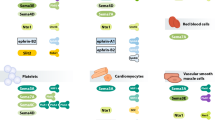
Abstract
Acute kidney injury is an increasingly common global health problem and is associated with severe morbidity and mortality. In addition to facing high mortality rates, the survivors of acute kidney injury are at increased risk of developing chronic kidney disease and end-stage renal disease. Renal ischemia-reperfusion injury (IRI) is the most common cause of acute kidney injury, and results from impaired delivery of oxygen and nutrients to the kidney. Massive leukocyte influx into the post-ischemic kidney is one of the hallmarks of IRI. The recruited leukocytes exacerbate tissue damage and, if uncontrolled, initiate the progressive changes that lead to renal fibrosis and chronic kidney disease. Early on, recruitment and activation of platelets promotes microthrombosis in the injured kidney, further exacerbating kidney damage. The diversity, complexity, and multiplicity of pathways involved in leukocyte recruitment and platelet activation make it extremely challenging to control these processes, and past efforts have met with limited success in human trials. A generalized strategy to inhibit infiltration of inflammatory leukocytes and platelets, thereby reducing inflammation and injury, may prove to be more beneficial. In this review, we summarize recent findings demonstrating that the neuronal guidance cues, Slit and Roundabout (Robo), prevent the migration of multiple leukocyte subsets towards diverse inflammatory chemoattractants, and have potent anti-platelet functions in vitro and in vivo. These properties uniquely position Slit2 as a novel therapeutic that could be used to prevent acute kidney injury associated with IRI.

Similar content being viewed by others
References
Coca SG, Singanamala S, Parikh CR (2012) Chronic kidney disease after acute kidney injury: a systematic review and meta-analysis. Kidney Int 81:442–448
Lameire NH, Bagga A, Cruz D, De Maeseneer J, Endre Z, Kellum JA, Liu KD, Mehta RL, Pannu N, Van Biesen W, Vanholder R (2013) Acute kidney injury: an increasing global concern. Lancet 382:170–179
Murugan R, Kellum JA (2011) Acute kidney injury: what’s the prognosis? Nat Rev Nephrol 7:209–217
Ricci Z, Cruz D, Ronco C (2008) The RIFLE criteria and mortality in acute kidney injury: a systematic review. Kidney Int 73:538–546
Amdur RL, Chawla LS, Amodeo S, Kimmel PL, Palant CE (2009) Outcomes following diagnosis of acute renal failure in U.S. veterans: focus on acute tubular necrosis. Kidney Int 76:1089–1097
Chawla LS, Kimmel PL (2012) Acute kidney injury and chronic kidney disease: an integrated clinical syndrome. Kidney Int 82:516–524
Lo LJ, Go AS, Chertow GM, McCulloch CE, Fan D, Ordoñez JD, Hsu C-Y (2009) Dialysis-requiring acute renal failure increases the risk of progressive chronic kidney disease. Kidney Int 76:893–899
Wald R, Quinn RR, Adhikari NK, Burns KE, Friedrich JO, Garg AX, Harel Z, Hladunewich MA, Luo J, Mamdani M, Perl J, Ray JG (2012) Risk of chronic dialysis and death following acute kidney injury. Am J Med 125:585–593
Bonventre JV, Yang L (2011) Cellular pathophysiology of ischemic acute kidney injury. J Clin Invest 121:4210–4221
Devarajan P (2006) Update on mechanisms of ischemic acute kidney injury. J Am Soc Nephrol 17:1503–1520
Jo SK, Rosner MH, Okusa MD (2007) Pharmacologic treatment of acute kidney injury: why drugs haven’t worked and what is on the horizon. Clin J Am Soc Nephrol 2:356–365
Remuzzi G, Horton R (2013) Acute renal failure: an unacceptable death sentence globally. Lancet 382:2041–2042
Bonventre JV, Weinberg JM (2003) Recent advances in the pathophysiology of ischemic acute renal failure. J Am Soc Nephrol 14:2199–2210
Okusa MD (2002) The inflammatory cascade in acute ischemic renal failure. Nephron 90:133–138
Legrand M, Mik EG, Johannes T, Payen D, Ince C (2008) Renal hypoxia and dysoxia after reperfusion of the ischemic kidney. Mol Med 14:502–516
Baggiolini M, Dewald B, Moser B (1994) Interleukin-8 and related chemotactic cytokines–CXC and CC chemokines. Adv Immunol 55:97–179
Daemen MA, van de Ven MW, Heineman E, Buurman WA (1999) Involvement of endogenous interleukin-10 and tumor necrosis factor-alpha in renal ischemia-reperfusion injury. Transplantation 67:792–800
Donnahoo KK, Meldrum DR, Shenkar R, Chung CS, Abraham E, Harken AH (2000) Early renal ischemia, with or without reperfusion, activates NFkappaB and increases TNF-alpha bioactivity in the kidney. J Urol 163:1328–1332
Donnahoo KK, Meng X, Ayala A, Cain MP, Harken AH, Meldrum DR (1999) Early kidney TNF-alpha expression mediates neutrophil infiltration and injury after renal ischemia-reperfusion. Am J Physiol 277:R922–R929
Furuichi K, Wada T, Iwata Y, Kitagawa K, Kobayashi K, Hashimoto H, Ishiwata Y, Asano M, Wang H, Matsushima K, Takeya M, Kuziel WA, Mukaida N, Yokoyama H (2003) CCR2 signaling contributes to ischemia-reperfusion injury in kidney. J Am Soc Nephrol 14:2503–2515
Furuichi K, Wada T, Yokoyama H, Kobayashi K (2002) Role of cytokines and chemokines in renal ischemia-reperfusion injury. Drug News Perspect 15:477–482
Miura M, Fu X, Zhang QW, Remick DG, Fairchild RL (2001) Neutralization of Gro alpha and macrophage inflammatory protein-2 attenuates renal ischemia/reperfusion injury. Am J Pathol 159:2137–2145
Ley K, Laudanna C, Cybulsky MI, Nourshargh S (2007) Getting to the site of inflammation: the leukocyte adhesion cascade updated. Nat Rev Immunol 7:678–689
Hellberg PO, Kallskog TO (1989) Neutrophil-mediated post-ischemic tubular leakage in the rat kidney. Kidney Int 36:555–561
Klausner JM, Paterson IS, Goldman G, Kobzik L, Rodzen C, Lawrence R, Valeri CR, Shepro D, Hechtman HB (1989) Postischemic renal injury is mediated by neutrophils and leukotrienes. Am J Physiol Ren Physiol 256:F794–F802
Linas SL, Shanley PF, Whittenburg D, Berger E, Repine JE (1988) Neutrophils accentuate ischemia-reperfusion injury in isolated perfused rat kidneys. Am J Physiol 255:F728–F735
Solez K, Morel-Maroger L, Sraer JD (1979) The morphology of “acute tubular necrosis” in man: analysis of 57 renal biopsies and a comparison with the glycerol model. Medicine (Baltimore) 58:362–376
Willinger CC, Schramek H, Pfaller K, Pfaller W (1992) Tissue distribution of neutrophils in postischemic acute renal failure. Virchows Archiv B Cell Pathol 62:237–243
Hayama T, Matsuyama M, Funao K, Tanaka T, Tsuchida K, Takemoto Y, Kawahito Y, Sano H, Nakatani T, Yoshimura R (2006) Beneficial effect of neutrophil elastase inhibitor on renal warm ischemia-reperfusion injury in the rat. Transplant Proc 38:2201–2202
Jang HR, Rabb H (2009) The innate immune response in ischemic acute kidney injury. Clin Immunol 130:41–50
Bolisetty S, Agarwal A (2009) Neutrophils in acute kidney injury: not neutral anymore. Kidney Int 75:674–676
Awad AS, Rouse M, Huang L, Vergis AL, Reutershan J, Cathro HP, Linden J, Okusa MD (2009) Compartmentalization of neutrophils in the kidney and lung following acute ischemic kidney injury. Kidney Int 75:689–698
Awad AS, Ye H, Huang L, Li L, Foss FW Jr, Macdonald TL, Lynch KR, Okusa MD (2006) Selective sphingosine 1-phosphate 1 receptor activation reduces ischemia-reperfusion injury in mouse kidney. Am J Physiol Ren Physiol 290:F1516–F1524
Chiao H, Kohda Y, McLeroy P, Craig L, Housini I, Star RA (1997) Alpha-melanocyte-stimulating hormone protects against renal injury after ischemia in mice and rats. J Clin Invest 99:1165–1172
Day YJ, Huang L, Ye H, Linden J, Okusa MD (2005) Renal ischemia-reperfusion injury and adenosine 2A receptor-mediated tissue protection: role of macrophages. Am J Physiol Ren Physiol 288:F722–F731
De Greef KE, Ysebaert DK, Dauwe S, Persy V, Vercauteren SR, Mey D, De Broe ME (2001) Anti-B7-1 blocks mononuclear cell adherence in vasa recta after ischemia. Kidney Int 60:1415–1427
Jo SK, Sung SA, Cho WY, Go KJ, Kim HK (2006) Macrophages contribute to the initiation of ischaemic acute renal failure in rats. Nephrol Dial Transplant 21:1231–1239
Loverre A, Capobianco C, Stallone G, Infante B, Schena A, Ditonno P, Palazzo S, Battaglia M, Crovace A, Castellano G, Ranieri E, Schena FP, Gesualdo L, Grandaliano G (2007) Ischemia–reperfusion injury-induced abnormal dendritic cell traffic in the transplanted kidney with delayed graft function. Kidney Int 72:994–1003
Rabb H, Daniels F, O’Donnell M, Haq M, Saba SR, Keane W, Tang WW (2000) Pathophysiological role of T lymphocytes in renal ischemia-reperfusion injury in mice. Am J Physiol Ren Physiol 279:F525–F531
Schlichting CL, Schareck WD, Weis M (2006) Renal ischemia-reperfusion injury: new implications of dendritic cell-endothelial cell interactions. Transplant Proc 38:670–673
Salmela K, Wramner L, Ekberg H, Hauser I, Bentdal O, Lins LE, Isoniemi H, Backman L, Persson N, Neumayer HH, Jorgensen PF, Spieker C, Hendry B, Nicholls A, Kirste G, Hasche G (1999) A randomized multicenter trial of the anti-ICAM-1 monoclonal antibody (enlimomab) for the prevention of acute rejection and delayed onset of graft function in cadaveric renal transplantation: a report of the European Anti-ICAM-1 Renal Transplant Study Group. Transplantation 67:729–736
Brose K, Bland KS, Wang KH, Arnott D, Henzel W, Goodman CS, Tessier-Lavigne M, Kidd T (1999) Slit proteins bind Robo receptors and have an evolutionarily conserved role in repulsive axon guidance. Cell 96:795–806
Kidd T, Bland KS, Goodman CS (1999) Slit is the midline repellent for the Robo receptor in Drosophila. Cell 96:785–794
Kidd T, Brose K, Mitchell KJ, Fetter RD, Tessier-Lavigne M, Goodman CS, Tear G (1998) Roundabout controls axon crossing of the CNS midline and defines a novel subfamily of evolutionarily conserved guidance receptors. Cell 92:205–215
Wu JY, Feng L, Park HT, Havlioglu N, Wen L, Tang H, Bacon KB, Jiang Z, Zhang X, Rao Y (2001) The neuronal repellent Slit inhibits leukocyte chemotaxis induced by chemotactic factors. Nature 410:948–952
Wong K, Park HT, Wu JY, Rao Y (2002) Slit proteins: molecular guidance cues for cells ranging from neurons to leukocytes. Curr Opin Genet Dev 12:583–591
Yuan W, Zhou L, Chen JH, Wu JY, Rao Y, Ornitz DM (1999) The mouse SLIT family: secreted ligands for ROBO expressed in patterns that suggest a role in morphogenesis and axon guidance. Dev Biol 212:290–306
Battye R, Stevens A, Perry RL, Jacobs JR (2001) Repellent signaling by Slit requires the leucine-rich repeats. J Neurosci 21:4290–4298
Chen JH, Wen L, Dupuis S, Wu JY, Rao Y (2001) The N-terminal leucine-rich regions in Slit are sufficient to repel olfactory bulb axons and subventricular zone neurons. J Neurosci 21:1548–1556
Dickson BJ, Gilestro GF (2006) Regulation of commissural axon pathfinding by slit and its Robo receptors. Annu Rev Cell Dev Biol 22:651–675
Tole S, Mukovozov IM, Huang YW, Magalhaes MA, Yan M, Crow MR, Liu GY, Sun CX, Durocher Y, Glogauer M, Robinson LA (2009) The axonal repellent, Slit2, inhibits directional migration of circulating neutrophils. J Leukoc Biol 86:1403–1415
Prasad A, Fernandis AZ, Rao Y, Ganju RK (2004) Slit protein-mediated inhibition of CXCR4-induced chemotactic and chemoinvasive signaling pathways in breast cancer cells. J Biol Chem 279:9115–9124
Acevedo LM, Weis SM, Cheresh DA (2008) Robo4 counteracts VEGF signaling. Nat Med 14:372–373
Park KW, Morrison CM, Sorensen LK, Jones CA, Rao Y, Chien CB, Wu JY, Urness LD, Li DY (2003) Robo4 is a vascular-specific receptor that inhibits endothelial migration. Dev Biol 261:251–267
Qian L, Liu J, Bodmer R (2005) Slit and Robo control cardiac cell polarity and morphogenesis. Curr Biol 15:2271–2278
MacMullin A, Jacobs JR (2006) Slit coordinates cardiac morphogenesis in Drosophila. Dev Biol 293:154–164
Xian J, Clark KJ, Fordham R, Pannell R, Rabbitts TH, Rabbitts PH (2001) Inadequate lung development and bronchial hyperplasia in mice with a targeted deletion in the Dutt1/Robo1 gene. Proc Natl Acad Sci U S A 98:15062–15066
Grieshammer U, Le M, Plump AS, Wang F, Tessier-Lavigne M, Martin GR (2004) SLIT2-mediated ROBO2 signaling restricts kidney induction to a single site. Dev Cell 6:709–717
Liu J, Zhang L, Wang D, Shen H, Jiang M, Mei P, Hayden PS, Sedor JR, Hu H (2003) Congenital diaphragmatic hernia, kidney agenesis and cardiac defects associated with Slit3-deficiency in mice. Mech Dev 120:1059–1070
Bertoli-Avella AM, Conte ML, Punzo F, de Graaf BM, Lama G, La Manna A, Polito C, Grassia C, Nobili B, Rambaldi PF, Oostra BA, Perrotta S (2008) ROBO2 gene variants are associated with familial vesicoureteral reflux. J Am Soc Nephrol 19:825–831
Wong K, Ren XR, Huang YZ, Xie Y, Liu G, Saito H, Tang H, Wen L, Brady-Kalnay SM, Mei L, Wu JY, Xiong WC, Rao Y (2001) Signal transduction in neuronal migration: roles of GTPase activating proteins and the small GTPase Cdc42 in the Slit-Robo pathway. Cell 107:209–221
Jones CA, Nishiya N, London NR, Zhu W, Sorensen LK, Chan AC, Lim CJ, Chen H, Zhang Q, Schultz PG, Hayallah AM, Thomas KR, Famulok M, Zhang K, Ginsberg MH, Li DY (2009) Slit2–Robo4 signalling promotes vascular stability by blocking Arf6 activity. Nat Cell Biol 11:1325–1331
Patel S, Huang YW, Reheman A, Pluthero FG, Chaturvedi S, Mukovozov IM, Tole S, Liu GY, Li L, Durocher Y, Ni H, Kahr WH, Robinson LA (2012) The cell motility modulator Slit2 is a potent inhibitor of platelet function. Circulation 126:1385–1395
Bashaw GJ, Kidd T, Murray D, Pawson T, Goodman CS (2000) Repulsive axon guidance: Abelson and enabled play opposing roles downstream of the roundabout receptor. Cell 101:703–715
Ypsilanti AR, Zagar Y, Chedotal A (2010) Moving away from the midline: new developments for Slit and Robo. Development 137:1939–1952
Guan H, Zu G, Xie Y, Tang H, Johnson M, Xu X, Kevil C, Xiong WC, Elmets C, Rao Y, Wu JY, Xu H (2003) Neuronal repellent Slit2 inhibits dendritic cell migration and the development of immune responses. J Immunol 171:6519–6526
Kanellis J, Garcia GE, Li P, Parra G, Wilson CB, Rao Y, Han S, Smith CW, Johnson RJ, Wu JY, Feng L (2004) Modulation of inflammation by slit protein in vivo in experimental crescentic glomerulonephritis. Am J Pathol 165:341–352
Prasad A, Qamri Z, Wu J, Ganju RK (2007) Slit-2/Robo-1 modulates the CXCL12/CXCR4-induced chemotaxis of T cells. J Leukoc Biol 82:465–476
Chaturvedi S, Yuen DA, Bajwa A, Huang YW, Sokollik C, Huang L, Lam GY, Tole S, Liu GY, Pan J, Chan L, Sokolskyy Y, Puthia M, Godaly G, John R, Wang C, Lee WL, Brumell JH, Okusa MD, Robinson LA (2013) Slit2 prevents neutrophil recruitment and renal ischemia-reperfusion injury. J Am Soc Nephrol 24:1274–1287
Bu-Qing Y, Zhen HG, Li M, Jian-Guo G (2010) Slit2 regulates attractive eosinophil and repulsive neutrophil chemotaxis through differential srGAP1 expression during lung inflammation. J Immunol 185:6294–6305
Jones CA, London NR, Chen H, Park KW, Sauvaget D, Stockton RA, Wythe JD, Suh W, Larrieu-Lahargue F, Mukouyama YS, Lindblom P, Seth P, Frias A, Nishiya N, Ginsberg MH, Gerhardt H, Zhang K, Li DY (2008) Robo4 stabilizes the vascular network by inhibiting pathologic angiogenesis and endothelial hyperpermeability. Nat Med 14:448–453
Zhao H, Appakkudal RA, Ganju RK (2014) Slit2–Robo4 pathway modulates lipopolysaccharide-induced endothelial inflammation and its expression is dysregulated during endotoxemia. J Immunol 192:385–393
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chaturvedi, S., Robinson, L.A. Slit2-Robo signaling in inflammation and kidney injury. Pediatr Nephrol 30, 561–566 (2015). https://doi.org/10.1007/s00467-014-2825-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-014-2825-4