros T, Richter K, Trognitz FC, Trognitz B, Hanke MV, Flachowsky H (2007) Strong evidence for fire blight resistance gene of Malus robusta located on linkage group 3. Plant Breed 126:470–475
Peil A, Hanke MV, Flachowsky H, Richter K, Garcia-Libreros T, Celton JM, Gardiner S, Horner M, Bus V (2008) Confirmation of the fire blight QTL of Malus × robusta 5 on linkage group 3. Acta Hortic 793:297–303
Persen U, Gottsberger R, Reisenzein H (2011) Spread of Erwinia amylovora in apple and pear trees of different cultivars after artificial inoculation. Acta Hortic 896:319–330
Postman JD (2008) The USDA Quince and Pear Genebank in Oregon, a world source of fire blight resistance. Acta Hortic 793:357–362
Sobiczewski P, Żurawicz E, Berczyński S (2005) Trends in breeding of cultivars and rootstocks of apple and pear resistant to fire blight (in Polish, English summary). Postępy Nauk Rolniczych 6:39–49
StatSoft Inc (2005) STATISTICA (data analysis software system), version 7.1. www.statsoft.com
Steel RGD, Torrie JH, Dickey DA (1997) Principles and procedures of statistics: a biometrical approach. McGraw-Hill, New York
Thibault B (1981) Pear breeding for fire blight resistance: program and first studies in France. Acta Hortic 117:63–69
Toth M (2005) Pomology of gene resources bearing resistance to Erwinia amylovora. Int J Hortic Sci 11(3):29–33
Toth M, Kasa K, Gondor M, Honty K, Hevesi M (2006) First results of fire blight resistance screening in a Hungarian breeding programme. Acta Hortic 704:545–549
Van der Zwet T, Keil HL (1979) Fire blight: a bacterial disease of rosaceous plants. USDA Agr Hdbk 510
Van der Zwet T, Oitto WA, Westwood MN (1974) Variability in degree of fire blight resistance within and between Pyrus species, interspecific hybrids, and seedling progenies. Euphytica 23:295–303
Waite MB (1896) The cause of prevention of pear blight. USDA Yearbook 1895:295–300
Acknowledgments
This research was conducted in the framework of COST Action 864.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by D. Treutter.
A contribution to the Special Issue: Pome Fruit Health
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Przybyla, A.A., Bokszczanin, K.L., Schollenberger, M. et al. Fire blight resistance of pear genotypes from different European countries. Trees 26, 191–197 (2012). https://doi.org/10.1007/s00468-011-0646-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-011-0646-7





