Abstract
The primary hyperoxalurias (PH 1, 2, and 3) are rare autosomal recessive disorders of glyoxylate metabolism resulting in hepatic overproduction of oxalate. Clinical presentations that should prompt consideration of PH include kidney stones, nephrocalcinosis, and kidney failure of unknown etiology, especially with echogenic kidneys on ultrasound. PH1 is the most common and severe of the primary hyperoxalurias with a high incidence of kidney failure as early as infancy. Until the recent availability of a novel RNA interference (RNAi) agent, PH care was largely supportive of eventual need for kidney/liver transplantation in PH1 and PH2. Together with the Oxalosis and Hyperoxaluria Foundation, the authors developed a diagnostic algorithm for PH1 and in this report outline best clinical practices related to its early diagnosis, supportive treatment, and long-term management, including the use of the novel RNAi. PH1-focused approaches to dialysis and kidney/liver transplantation for PH patients with progression to chronic kidney disease/kidney failure and systemic oxalosis are suggested. Therapeutic advances for this devastating disease heighten the importance of early diagnosis and informed treatment.
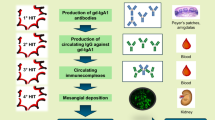
Graphical abstract

A higher resolution version of the Graphical abstract is available as Supplementary information

Similar content being viewed by others
References
Demoulin N, Aydin S, Gillion V, Morelle J, Jadoul M (2022) Pathophysiology and management of hyperoxaluria and oxalate nephropathy: a review. Am J Kidney Dis 79:717–727
Fargue S, Acquaviva Bourdain C (2022) Primary hyperoxaluria type 1: pathophysiology and genetics. Clin Kidney J 15(Suppl 1):i4–i8
Danpure CJ, Jennings R (1986) Peroxisomal alanine:glyoxylate aminotransferase deficiency in primary hyperoxaluria type I. FEBS Lett 201:20–24
Webster KE, Ferree PM, Holmes RP, Cramer SD (2000) Identification of missense, nonsense, and deletion mutations in the GRHPR gene in patients with primary hyperoxaluria type II (PH2). Hum Genet 107:176–185
Monico CG, Rossetti S, Belostotsky R, Cogal AG, Herges RM, Seide BM, Olson JB, Bergstrahl EJ, Williams HJ, Haley WE, Frishberg Y, Milliner DS (2011) Primary hyperoxaluria type III gene HOGA1 (formerly DHDPSL) as a possible risk factor for idiopathic calcium oxalate urolithiasis. Clin J Am Soc Nephrol 6:2289–2295
Martin-Higueras C, Garrelfs SF, Groothoff JW, Jacob DE, Moochhala SH, Bacchetta J, Acquaviva C, Zaniew M, Sikora P, Beck BB, Hoppe B (2021) A report from the European Hyperoxaluria Consortium (OxalEurope) Registry on a large cohort of patients with primary hyperoxaluria type 3. Kidney Int 100:621–635
Groothoff JW, Metry E, Deesker L, Garrelfs S, Acquaviva C, Almardini R, Beck BB, Boyer O, Cerkauskiene R, Ferraro PM, Groen LA, Gupta A, Knebelmann B, Mandrile G, Moochhala SS, Prytula A, Putnik J, Rumsby G, Soliman NA, Somani B, Bacchetta J (2023) Clinical practice recommendations for primary hyperoxaluria: an expert consensus statement from ERKNet and OxalEurope. Nat Rev Nephrol 19:194-211
Niederberger M, Spranger J (2020) Delphi technique in health sciences: a map. Front Public Health 8:457
Sas DJ, Harris C, Milliner DS (2019) Recent advances in the identification and management of inherited hyperoxalurias. Urolithiasis 47:79–89
Edvardsson VO, Goldfarb DS, Lieske JC, Beara-Lasic L, Anglani F, Milliner DS, Palsson R (2013) Hereditary causes of kidney stones and chronic kidney disease. Pediatr Nephrol 28:1923–1942
Edvardsson V, Sas D (2022) Urinary stone disease and nephrocalcinosis. In Emma F, et al (ed) Pediatric Nephrology, 8th edn. Springer, Cham
Marangella M, Petrarulo M, Vitale C, Daniele PG, Sammartano S, Cosseddu D, Linari F (1991) Serum calcium oxalate saturation in patients on maintenance haemodialysis for primary hyperoxaluria or oxalosis-unrelated renal diseases. Clin Sci (Lond) 81:483–490
Milliner DS, McGregor TL, Thompson A, Dehmel B, Knight J, Rosskamp R, Blank M, Yang S, Fargue S, Rumsby G, Groothoff J, Allain M, West M, Hollander K, Lowther WT, Lieske JC (2020) End points for clinical trials in primary hyperoxaluria. Clin J Am Soc Nephrol 15:1056–1065
Milliner DS, Cochat P, Hulton SA, Harambat J, Banos A, Dehmel B, Lindner E (2021) Plasma oxalate and eGFR are correlated in primary hyperoxaluria patients with maintained kidney function-data from three placebo-controlled studies. Pediatr Nephrol 36:1785–1793
Hoppe B, Kemper MJ, Bökenkamp A, Portale AA, Cohn RA, Langman CB (1999) Plasma calcium oxalate supersaturation in children with primary hyperoxaluria and end-stage renal failure. Kidney Int 56:268–274
Mandrile G, Beck B, Acquaviva C, Rumsby G, Deesker L, Garrelfs S, Gupta A, Bacchetta J, Groothoff J; OxalEurope Consortium/Erknet Guideline Workgroup On Hyperoxaluria (2023) Genetic assessment in primary hyperoxaluria: why it matters. Pediatr Nephrol 38:625–634
Sas DJ, Enders FT, Mehta RA, Tang X, Zhao F, Seide BM, Milliner DS, Lieske JC (2020) Clinical features of genetically confirmed patients with primary hyperoxaluria identified by clinical indication versus familial screening. Kidney Int 97:786–792
Pearle MS, Goldfarb DS, Assimos DG, Curhan G, Denu-Ciocca CJ, Matlaga BR, Monga M, Penniston KL, Preminger GM, Turk TM, White JR; American Urological Assocation (2014) Medical management of kidney stones: AUA guideline. J Urol 192:316–324
Gupta A, Somers MJG, Baum MA (2022) Treatment of primary hyperoxaluria type 1. Clin Kidney J 15(Suppl 1):i9–i13
Cochat P, Groothoff J (2013) Primary hyperoxaluria type 1: practical and ethical issues. Pediatr Nephrol 28:2273–2281
Leumann E, Hoppe B, Neuhaus T (1993) Management of primary hyperoxaluria: efficacy of oral citrate administration. Pediatr Nephrol 7:207–211
Meschi T, Maggiore U, Fiaccadori E, Schianchi T, Bosi S, Adorni G, Ridolo E, Guerra A, Allegri F, Novarini A, Borghi L (2004) The effect of fruits and vegetables on urinary stone risk factors. Kidney Int 66:2402–2410
Massey L (2005) Magnesium therapy for nephrolithiasis. Magnes Res 18:123–126
Cochat P, Hulton SA, Acquaviva C, Danpure CJ, Daudon M, De Marchi M, Fargue S, Groothoff J, Harambat J, Hoppe B, Jamieson NV, Kemper MJ, Mandrile G, Marangella M, Picca S, Rumsby G, Salido E, Straub M, van Woerden CS; OxalEurope (2012) Primary hyperoxaluria type 1: indications for screening and guidance for diagnosis and treatment. Nephrol Dial Transplant 27:1729–1736
Noori N, Honarkar E, Goldfarb DS, Kalantar-Zadeh K, Taheri M, Shakhssalim N, Parvin M, Basiri A (2014) Urinary lithogenic risk profile in recurrent stone formers with hyperoxaluria: a randomized controlled trial comparing DASH (Dietary Approaches to Stop Hypertension)-style and low-oxalate diets. Am J Kidney Dis 63:456–463
Singh P, Chebib FT, Cogal AG, Gavrilov DK, Harris PC, Lieske JC (2020) Pyridoxine responsiveness in a type 1 primary hyperoxaluria patient with a rare (atypical). Kidney Int Rep 5:955–958
Mandrile G, van Woerden CS, Berchialla P, Beck BB, Acquaviva Bourdain C, Hulton SA, Rumsby G; OxalEurope Consortium (2014) Data from a large European study indicate that the outcome of primary hyperoxaluria type 1 correlates with the AGXT mutation type. Kidney Int 86:1197–1204
Fargue S, Rumsby G, Danpure CJ (2013) Multiple mechanisms of action of pyridoxine in primary hyperoxaluria type 1. Biochim Biophys Acta 1832:1776–1783
Garrelfs SF, Frishberg Y, Hulton SA, Koren MJ, O’Riordan WD, Cochat P, Deschênes G, Shasha-Lavsky H, Saland JM, Van’t Hoff WG, Fuster DG, Magen D, Moochhala SH, Schalk G, Simkova E, Groothoff JW, Sas DJ, Meliambro KA, Lu J, Sweetser MT, Garg PP, Vaishnaw AK, Gansner JM, McGregor TL, Lieske JC; ILLUMINATE-A Collaborators (2021) Lumasiran, an RNAi Therapeutic for Primary Hyperoxaluria Type 1. N Engl J Med 384(13):1216–1226. https://doi.org/10.1056/NEJMoa2021712. PMID: 33789010
Frishberg Y, Deschênes G, Groothoff JW, Hulton SA, Magen D, Harambat J, Van't Hoff WG, Lorch U, Milliner DS, Lieske JC, Haslett P, Garg PP, Vaishnaw AK, Talamudupula S, Lu J, Habtemariam BA, Erbe DV, McGregor TL, Cochat P; study collaborators (2021) Phase 1/2 study of lumasiran for treatment of primary hyperoxaluria type 1: a placebo-controlled randomized clinical trial. Clin J Am Soc Nephrol 16:1025–1036
Sas DJ, Magen D, Hayes W, Shasha-Lavsky H, Michael M, Schulte I, Sellier-Leclerc AL, Lu J, Seddighzadeh A, Habtemariam B, McGregor TL, Fujita KP, Frishberg Y; ILLUMINATE-B Workgroup (2022) Phase 3 trial of lumasiran for primary hyperoxaluria type 1: a new RNAi therapeutic in infants and young children. Genet Med 24:654–662
Hayes W, Sas DJ, Magen D, Shasha-Lavsky H, Michael M, Sellier-Leclerc AL, Hogan J, Ngo T, Sweetser MT, Gansner JM, McGregor TL, Frishberg Y (2023) Efficacy and safety of lumasiran for infants and young children with primary hyperoxaluria type 1: 12-month analysis of the phase 3 ILLUMINATE-B trial. Pediatr Nephrol 38:1075–1086
Michael M, Groothoff JW, Shasha-Lavsky H, Lieske JC, Frishberg Y, Simkova E, Sellier-Leclerc AL, Devresse A, Guebre-Egziabher F, Bakkaloglu SA, Mourani C, Saqan R, Singer R, Willey R, Habtemariam B, Gansner JM, Bhan I, McGregor T, Magen D (2023) Lumasiran for advanced primary hyperoxaluria type 1: phase 3 ILLUMINATE-C trial. Am J Kidney Dis 81(2):145-155.e1
Stokes F, Acquaviva-Bourdain C, Hoppe B, Lieske JC, Lindner E, Toulson G, Vaz FM, Rumsby G (2020) Plasma oxalate: comparison of methodologies. Urolithiasis 48:473–480
Stevens PE, Levin A; Kidney Disease: Improving Global Outcomes Chronic Kidney Disease Guideline Development Work Group Members (2013) Evaluation and management of chronic kidney disease: synopsis of the kidney disease: improving global outcomes 2012 clinical practice guideline. Ann Intern Med 158:825–830
Derveaux T, Delbeke P, Walraedt S, Raes A, Van Laecke S, Leroy BP, De Zaeytijd J (2016) Detailed clinical phenotyping of oxalate maculopathy in primary hyperoxaluria type 1 and review of the literature. Retina 36:2227–2235
Lagies R, Beck BB, Hoppe B, Sreeram N, Udink Ten Cate FE (2013) Apical sparing of longitudinal strain, left ventricular rotational abnormalities, and short-axis dysfunction in primary hyperoxaluria type 1. Circ Heart Fail 6:e45–e47
Sas DJ, Enders FT, Gunderson TM, Mehta RA, Olson JB, Seide BM, Banks CJ, Dehmel B, Pellikka PA, Lieske JC, Milliner DS (2021) Natural history of clinical, laboratory, and echocardiographic parameters of a primary hyperoxaluria cohort on long term hemodialysis. Front Med (Lausanne) 8:592357
Fisher D, Hiller N, Drukker A (1995) Oxalosis of bone: report of four cases and a new radiological staging. Pediatr Radiol 25:293–295
Ben-Shalom E, Cytter-Kuint R, Rinat C, Becker-Cohen R, Tzvi-Behr S, Goichberg J, Peles V, Frishberg Y (2021) Long-term complications of systemic oxalosis in children-a retrospective single-center cohort study. Pediatr Nephrol 36:3123–3132
Julian BA, Faugere MC, Malluche HH (1987) Oxalosis in bone causing a radiographical mimicry of renal osteodystrophy. Am J Kidney Dis 9:436–440
Rootman MS, Mozer-Glassberg Y, Gurevich M, Schwartz M, Konen O (2018) Imaging features of primary hyperoxaluria. Clin Imaging 52:370–376
El Hage S, Ghanem I, Baradhi A, Mourani C, Mallat S, Dagher F, Kharrat K (2008) Skeletal features of primary hyperoxaluria type 1, revisited. J Child Orthop 2:205–210
Merz LM, Born M, Kukuk G, Sprinkart AM, Becker I, Martin-Higueras C, Hoppe B (2023) Correction to: three Tesla magnetic resonance imaging detects oxalate osteopathy in patients with primary hyperoxaluria type I. Pediatr Nephrol 38:2497–2498
Merz LM, Born M, Kukuk G, Sprinkart AM, Becker I, Martin-Higueras C, Hoppe B (2023) Three Tesla magnetic resonance imaging detects oxalate osteopathy in patients with primary hyperoxaluria type I. Pediatr Nephrol 38:2083–2092
Bacchetta J, Fargue S, Boutroy S, Basmaison O, Vilayphiou N, Plotton I, Guebre-Egziabher F, Dohin B, Kohler R, Cochat P (2010) Bone metabolism in oxalosis: a single-center study using new imaging techniques and biomarkers. Pediatr Nephrol 25:1081–1089
Tonnelet D, Benali K, Rasmussen C, Goulenok T, Piekarski E (2020) Diffuse hypermetabolic bone marrow infiltration in severe primary hyperoxaluria on FDG PET. Clin Nucl Med 45(6):e296–e298
Bacchetta J, Boivin G, Cochat P (2016) Bone impairment in primary hyperoxaluria: a review. Pediatr Nephrol 31:1–6
Monico CG, Rossetti S, Olson JB, Milliner DS (2005) Pyridoxine effect in type I primary hyperoxaluria is associated with the most common mutant allele. Kidney Int 67:1704–1709
Lorenz EC, Lieske JC, Seide BM, Meek AM, Olson JB, Bergstralh EJ, Milliner DS (2014) Sustained pyridoxine response in primary hyperoxaluria type 1 recipients of kidney alone transplant. Am J Transplant 14:1433–1438
Hopp K, Cogal AG, Bergstralh EJ, Seide BM, Olson JB, Meek AM, Lieske JC, Milliner DS, Harris PC; Rare Kidney Stone Consortium (2015) Phenotype-genotype correlations and estimated carrier frequencies of primary hyperoxaluria. J Am Soc Nephrol 26:2559–2570
Mandrile G, Beck B, Acquaviva C, Rumsby G, Deesker L, Garrelfs S, Gupta A, Bacchetta J, Groothoff J; OxalEurope Consortium/Erknet Guideline Workgroup On Hyperoxaluria (2022) Genetic assessment in primary hyperoxaluria: why it matters. Pediatr Nephrol 38:625-634
Milliner DS, Eickholt JT, Bergstralh EJ, Wilson DM, Smith LH (1994) Results of long-term treatment with orthophosphate and pyridoxine in patients with primary hyperoxaluria. N Engl J Med 331:1553–1558
Berini SE, Tracy JA, Engelstad JK, Lorenz EC, Milliner DS, Dyck PJ (2015) Progressive polyradiculoneuropathy due to intraneural oxalate deposition in type 1 primary hyperoxaluria. Muscle Nerve 51:449–454
Biebuyck N, Destombes C, Prakash R, Boyer O (2023) Is withdrawal of nocturnal hyperhydration possible in children with primary hyperoxaluria treated with RNAi? J Nephrol 36:1473–1476
Baum MA, Langman C, Cochat P, Lieske JC, Moochhala SH, Hamamoto S, Satoh H, Mourani C, Ariceta G, Torres A, Wolley M, Belostotsky V, Forbes TA, Groothoff J, Hayes W, Tönshoff B, Takayama T, Rosskamp R, Russell K, Zhou J, Amrite A, Hoppe B; PHYOX2 study investigators (2023) PHYOX2: a pivotal randomized study of nedosiran in primary hyperoxaluria type 1 or 2. Kidney Int 103:207–217
Martin-Higueras C, Feldkötter M, Hoppe B (2021) Is stiripentol truly effective for treating primary hyperoxaluria? Clin Kidney J 14:442–444
Martinez-Turrillas R, Martin-Mallo A, Rodriguez-Diaz S, Zapata-Linares N, Rodriguez-Marquez P, San Martin-Uriz P, Vilas-Zornoza A, Calleja-Cervantes ME, Salido E, Prosper F, Rodriguez-Madoz JR (2022) CRISPR-Cas9 inhibition of hepatic LDH as treatment of primary hyperoxaluria. Mol Ther Methods Clin Dev 25:137–146
Sellier-Leclerc AL, Metry E, Clave S, Perrin P, Acquaviva-Bourdain C, Levi C, Crop M, Caillard S, Moulin B, Groothoff J, Bacchetta J (2023) Isolated kidney transplantation under lumasiran therapy in primary hyperoxaluria type 1: a report of five cases. Nephrol Dial Transplant 38:517–521
Breeggemann MC, Gluck SL, Stoller ML, Lee MM (2023) A case report of kidney-only transplantation in primary hyperoxaluria type 1: a novel approach with the use of nedosiran. Case Rep Nephrol Dial 13:63–69
Zhao F, Bergstralh EJ, Mehta RA, Vaughan LE, Olson JB, Seide BM, Meek AM, Cogal AG, Lieske JC, Milliner DS; Investigators of Rare Kidney Stone Consortium (2016) Predictors of incident ESRD among patients with primary hyperoxaluria presenting prior to kidney failure. Clin J Am Soc Nephrol 11:119–126
Shah RJ, Vaughan LE, Enders FT, Milliner DS, Lieske JC (2020) Plasma oxalate as a predictor of kidney function decline in a primary hyperoxaluria cohort. Int J Mol Sci 21:3608
Watts RW, Veall N, Purkiss P (1984) Oxalate dynamics and removal rates during haemodialysis and peritoneal dialysis in patients with primary hyperoxaluria and severe renal failure. Clin Sci (Lond) 66:591–597
Marangella M, Cosseddu D, Petrarulo M, Vitale C, Linari F (1993) Thresholds of serum calcium oxalate supersaturation in relation to renal function in patients with or without primary hyperoxaluria. Nephrol Dial Transplant 8:1333–1337
Marangella M, Petrarulo M, Cosseddu D, Vitale C, Linari F (1992) Oxalate balance studies in patients on hemodialysis for type I primary hyperoxaluria. Am J Kidney Dis 19:546–553
Illies F, Bonzel KE, Wingen AM, Latta K, Hoyer PF (2006) Clearance and removal of oxalate in children on intensified dialysis for primary hyperoxaluria type 1. Kidney Int 70:1642–1648
Ermer T, Kopp C, Asplin JR, Granja I, Perazella MA, Reichel M, Nolin TD, Eckardt KU, Aronson PS, Finkelstein FO, Knauf F (2017) Impact of regular or extended hemodialysis and hemodialfiltration on plasma oxalate concentrations in patients with end-stage renal disease. Kidney Int Rep 2:1050–1058
Franssen CF (2005) Oxalate clearance by haemodialysis–a comparison of seven dialysers. Nephrol Dial Transplant 20:1916–1921
Tang X, Bergstralh EJ, Mehta RA, Vrtiska TJ, Milliner DS, Lieske JC (2015) Nephrocalcinosis is a risk factor for kidney failure in primary hyperoxaluria. Kidney Int 87:623–631
Díaz C, Catalinas FG, de Alvaro F, Torre A, Sánchez C, Costero O (2004) Long daily hemodialysis sessions correct systemic complications of oxalosis prior to combined liver-kidney transplantation: case report. Ther Apher Dial 8:52–55
Plumb TJ, Swee ML, Fillaus JA (2013) Nocturnal home hemodialysis for a patient with type 1 hyperoxaluria. Am J Kidney Dis 62:1155–1159
Yamauchi T, Quillard M, Takahashi S, Nguyen-Khoa M (2001) Oxalate removal by daily dialysis in a patient with primary hyperoxaluria type 1. Nephrol Dial Transplant 16:2407–2411
Tang X, Voskoboev NV, Wannarka SL, Olson JB, Milliner DS, Lieske JC (2014) Oxalate quantification in hemodialysate to assess dialysis adequacy for primary hyperoxaluria. Am J Nephrol 39:376–382
Cochat P, Fargue S, Harambat J (2010) Primary hyperoxaluria type 1: strategy for organ transplantation. Curr Opin Organ Transplant 15:590–593
Cornell LD, Amer H, Viehman JK, Mehta RA, Lieske JC, Lorenz EC, Heimbach JK, Stegall MD, Milliner DS; Rare Kidney Stone Consortium Primary Hyperoxaluria (RKSC PH) investigators (2022) Posttransplant recurrence of calcium oxalate crystals in patients with primary hyperoxaluria: incidence, risk factors, and effect on renal allograft function. Am J Transplant 22:85–95
Villani V, Gupta N, Elias N, Vagefi PA, Markmann JF, Paul E, Traum AZ, Yeh H (2017) Bilateral native nephrectomy reduces systemic oxalate level after combined liver-kidney transplant: a case report. Pediatr Transplant 21(3).
Mor E, Nesher E, Ben-Ari Z, Weissman I, Shaharabani E, Eizner S, Solomonov E, Rahamimov R, Braun M (2013) Sequential liver and kidney transplantation from a single living donor in two young adults with primary hyperoxaluria type 1. Liver Transpl 19:646–648
Metry EL, van Dijk LMM, Peters-Sengers H, Oosterveld MJS, Groothoff JW, Ploeg RJ, Stel VS, Garrelfs SF (2021) Transplantation outcomes in patients with primary hyperoxaluria: a systematic review. Pediatr Nephrol 36:2217–2226
Xiang J, Chen Z, Xu F, Mei S, Li Z, Zhou J, Dong Y, Gu Y, Huang Z, Hu Z (2020) Outcomes of liver-kidney transplantation in patients with primary hyperoxaluria: an analysis of the scientific registry of transplant recipients database. BMC Gastroenterol 20:208
Dhondup T, Lorenz EC, Milliner DS, Lieske JC. (2018) Combined liver-kidney transplantation for primary hyperoxaluria type 2: a case report. Am J Transplant 18:253–257
Bergstralh EJ, Monico CG, Lieske JC, Herges RM, Langman CB, Hoppe B, Milliner DS; IPHR Investigators (2010) Transplantation outcomes in primary hyperoxaluria. Am J Transplant 10:2493–2501
Devresse A, Cochat P, Godefroid N, Kanaan N (2020) Transplantation for primary hyperoxaluria type 1: designing new strategies in the era of promising therapeutic perspectives. Kidney Int Rep 5:2136–2145
Lee E, Ramos-Gonzalez G, Rodig N, Elisofon S, Vakili K, Kim HB (2018) Bilateral native nephrectomy to reduce oxalate stores in children at the time of combined liver–kidney transplantation for primary hyperoxaluria type 1. Pediatr Nephrol 33:881–887
Acknowledgements
We are grateful to the Oxalosis and Hyperoxaluria Foundation for convening the panel of international experts in PH. Kim Hollander, Executive Director, and Julie Bertarelli, Operations Coordinator, provided invaluable guidance and administrative support. We thank Maya Vaishnaw for design assistance with the diagnostic algorithm and graphic abstract.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
MM: Alnylam Pharmaceuticals-served as site PI for use of lumasiran for PH1; PI longitudinal study (BONAPH1DE); Novo Nordisk Inc: served in the advisory board; EH: Research funding: Dicerna; Consulting fees, Advisory Board chair, speaker fees and travel funding from Alnylam; Consulting fees from Arbor Biotechnologies; DM: Research funding—Dicerna, Alnylam, OxThera; Advisory Boards—Synlogic, Mirum Pharmaceuticals, Consulting agreements—Oxthera, Dicerna, Alnylam; YF: Alnylam – consulting fees, research funding; Dicerna – travel grant to attend investigators’ meeting; DJS: Received grant funding for research from Alnylam and Dicerna Pharmaceuticals; JC: None; LC: Nuwellis-Consultant; KP: None; JS: Alynylam: research funding, consulting fees, NovoNordisk: scientific advisory; MJGS: Advisory Boards for Alnylam Pharmaceuticals, Cantero Therapeutics, Dicerna Pharmaceuticals, and BioPorto. Chairs a Data Safety Monitoring Board for a clinical trial for Dicerna. Member of the Scientific Advisory Board of the Oxalosis and Hyperoxaluria Foundation; MAB: Novo Nordisk (previously Dicerna): scientific advisory, PI clinical trial Nedosiran; Alnylam: scientific advisory, PI longitudinal study (BONAPH1DE); Chinook: scientific advisory; Cantero (previously Orfan): scientific advisory; UpToDate: author and section editor.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Michael J. G. Somers and Michelle A. Baum contributed equally as final or last authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Michael, M., Harvey, E., Milliner, D.S. et al. Diagnosis and management of primary hyperoxalurias: best practices. Pediatr Nephrol (2024). https://doi.org/10.1007/s00467-024-06328-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00467-024-06328-2