Summary
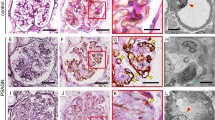
Polymorphonuclear neutrophil granulocytes (PMNs) seem to participate in the pathogenesis of renal ischemic reperfusion injury. The kidneys from male Sprague Dawley rats were immersion-fixed after 45 min of renal artery clamping followed by reperfusion for 0, 5, 20, and 120 min, respectively. The tissue distribution of PMNs in the kidneys was studied histochemically using naphthol AS-D chloroacetate esterase as a specific marker for these cells. Neutrophil counts per unit sectional area were obtained for renal cortex, outer and inner medulla. In the cortex separate intraglomerular and peritubular counts, and in the outer medulla separate outer and inner stripe counts were made. After 120 min of reperfusion the total renal PMN counts were 488 ±62 (n = 4) compared with 54 ±4 (n = 4) per cm2 in nonischemic controls. Within 120 min of reperfusion PMN counts increased by a factor of 8 in the cortex, of 12 in the outer medulla and of 14 in the inner medulla, compared with controls. The ratio of intraglomerular against peritubular PMN counts was approximately 2 in controls, but 0.5 after a 120-min reperfusion interval. The outer stripe of the outer medulla contained only a small number of PMNs whereas PMN counts of 923 ±197 (n = 4) per cm2 were found in the inner stripe after 120 min reperfusion. Interestingly, there was a marked increase in PMNs in the inner stripe during the first 5 min of reperfusion but no extravasation of PMNs was observed. Taken together, these data provide the first evidence that PMNs accumulate particularly within peritubular capillaries in the cortex and within the inner stripe of the outer medulla. This distribution pattern is consistent with the hypothesis that PMN-augmented cell injury occurs in the early phase of postischemic acute renal failure. In addition the steady increase in PMNs during reperfusion may further contribute to impaired renal function.
Similar content being viewed by others
References
AndreoliSP, McAteer JA, Mallett C (1990) Reactive oxygen mole-cule-mediated injury in endothelial and renal tubular epithelial cells in vitro. Kidney Int 38:785–794
BaggioliniM, Dewald B (1985) The Neutrophil. Int Archs Allergy Appl Immunol 76:[Suppl 1] 13–20
Bayati A, Christofferson R, Källskog Ö, Wolgast M (1990) Mecha-nism of erythrocyte trapping in ischemic acute renal failure. Acta Physiol Scand 138:13–23
Bolli R, Jeroudi MO, Patel BS, Aruoma OI, Halliwell B, Lai EK, McCay PB (1989) Marked reduction of free radical generation and contractile dysfunction by antioxidant therapy begun at the time of reperfusion. Evidence that myocardial “stunning” is a manifestation of reperfusion injury. Circ Res 65:607–622
Davis BJ, Ornstein L (1959) High resolution enzyme localization with a new diazo reagent, “hexazonium pararosaniline”. J Histochem Cytochem 7:297–298
DeMay JG, Vanhoutte PM (1982) Heterogeneous behaviour of the canine arterial and venous wall. Importance of endothelium. Circ Res 51:439–447
Engler RL, Schmid-Schönbein GW, Pavelec RS (1983) Leucocyte capillary plugging in myocardial ischemia and reperfusion in the dog. Am J Pathol 111:98–111
Gräser T, Vanhoutte PM (1991) Hypoxic contraction of canine coronary arteries: role of endothelium and cGMP. Am J Phys-iol 261:H1769-H1777
Hellberg POA, Källskog TÖK (1989) Neutrophil-mediated post-ischemic tubular leakage in the rat kidney. Kidney Int 36:555–561
Hellberg POA, Källskog ÖT, Öjteg G, Wolgast M (1990)a Peritu-bular capillary permeability and intravascular RBC aggregation after ischemia: effects of neutrophils. Am J Physiol 258: Fl018-F1025
Hellberg POA, Bayati A, Källskog Ö, Wolgast M (1990)b Red cell trapping after ischemia and long-term kidney damage. In-fluence of hematocrit. Kidney Int 37:1240–1247
Hellberg POA, Källskog Ö, Wolgast M (1990)c Nephron function in the early phase of ischemic renal failure. Significance of erythrocyte trapping. Kidney Int 38:432–439
Hernandez LA, Grisham MB, Twohig B, Arfors KE, Harlan JM, Granger DN (1987) Role of neutrophils in ischemia-reperfu-sion-induced microvascular injury. Am J Physiol 253:H699-H703
Jaeschke H, Farhood A, Smith CW (1990) Neutrophils contribute to ischemia/reperfusion injury in rat liver in vivo. FASEB J 4:3355–3359
Joannidis M, Bonn G, Pfaller W (1989) Lipid peroxidation -an initial event in experimental acute renal failure. Renal Physiol Biochem 12:47–55
Klausner JM, Paterson IS, Goldman G, Kobzik L, Rodzen C, Lawrence R, Valeri CR, Shepro D, Hechtman HB (1989) Post-ischemic renal injury is mediated by neutrophils and leuko-trienes. Am J Physiol 256:F794-F802
Korthius RJ, Grisham MB, Granger DN (1988) Leucocyte deple-tion attenuates vascular injury in postischemic skeletal muscle. Am J Physiol 254:H823-H827
Leder LD (1964) Über die selektive fermentcytochemische Darstel-lung von neutrophilen myeloischen Zellen und Gewebsmastzel-len im Paraffinschnitt. Klin Wochenschr 42:553
Linas SL, Shanley PF, Whittenburg D, Berger E, Repine JE (1988) Neutrophils accentuate ischemia-reperfusion injury in isolated perfused rat kidneys. Am J Physiol 255:F728-F735
Mason J, Torhorst J, Welsch J (1984) Role of the medullary perfu-sion defect in the pathogenesis of ischemic renal failure. Kidney Int 26:283–293
Mason J (1986) The pathophysiology of ischemic acute renal fail-ure. Renal Physiol Biochem 9:129–147
McEver RP, Beckstead JH, Moore KL, Marshall-Carlson L, Bain-ton DF (1989) GMP-140, a platelet alpha-granule membrane protein, is also synthesized by vascular endothelial cells and is localized in Weibel-Palade bodies. J Clin Invest 84:92–99
McEver RP (1991) GMP-140: a receptor for neutrophils and monocytes on activated platelets and endothelium. J Cell Bio-chem 45:156–161
Moloney WC, McPherson K, Fliegelman L (1960) Esterase activity in leucocytes demonstrated by the use of naphthol AS-D chloro-acetate substrate. J Histochem Cytochem 8:200–207
Pfaller W, Rittinger M (1980): Quantitative morphology of the rat kidney. Int J Biochem 12:17–22
Pfaller W (1982) Structure function correlation on rat kidney. Quantitative correlation of structure and function in the normal and injured rat kidney. Adv Anat Embryol Cell Biol 70:1–106
Rasmussen SN (1973) Intrarenal red cell and plasma volumes in the non-diuretic rat. Determination by means of51Cr-labelled red cells and125I-γM-immunoglobulin. Pflügers Arch 342:61–72
Rindler-Ludwig R, Schmalzl F, Braunsteiner H (1974) Esterases in human neutrophil granulocytes: Evidence for their protease nature. Br J Haematol 27:57–64
Romson JL, Hook BG, Kunkel SL, Abrams GD, Schork A, Lucce-si BR (1983) Reduction of the extent of ischemic myocardial injury by neutrophil depletion in the dog. Circulation 67:1016–1023
Schlondorff D, Neuwirth R (1986) Platelet activating factor and the kidney. Am J Physiol 251:F1-F11
Sussman MS, Bulkley GB (1990) Oxygen-derived free radicals in reperfusion injury. Meth Enzymol 186:711–723
Weiss SJ (1989) Tissue destruction by neutrophils. N Engl J Med 320:365–376
Yoshioka T, Ichikawa I (1989) Glomerular dysfunction induced by polymorphonuclear leucocyte-derived reactive oxygen spe-cies. Am J Physiol 257: F53-F59
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Willinger, C.C., Schramek, H., Pfaller, K. et al. Tissue distribution of Neutrophils in postischemic acute renal failure. Virchows Archiv B Cell Pathol 62, 237–243 (1992). https://doi.org/10.1007/BF02899687
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02899687