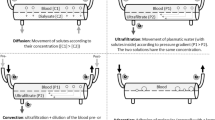
Abstract
Background
Continuous kidney replacement therapy (CKRT) has been expanded from simple kidney replacement therapy to the field of critical illness in children. However, CKRT is rarely used in critically ill neonates in the neonatal intensive care unit (NICU). This study aimed to describe patients’ clinical characteristics at admission and CKRT initiation, CKRT effects, short-term outcomes, and predictors of death in critically ill neonates.
Methods
A 7-year single-center retrospective study in a tertiary NICU.
Results
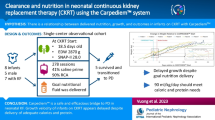
Thirty-nine critically ill neonates received CKRT between May 2015 and April 2022 with a mortality rate of 35.9%. The most common primary diagnosis was neonatal sepsis in 15 cases (38.5%). Continuous veno-venous hemodiafiltration and continuous veno-venous hemofiltration were applied in 43.6% and 56.4% of neonates, respectively. The duration of CKRT was 44 (18, 72) h. Thirty-one patients (79.5%) had complications due to CKRT-related adverse events, and the most common complication was thrombocytopenia. Approximately 12 h after the CKRT initiation, urine volume, mean arterial pressure, and pH were increased, and serum creatinine, blood urea nitrogen, and blood lactate were decreased. In the multivariate logistic regression analysis, neonatal critical illness score [odds ratio 0.886 (0.786 ~ 0.998), P = 0.046] was an independent risk factor for death in critically ill neonates who received CKRT.
Conclusions
CKRT can be an effective and feasible technique in critically ill neonates, but the overall mortality and CKRT-related complications are relatively high. Furthermore, the probability of death is greater among neonates with greater severity of illness at CKRT initiation.
Graphical abstract

A higher resolution version of the Graphical abstract is available as Supplementary information


Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Gaudry S, Grolleau F, Barbar S, Martin-Lefevre L, Pons B, Boulet É, Boyer A, Chevrel G, Montini F, Bohe J, Badie J, Rigaud JP, Vinsonneau C, Porcher R, Quenot JP, Dreyfuss D (2022) Continuous renal replacement therapy versus intermittent hemodialysis as first modality for renal replacement therapy in severe acute kidney injury: a secondary analysis of AKIKI and IDEAL-ICU studies. Crit Care 26:93
Yu G, Tao S, Jin Y, Li W, Hu Z, Fang X (2022) Ultrasound dynamic monitoring of IVCD to guide application of CRRT in patients with renal failure combined with acute heart failure. Sci Rep 12:14041
John JC, Taha S, Bunchman TE (2019) Basics of continuous renal replacement therapy in pediatrics. Kidney Res Clin Pract 38:455–461
Ames EG, Powell C, Engen RM, Weaver DJ Jr, Mansuri A, Rheault MN, Sanderson K, Lichter-Konecki U, Daga A, Burrage LC, Ahmad A, Wenderfer SE, Luckritz KE (2022) Multisite retrospective review of outcomes in renal replacement therapy for neonates with inborn errors of metabolism. J Pediatr 246:116–122
Jarrell AS, Crow JR, Strout SE, Kruer RM, Toman LP, Dioverti-Prono MV, Lees L, Avery RK, Marzinke MA (2021) Valganciclovir dosing for cytomegalovirus prophylaxis in solid-organ transplant recipients on continuous veno-venous hemodialysis. Clin Infect Dis 73:101–106
Sohn YB, Paik KH, Cho HY, Kim SJ, Park SW, Kim ES, Chang YS, Park WS, Choi YH, Jin DK (2012) Continuous renal replacement therapy in neonates weighing less than 3 kg. Korean J Pediatr 55:286–292
Wu CY, Lin YC, Chen CC (2021) A case series: continuous kidney replacement therapy in neonates with low body weight. Front Pediatr 9:769220
Zarbock A, Küllmar M, Kindgen-Milles D, Wempe C, Gerss J, Brandenburger T, Dimski T, Tyczynski B, Jahn M, Mülling N, Mehrländer M, Rosenberger P, Marx G, Simon TP, Jaschinski U, Deetjen P, Putensen C, Schewe JC, Kluge S, Jarczak D, Slowinski T, Bodenstein M, Meybohm P, Wirtz S, Moerer O, Kortgen A, Simon P, Bagshaw SM, Kellum JA, Meersch M (2020) Effect of regional citrate anticoagulation vs systemic heparin anticoagulation during continuous kidney replacement therapy on dialysis filter life span and mortality among critically ill patients with acute kidney injury: a randomized clinical trial. JAMA 324:1629–1639
Buccione E, Guzzi F, Colosimo D, Tedesco B, Romagnoli S, Ricci Z, L’Erario M, Villa G (2021) Continuous renal replacement therapy in critically ill children in the pediatric intensive care unit: a retrospective analysis of real-life prescriptions, complications, and outcomes. Front Pediatr 9:696798
Nishimi S, Sugawara H, Onodera C, Toya Y, Furukawa H, Konishi Y, Sotodate G, Matsumoto A, Ishikawa K, Oyama K (2019) Complications during continuous renal replacement therapy in critically ill neonates. Blood Purif 47(Suppl 2):74–80
Sanderson KR, Warady B, Carey W, Tolia V, Boynton MH, Benjamin DK, Jackson W, Laughon M, Clark RH, Greenberg RG (2022) Mortality risk factors among infants receiving dialysis in the neonatal intensive care unit. J Pediatr 242:159–165
Nada A, Bonachea EM, Askenazi DJ (2017) Acute kidney injury in the fetus and neonate. Semin Fetal Neonatal Med 22:90–97
Pediatric Group of Emergency Branch of Chinese Medical Association, Emergency Group and Neonatal Group of Pediatric Branch of Chinese Medical Association (2001) Neonatal critical illness score (draft). Chin J Pediatr 39:42–43
Sandrio S, Krebs J, Leonardy E, Thiel M, Schoettler JJ (2022) Vasoactive inotropic score as a prognostic factor during (cardio-) respiratory ECMO. J Clin Med 11:2390
Mduma E, Halidou T, Kaboré B, Walongo T, Lompo P, Museveni J, Gidabayda J, Gratz J, Guga G, Kimathi C, Liu J, Mdoe P, Moshiro R, Petzold M, Singlovic J, Guillerm M, Gomes MF, Houpt ER, Halleux CM (2022) Etiology of severe invasive infections in young infants in rural settings in sub-Saharan Africa. PLoS One 17:e0264322
Hingorani S, Schmicker RH, Brophy PD, Heagerty PJ, Juul SE, Goldstein SL, Askenazi D (2021) Severe acute kidney injury and mortality in extremely low gestational age neonates. Clin J Am Soc Nephrol 16:862–869
Matsushita FY, Krebs VLJ, de Carvalho WB (2022) Association between fluid overload and mortality in newborns: a systematic review and meta-analysis. Pediatr Nephrol 37:983–992
Pontoizeau C, Roda C, Arnoux JB, Vignolo-Diard P, Brassier A, Habarou F, Barbier V, Grisel C, Abi-Warde MT, Boddaert N, Kuster A, Servais A, Kaminska A, Hennequin C, Dupic L, Lesage F, Touati G, Valayannopoulos V, Chadefaux-Vekemans B, Oualha M, Eisermann M, Ottolenghi C, de Lonlay P (2020) Neonatal factors related to survival and intellectual and developmental outcome of patients with early-onset urea cycle disorders. Mol Genet Metab 130:110–117
Starr MC, Charlton JR, Guillet R, Reidy K, Tipple TE, Jetton JG, Kent AL, Abitbol CL, Ambalavanan N, Mhanna MJ, Askenazi DJ, Selewski DT, Harer MW (2021) Advances in neonatal acute kidney injury. Pediatrics 148:e2021051220
Crosier J, Whitaker M, Lambert HJ, Wellman P, Nyman A, Coulthard MG (2022) In vitro measurements of ultrafiltration precision in hemofiltration and hemodialysis devices used in infants. Pediatr Nephrol 37:3189–3194
Celik M, Akdeniz O, Ozgun N, Ipek MS, Ozbek MN (2019) Short-term results of continuous venovenous haemodiafiltration versus peritoneal dialysis in 40 neonates with inborn errors of metabolism. Eur J Pediatr 178:829–836
Garzotto F, Vidal E, Ricci Z, Paglialonga F, Giordano M, Laforgia N, Peruzzi L, Bellettato M, Murer L, Ronco C (2020) Continuous kidney replacement therapy in critically ill neonates and infants: a retrospective analysis of clinical results with a dedicated device. Pediatr Nephrol 35:1699–1705
Diane Mok TY, Tseng MH, Chiang MC, Lin JL, Chu SM, Hsu JF, Lien R (2018) Renal replacement therapy in the neonatal intensive care unit. Pediatr Neonatol 59:474–480
Cai C, Qiu G, Hong W, Shen Y, Gong X (2020) Clinical effect and safety of continuous renal replacement therapy in the treatment of neonatal sepsis-related acute kidney injury. BMC Nephrol 21:286
Weil MH, Afifi AA (1970) Experimental and clinical studies on lactate and pyruvate as indicators of the severity of acute circulatory failure (shock). Circulation 41:989–1001
Daga MK, Rohatgi I, Mishra R, Kumar N, Mawari G, Mishra TK, Singh S, Shukla J (2021) Lactate enhanced-quick Sequential Organ Failure Assessment 2 (LqSOFA2): a new score for bedside prognostication of patients with sepsis. Indian J Med Res 154:607–614
Sık G, Demirbuga A, Günhar S, Nisli K, Citak A (2019) Clinical features and indications associated with mortality in continuous renal replacement therapy for pediatric patients. Indian J Pediatr 86:360–364
Li X, Liu C, Mao Z, Li Q, Zhou F (2021) Timing of renal replacement therapy initiation for acute kidney injury in critically ill patients: a systematic review of randomized clinical trials with meta-analysis and trial sequential analysis. Crit Care 25:15
Daugirdas JT, Bernardo AA (2012) Hemodialysis effect on platelet count and function and hemodialysis-associated thrombocytopenia. Kidney Int 82:147–157
Gould DW, Doidge J, Sadique MZ, Borthwick M, Hatch R, Caskey FJ, Forni L, Lawrence RF, MacEwen C, Ostermann M, Mouncey PR, Harrison DA, Rowan KM, Young JD, Watkinson PJ (2022) Heparin versus citrate anticoagulation for continuous renal replacement therapy in intensive care: the RRAM observational study. Health Technol Assess 26:1–58
Author information
Authors and Affiliations
Contributions
Jinglin Xu and Dongmei Chen contributed to the study’s conception and design. Material preparation, data collection, and analysis were performed by Jinglin Xu, Lingyu Fang, and Lianqiang Wu. The first draft of the manuscript was written by Jinglin Xu, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
Considering this is an observational study, the Medical Ethics Committee of Quanzhou Maternity and Children’s Hospital has confirmed that no ethical approval is required.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, J., Fang, L., Chen, J. et al. Real-life effects, complications, and outcomes in 39 critically ill neonates receiving continuous kidney replacement therapy. Pediatr Nephrol 38, 3145–3152 (2023). https://doi.org/10.1007/s00467-023-05944-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-023-05944-8