Abstract
Background
The sequential organ failure assessment (SOFA) score is easy to calculate and has been well validated as an outcome predictor in critically ill adult patients. However, its use in children has been limited, mainly because of differences in basal reference levels of serum creatinine.
Methods
Data include 87 patients requiring continuous renal replacement therapy (CRRT) between January 2005 and July 2011. We modified the SOFA score by excluding the renal component to an extrarenal SOFA score, based on the assumption that CRRT may mitigate the renal effect on outcome and investigated the utility in predicting outcome with comparison with pediatric risk of mortality (PRISM) III, pediatric logistic organ dysfunction (PELOD), and SOFA scores.
Results
Results showed that 95.4 % (n = 83) had multiple organ dysfunction syndrome with an overall mortality of 50.6 %. The extrarenal SOFA score at CRRT initiation and ≥20 % fluid overload were significantly associated with mortality. In comparison with the predictive power of various scoring systems, the extrarenal SOFA score showed the largest area under the receiver operating characteristic curve (extrarenal SOFA 0.774, SOFA 0.770, PRISM III 0.660, and PELOD 0.650).
Conclusions
The extrarenal SOFA score may be a useful prognostic marker in critically ill children treated with CRRT.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pediatric patients with multiple organ dysfunction syndrome (MODS) often require several types of medications, blood product transfusions and nutritional support, which makes controlling and maintaining an adequate volume status difficult. Hence, these patients often require continuous renal replacement therapy (CRRT). CRRT has advantages in that it allows continuous and programmed removal of fluid and metabolic correction that is well tolerated in hemodynamically unstable, critically ill patients [1, 2].
To evaluate the severity of MODS and to predict outcome, several scoring systems have been developed. Among them, the sequential organ failure assessment (SOFA) score is widely used to assess morbidity and has been well validated as an outcome predictor in various settings for critically ill adult patients [3–7]. SOFA provides a direct assessment of dysfunction in each of six organs using simple and easily measurable methods and without the need for complicated formulas or invasive procedures. In addition, it is specifically designed to account for several therapeutic interventions such as mechanical ventilation and the use of vasoactive drugs.
Despite its advantages, SOFA use in children has been limited mainly because of differences in the basal reference level of serum creatinine between children and adults. However, the impact of the renal component of the SOFA score on outcome prediction may be mitigated by CRRT management. Therefore, we modified the SOFA score by excluding the renal component to develop an extrarenal SOFA score for application in pediatric patients treated with CRRT.
The purpose of this study was to evaluate the usefulness of our extrarenal SOFA score in the prediction of outcome in critically ill pediatric patients requiring CRRT treatment.
Materials and methods
Participants
All consecutive patients admitted to our pediatric intensive care unit (PICU) and requiring CRRT management between January 2005 and July 2011 were included in this study. Patients who were in a vegetative state, had chronic renal failure, or who underwent CRRT for less than 24 h were excluded. This study was approved by the institutional review board of the Asan Medical Center, Seoul, Korea with waived parental consent owing to the retrospective nature of the analyses.
Data collection
The following baseline demographic data were gathered: age, weight, sex, underlying disease, diagnosis on admission to the PICU, leading causes of CRRT initiation, number of organs in failure, blood urea nitrogen (BUN), glomerular filtration rate (GFR), and levels of creatinine (Cr), C-reactive protein (CRP), and brain natriuretic peptide (BNP). The length of PICU and hospital stay, the interval from PICU admission to CRRT initiation, and the duration of CRRT support were also calculated. The percentage fluid overload (% FO) was calculated for each patient using the following formula: (total patient fluid input – total patient fluid output) (liters)/PICU admission body weight (kg) × 100 (%) [8]. To evaluate disease severity, the pediatric risk of mortality (PRISM) III, pediatric logistic organ dysfunction (PELOD), and SOFA scores were calculated on admission to the PICU and at CRRT initiation.
Use of the SOFA score in pediatric patients required several modifications owing to variations in normal ranges according to age, especially in the definition of hypotension and in the renal scoring system. We modified the criterion of hypotension of the cardiovascular component of SOFA to systolic blood pressure below the fifth percentile for age and we assigned 1 point to patients showing hypotension in the absence of any inotropic agent use (Table 1). The extrarenal SOFA score was defined as the standard SOFA score excluding the renal component, which was also calculated on admission to the PICU and at CRRT initiation. For these scorings, we recorded the worst or the most abnormal value over 24 h of PICU stay. The pre-sedation status was used to evaluate neurological status in patients under sedation. Organ failure was defined using previously reported criteria [9], and MODS was defined as the simultaneous presence of two or more organs in failure. The outcome variable was defined as PICU mortality.
Statistical analysis
Data were analyzed using the Statistical Package for Social Science (SPSS version 14.0 for windows, SPSS, Chicago, IL, USA) software. Categorical variables are expressed as number and proportion. Continuous data are presented as means ± standard deviations. Comparisons between two subgroups were made using the Student’s t test for continuous data and the Chi-squared test for categorical variables. Risk factors for mortality were evaluated in univariate and multivariate logistic regression analyses. Multivariate logistic regression analysis with backward elimination was conducted using variables yielding p values < 0.05 by univariate analysis to estimate the independent association of each covariate. Results are summarized as odds ratio (OR) and the respective 95 % confidence intervals (CI). The discriminative power of scores for predicting PICU mortality was evaluated by constructing receiver operating characteristic (ROC) curves and defined by the calculated area under the ROC curve (AUROC) with a 95 % confidence interval (95 % CI), with an optimal cutoff point chosen during analysis. For all comparisons, variables with a p value less than 0.05 were considered statistically significant.
Results
Demographic data
During the 78-month study period, 87 patients with a mean age of 7.9 ± 6.4 years were included. There were 39 boys and 48 girls. CRRT was carried out with a mean PICU stay of 6.45 ± 17.26 days, with 66 patients (75.8 %) given CRRT within 7 days of the PICU stay. Of the 87 patients, 73 (83.9 %) required mechanical ventilator support at CRRT initiation, and 60 (68.9 %) patients were managed with inotropic agents at CRRT initiation. The most common co-morbid disease was hemato-oncological disease in 43 cases (52.8 %). Sepsis-induced acute kidney injury (AKI) was the most common leading cause of CRRT (37.9 %; Table 2), with a significant difference in incidence between the survivor group (23.3 %) and non-survivor group (59.1 %; p = 0.001). In addition, 83 (95.4 %) patients presented with two or more organ dysfunctions and were therefore given a diagnosis of MODS.
Outcome data
The total ICU mortality rate was 50.6 %, which was significantly higher than the overall mortality of critically ill patients admitted to the PICU during the same period (12.0 %). Mortality among patients of less than 1 year of age was 61.1 %, which was higher than that of older children (47.8 %); however, this difference did not reach statistical significance. There was no significant difference between survivors and nonsurvivors with respect to age, baseline body weight, BUN, Cr, GFR, and length of PICU stay (Table 3). The % FO at CRRT initiation was higher in nonsurvivors (19.84 ± 24.61 %) than in survivors (13.10 ± 16.97 %), but this difference failed to reach statistical significance. When patients were stratified by fluid overload to ≥20 % and <20 %, survival rates were better for patients with <20 % FO (56.3 %) versus ≥20 % FO (31.8 %). In multivariate logistic regression analysis, the 20 % FO was significantly associated with mortality (OR 4.462, 95 % CI 1.260–15.805, p = 0.020, Table 4).
Comparisons of scoring systems
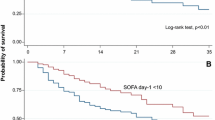
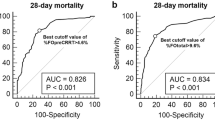
The PRISM III, PELOD, SOFA, and extrarenal SOFA scores on PICU admission did not show significant differences between survivors and nonsurvivors. However, all these scores at CRRT initiation showed significant differences between the two groups (Table 3). In terms of specific organ components of the SOFA score, higher individual organ failure scores except in the renal component had a tendency toward increased mortality. However, this was only statistically significant for respiratory, cardiovascular, and liver components (Fig. 1). Comparing the predictive capacity of the scores for PICU mortality, extrarenal SOFA score at CRRT initiation presented the largest AUROC curve (0.774, 95 % CI 0.676–0.872) followed by the SOFA score at CRRT initiation (0.770, 95 % CI 0.672–0.867), PRISM III at CRRT initiation (0.660, 95 % CI 0.545–0.775), and PELOD score at CRRT initiation (0.650, 95 % CI 0.535–0.766) (Fig. 2). Multivariate logistic regression analysis also showed that the extrarenal SOFA score at CRRT initiation (OR 1.382, 95 % CI 1.159–1.648, p < 0.001) was significantly correlated with PICU mortality (Table 4). A cutoff point of 10.5 was obtained for the extrarenal SOFA score at CRRT initiation and showed the maximum sum of sensitivity (72.7 %) and specificity (72.1 %) for predicting PICU mortality. When patients were categorized as having a low extrarenal SOFA score (<10.5) or a high extrarenal SOFA score (≥10.5) at CRRT initiation, the mortality rate was significantly higher in the high extrarenal SOFA score at CRRT initiation group (72.7 %) than in the low extrarenal SOFA score at CRRT initiation group (27.9 %;p < 0.001).
Discussion
Acute kidney injury (AKI) is a serious complication that commonly occurs in patients in the ICU [10, 11]. It is often accompanied by MODS [12–15], which can complicate the clinical course of AKI and is the major cause of high morbidity and mortality in the ICU. In our present study, 95.4 % of patients had MODS, and the overall mortality was 50.6 %. Among the patients included, 22 (25.6 %) showed ≥20 % FO, which was significantly associated with mortality by multivariate logistic regression analysis (OR 4.462, 95 % CI 1.260–15.805, p = 0.020). FO has been reported to be an important risk factor in patients with CRRT [13, 14, 16, 17]. Patients with MODS frequently required CRRT because it is an established safe renal support method and it can manage FO effectively.
It is important to objectively assess the severity of illness and organ dysfunction to predict outcome in critically ill patients and several organ dysfunction scoring systems have been developed, such as the MODS score, SOFA score, and PELOD score [18–20]. While the PELOD score is most commonly used for pediatric patients, it has several disadvantages: it is too complex to use repeatedly, and it does not take into consideration the use of vasoactive drugs, which are known to have an impact on patient prognosis [20]. In contrast, the SOFA score is easy to calculate and takes into account therapeutic interventions such as mechanical ventilation and vasoactive drugs, which makes it more appropriate to assess the severity of organ dysfunction after therapeutic management. Moreover, this score has been extensively evaluated and is well validated as an outcome predictor in critically ill adult patients. However, despite several advantages, the SOFA score has rarely been used for pediatric patients owing to the differences between adults and children in the normal range of variables [21]. Thus, when we applied this scoring system to pediatric patients, we modified the criterion for hypotension in the cardiovascular component.
Specifically, when we first broke down the SOFA score into individual organ scores, we found that mortality did not increase with increasing renal score, but did tend to increase with increasing score for the other organ components. One of the reasons is thought to be that the basal reference level of serum Cr is different from that of adults and may vary by age or size. However, even when corrected for age-appropriate normal values the renal component of the SOFA score showed weak power for predicting outcome. Furthermore, we found no difference between the survivor and nonsurvivor groups with regard to the levels of serum Cr (p = 0.575). These results are consistent with those of previous studies showing no relationship between serum Cr level and mortality in pediatric patients with AKI managed with CRRT [14, 17, 22]. On the basis of these findings, we assumed that CRRT management mitigates the effect of the renal component on the outcome prediction of the SOFA score. Thus, for patients who were managed with CRRT, we developed an extrarenal SOFA score by excluding the renal component from the SOFA score.
We compared the scoring systems PRISM III, PELOD, SOFA, and extra-renal SOFA scores, not only at PICU admission, but also at CRRT initiation. At PICU admission, none of the scores correlated significantly with mortality. However, at CRRT initiation, all four scores positively correlated with mortality. We interpret these results as follows: organ dysfunction is a dynamic process in which the severity of dysfunction changes with time. Furthermore, therapeutic interventions performed after ICU admission can have an impact on the clinical course of the disease and on the progression of organ dysfunction. From this perspective, the initial scores on PICU admission may simply reflect the pre-admission state of organ function. Thus, in order to capture the dynamically changing states of critically ill patients, it is preferential to use scoring systems such as the SOFA or extra-renal SOFA scores that allow repeated serial assessments with easy calculations.
Our present results show that the extra-renal SOFA score at CRRT initiation has a good predictive value for PICU mortality of patients treated with CRRT, as shown by the AUC (0.774, 95 % CI 0.676–0.872). The results of a multivariate logistic regression using mortality as the outcome variable also showed that the extrarenal SOFA score at CRRT initiation is a significant predictor of mortality (OR 1.382, 95 % CI 1.159–1.648, p < 0.001).
Several limitations should be addressed. First, our study was retrospective and observational in nature, and all the scores were also calculated retrospectively. Second, in sedated patients, we assumed the neurological component to be the pre-sedated state, which may cause some differences in representing the actual neurological status. Third, it was performed in a single medical center and the sample size was relatively small. Fourth, we included only a specific group of critically ill pediatric patients requiring CRRT treatment, which may raise the issue of the validity of our estimation and the use of the extrarenal SOFA score in general. Fifth, our extrarenal SOFA score is calculated by excluding the renal component from the SOFA score. This is based on the assumption that CRRT might mitigate the renal effect on outcome prediction. However, we did not thoroughly explore other modifications that could evaluate renal dysfunction better.
In conclusion, proper assessment of the severity of organ dysfunction is critical in the management of MODS and in the prediction of outcome for critically ill pediatric patients requiring CRRT. Our results suggest that the extrarenal SOFA score might be a feasible and reliable outcome predictor in critically ill pediatric patients requiring CRRT.
References
Goldstein SL (2003) Overview of pediatric renal replacement therapy in acute renal failure. Artif Organs 27:781–785
Symons JM, Chua AN, Somers MJ, Baum MA, Bunchman TE, Benfield MR, Brophy PD, Blowey D, Fortenberry JD, Chand D, Flores FX, Hackbarth R, Alexander SR, Mahan J, McBryde KD, Goldstein SL (2007) Demographic characteristics of pediatric continuous renal replacement therapy: a report of the prospective pediatric continuous renal replacement therapy registry. Clin J Am Soc Nephrol 2:732–738
Patila T, Kukkonen S, Vento A, Pettila V, Suojaranta-Ylinen R (2006) Relation of the sequential organ failure assessment score to morbidity and mortality after cardiac surgery. Ann Thorac Surg 82:2072–2078
Zygun D, Berthiaume L, Laupland K, Kortbeek J, Doig C (2006) SOFA is superior to MOD score for the determination of non-neurologic organ dysfunction in patients with severe traumatic brain injury: a cohort study. Crit Care 10:R115
Ceriani R, Mazzoni M, Bortone F, Gandini S, Solinas C, Susini G, Parodi O (2003) Application of the sequential organ failure assessment score to cardiac surgical patients. Chest 123:1229–1239
Ferreira FL, Bota DP, Bross A, Melot C, Vincent JL (2001) Serial evaluation of the SOFA score to predict outcome in critically ill patients. JAMA 286:1754–1758
Minne L, Abu-Hanna A, de Jonge E (2008) Evaluation of SOFA-based models for predicting mortality in the ICU: a systematic review. Crit Care 12:R161
Goldstein SL, Currier H, Graf C, Cosio CC, Brewer ED, Sachdeva R (2001) Outcome in children receiving continuous venovenous hemofiltration. Pediatrics 107:1309–1312
Tantalean JA, Leon RJ, Santos AA, Sanchez E (2003) Multiple organ dysfunction syndrome in children. Pediatr Crit Care Med 4:181–185
Strazdins V, Watson AR, Harvey B (2004) Renal replacement therapy for acute renal failure in children: European guidelines. Pediatr Nephrol 19:199–207
Yuasa S, Takahashi N, Shoji T, Uchida K, Kiyomoto H, Hashimoto M, Fujioka H, Fujita Y, Hitomi H, Matsuo H (1998) A simple and early prognostic index for acute renal failure patients requiring renal replacement therapy. Artif Organs 22:273–278
De Mendonca A, Vincent JL, Suter PM, Moreno R, Dearden NM, Antonelli M, Takala J, Sprung C, Cantraine F (2000) Acute renal failure in the ICU: risk factors and outcome evaluated by the SOFA score. Intensive Care Med 26:915–921
Foland JA, Fortenberry JD, Warshaw BL, Pettignano R, Merritt RK, Heard ML, Rogers K, Reid C, Tanner AJ, Easley KA (2004) Fluid overload before continuous hemofiltration and survival in critically ill children: a retrospective analysis. Crit Care Med 32:1771–1776
Goldstein SL, Somers MJ, Baum MA, Symons JM, Brophy PD, Blowey D, Bunchman TE, Baker C, Mottes T, McAfee N, Barnett J, Morrison G, Rogers K, Fortenberry JD (2005) Pediatric patients with multi-organ dysfunction syndrome receiving continuous renal replacement therapy. Kidney Int 67:653–658
Dunham CM (2001) Clinical impact of continuous renal replacement therapy on multiple organ failure. World J Surg 25:669–676
Sutherland SM, Zappitelli M, Alexander SR, Chua AN, Brophy PD, Bunchman TE, Hackbarth R, Somers MJ, Baum M, Symons JM, Flores FX, Benfield M, Askenazi D, Chand D, Fortenberry JD, Mahan JD, McBryde K, Blowey D, Goldstein SL (2010) Fluid overload and mortality in children receiving continuous renal replacement therapy: the prospective pediatric continuous renal replacement therapy registry. Am J Kidney Dis 55:316–325
Hayes LW, Oster RA, Tofil NM, Tolwani AJ (2009) Outcomes of critically ill children requiring continuous renal replacement therapy. J Crit Care 24:394–400
Graciano AL, Balko JA, Rahn DS, Ahmad N, Giroir BP (2005) The pediatric multiple organ dysfunction score (P-MODS): development and validation of an objective scale to measure the severity of multiple organ dysfunction in critically ill children. Crit Care Med 33:1484–1491
Vincent JL, Moreno R, Takala J, Willatts S, De Mendonca A, Bruining H, Reinhart CK, Suter PM, Thijs LG (1996) The SOFA (sepsis-related organ failure assessment) score to describe organ dysfunction/failure. On behalf of the working group on sepsis-related problems of the European society of intensive care medicine. Intensive Care Med 22:707–710
Leteurtre S, Duhamel A, Grandbastien B, Lacroix J, Leclerc F (2006) Paediatric logistic organ dysfunction (PELOD) score. Lancet 367:897, author reply 900-892
Shime N, Kageyama K, Ashida H, Tanaka Y (2001) Application of modified sequential organ failure assessment score in children after cardiac surgery. J Cardiothorac Vasc Anesth 15:463–468
Oh HJ, Shin DH, Lee MJ, Ko KI, Kim CH, Koo HM, Doh FM, Kwon YE, Kim YL, Nam KH, Park KS, An SY, Park JT, Han SH, Yoo TH, Kang SW (2013) Urine output is associated with prognosis in patients with acute kidney injury requiring continuous renal replacement therapy. J Crit Care 28:379–388
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jhang, W.K., Kim, Y.A., Ha, E.J. et al. Extrarenal sequential organ failure assessment score as an outcome predictor of critically ill children on continuous renal replacement therapy. Pediatr Nephrol 29, 1089–1095 (2014). https://doi.org/10.1007/s00467-013-2741-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-013-2741-z