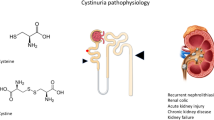
Abstract
Adenine phosphoribosyltransferase (APRT) deficiency, cystinuria, Dent disease, familial hypomagnesemia with hypercalciuria and nephrocalcinosis (FHHNC), and primary hyperoxaluria (PH) are rare but important causes of severe kidney stone disease and/or chronic kidney disease in children. Recurrent kidney stone disease and nephrocalcinosis, particularly in pre-pubertal children, should alert the physician to the possibility of an inborn error of metabolism as the underlying cause. Unfortunately, the lack of recognition and knowledge of the five disorders has frequently resulted in an unacceptable delay in diagnosis and treatment, sometimes with grave consequences. A high index of suspicion coupled with early diagnosis may reduce or even prevent the serious long-term complications of these diseases. In this paper, we review the epidemiology, clinical features, diagnosis, treatment, and outcome of patients with APRT deficiency, cystinuria, Dent disease, FHHNC, and PH, with an emphasis on childhood manifestations.







Similar content being viewed by others
References
Sas DJ (2011) An update on the changing epidemiology and metabolic risk factors in pediatric kidney stone disease. Clin J Am Soc Nephrol 6:2062–2068
Stamatelou KK, Francis ME, Jones CA, Nyberg LM, Curhan GC (2003) Time trends in reported prevalence of kidney stones in the United States: 1976–1994. Kidney Int 63:1817–1823
Beara-Lasic L, Edvardsson V, Palsson R, Lieske J, Goldfarb D, Milliner D (2011) Genetic causes of kidney stones and kidney failure. Clin Rev Bone Miner Metab 10(6):1–18
Greenwood MC, Dillon MJ, Simmonds HA, Barratt TM, Pincott JR, Metreweli C (1982) Renal failure due to 2,8-dihydroxyadenine urolithiasis. Eur J Pediatr 138:346–349
Konrad M, Hou J, Weber S, Dotsch J, Kari JA, Seeman T, Kuwertz-Broking E, Peco-Antic A, Tasic V, Dittrich K, Alshaya HO, von Vigier RO, Gallati S, Goodenough DA, Schaller A (2008) CLDN16 genotype predicts renal decline in familial hypomagnesemia with hypercalciuria and nephrocalcinosis. J Am Soc Nephrol 19:171–181
Hoppe B, Beck BB, Milliner DS (2009) The primary hyperoxalurias. Kidney Int 75:1264–1271
Rule AD, Krambeck AE, Lieske JC (2011) Chronic kidney disease in kidney stone formers. Clin J Am Soc Nephrol 6:2069–2075
Edvardsson V, Palsson R, Olafsson I, Hjaltadottir G, Laxdal T (2001) Clinical features and genotype of adenine phosphoribosyltransferase deficiency in Iceland. Am J Kidney Dis 38:473–480
Bollee G, Dollinger C, Boutaud L, Guillemot D, Bensman A, Harambat J, Deteix P, Daudon M, Knebelmann B, Ceballos-Picot I (2010) Phenotype and genotype characterization of adenine phosphoribosyltransferase deficiency. J Am Soc Nephrol 21:679–688
Kelley WN, Levy RI, Rosenbloom FM, Henderson JF, Seegmiller JE (1968) Adenine phosphoribosyltransferase deficiency: a previously undescribed genetic defect in man. J Clin Invest 47:2281–2289
Sahota AS, Tischfield AJ, Kamatani N, Simmonds HA (2001) Adenine phosphoribosyltransferase deficiency and 2,8-dihydroxyadenine lithiasis. In: Scriver CRBA, Sly WS, Valle D, Vogelstein B, Childs B (eds) The metabolic and molecular bases of inherited disease. McGraw-Hill, New York, pp 2571–2584
Broderick TP, Schaff DA, Bertino AM, Dush MK, Tischfield JA, Stambrook PJ (1987) Comparative anatomy of the human APRT gene and enzyme: nucleotide sequence divergence and conservation of a nonrandom CpG dinucleotide arrangement. Proc Natl Acad Sci U S A 84:3349–3353
Edvardsson VO, Palsson R, Sahota A (2012) Adenine Phosphoribosyltransferase Deficiency In: GeneReviews at GeneTests: Medical Genetics Information Resource [database online]. Copyright, University of Washington, Seattle, 1997–2010. Available at http://www.genetests.org
Kamatani N, Hakoda M, Otsuka S, Yoshikawa H, Kashiwazaki S (1992) Only three mutations account for almost all defective alleles causing adenine phosphoribosyltransferase deficiency in Japanese patients. J Clin Invest 90:130–135
Nasr SH, Sethi S, Cornell LD, Milliner DS, Boelkins M, Broviac J, Fidler ME (2010) Crystalline nephropathy due to 2,8-dihydroxyadeninuria: an under-recognized cause of irreversible renal failure. Nephrol Dial Transplant 25:1909–1915
Hidaka Y, Tarle SA, O’Toole TE, Kelley WN, Palella TD (1987) Nucleotide sequence of the human APRT gene. Nucleic Acids Res 15:9086
Sahota A, Chen J, Boyadjiev SA, Gault MH, Tischfield JA (1994) Missense mutation in the adenine phosphoribosyltransferase gene causing 2,8-dihydroxyadenine urolithiasis. Hum Mol Genet 3:817–818
Harambat J, Bollee G, Daudon M, Ceballos-Picot I, Bensman A (2012) Adenine phosphoribosyltransferase deficiency in children. Pediatr Nephrol 27:571–579
Debray H, Cartier P, Temstet A, Cendron J (1976) Child’s urinary lithiasis revealing a complete deficit in adenine phosphoribosyl transferase. Pediatr Res 10:762–766
Chiba P, Zwiauer K, Muller MM (1988) Characterization of an adenine phosphoribosyltransferase deficiency. Clin Chim Acta 172:141–147
Cassidy MJ, McCulloch T, Fairbanks LD, Simmonds HA (2004) Diagnosis of adenine phosphoribosyltransferase deficiency as the underlying cause of renal failure in a renal transplant recipient. Nephrol Dial Transplant 19:736–738
Benedetto B, Madden R, Kurbanov A, Braden G, Freeman J, Lipkowitz GS (2001) Adenine phosphoribosyltransferase deficiency and renal allograft dysfunction. Am J Kidney Dis 37:E37
Becker MA, Schumacher HR Jr, Wortmann RL, MacDonald PA, Palo WA, Eustace D, Vernillet L, Joseph-Ridge N (2005) Febuxostat, a novel nonpurine selective inhibitor of xanthine oxidase: a twenty-eight-day, multicenter, phase II, randomized, double-blind, placebo-controlled, dose–response clinical trial examining safety and efficacy in patients with gout. Arthritis Rheum 52:916–923
Milliner DS, Murphy ME (1993) Urolithiasis in pediatric patients. Mayo Clin Proc 68:241–248
Chillaron J, Font-Llitjos M, Fort J, Zorzano A, Goldfarb DS, Nunes V, Palacin M (2010) Pathophysiology and treatment of cystinuria. Nat Rev Nephrol 6:424–434
Fernandez E, Carrascal M, Rousaud F, Abian J, Zorzano A, Palacin M, Chillaron J (2002) rBAT-b(0,+)AT heterodimer is the main apical reabsorption system for cystine in the kidney. Am J Physiol Renal Physiol 283:F540–548
Martens K, Jaeken J, Matthijs G, Creemers JW (2008) Multi-system disorder syndromes associated with cystinuria type I. Curr Mol Med 8:544–550
Dello Strologo L, Laurenzi C, Legato A, Pastore A (2007) Cystinuria in children and young adults: success of monitoring free-cystine urine levels. Pediatr Nephrol 22:1869–1873
Lambert EH, Asplin JR, Herrell SD, Miller NL (2010) Analysis of 24-hour urine parameters as it relates to age of onset of cystine stone formation. J Endourol 24:1179–1182
Worcester EM, Coe FL, Evan AP, Parks JH (2006) Reduced renal function and benefits of treatment in cystinuria vs other forms of nephrolithiasis. BJU Int 97:1285–1290
Assimos DG, Leslie SW, Ng C, Streem SB, Hart LJ (2002) The impact of cystinuria on renal function. J Urol 168:27–30
Nakagawa Y, Coe FL (1999) A modified cyanide-nitroprusside method for quantifying urinary cystine concentration that corrects for creatinine interference. Clin Chim Acta 289:57–68
Evan AP, Coe FL, Lingeman JE, Shao Y, Matlaga BR, Kim SC, Bledsoe SB, Sommer AJ, Grynpas M, Phillips CL, Worcester EM (2006) Renal crystal deposits and histopathology in patients with cystine stones. Kidney Int 69:2227–2235
Boutros M, Vicanek C, Rozen R, Goodyer P (2005) Transient neonatal cystinuria. Kidney Int 67:443–448
Goldfarb DS, Coe FL, Asplin JR (2006) Urinary cystine excretion and capacity in patients with cystinuria. Kidney Int 69:1041–1047
Rodman JS, Blackburn P, Williams JJ, Brown A, Pospischil MA, Peterson CM (1984) The effect of dietary protein on cystine excretion in patients with cystinuria. Clin Nephrol 22:273–278
Chow GK, Streem SB (1996) Medical treatment of cystinuria: results of contemporary clinical practice. J Urol 156:1576–1578
Nakagawa Y, Asplin JR, Goldfarb DS, Parks JH, Coe FL (2000) Clinical use of cystine supersaturation measurements. J Urol 164:1481–1485
Dolin DJ, Asplin JR, Flagel L, Grasso M, Goldfarb DS (2005) Effect of cystine-binding thiol drugs on urinary cystine capacity in patients with cystinuria. J Endourol 19:429–432
Pareek G, Steele TH, Nakada SY (2005) Urological intervention in patients with cystinuria is decreased with medical compliance. J Urol 174:2250–2252
Trinchieri A, Montanari E, Zanetti G, Lizzano R (2007) The impact of new technology in the treatment of cystine stones. Urol Res 35:129–132
Wrong OM, Norden AG, Feest TG (1994) Dent’s disease; a familial proximal renal tubular syndrome with low-molecular-weight proteinuria, hypercalciuria, nephrocalcinosis, metabolic bone disease, progressive renal failure and a marked male predominance. Q J Med 87:473–493
Lloyd SE, Gunther W, Pearce SH, Thomson A, Bianchi ML, Bosio M, Craig IW, Fisher SE, Scheinman SJ, Wrong O, Jentsch TJ, Thakker RV (1997) Characterisation of renal chloride channel, CLCN5, mutations in hypercalciuric nephrolithiasis (kidney stones) disorders. Hum Mol Genet 6:1233–1239
Thakker RV (2000) Pathogenesis of Dent’s disease and related syndromes of X-linked nephrolithiasis. Kidney Int 57:787–793
Reinhart SC, Norden AG, Lapsley M, Thakker RV, Pang J, Moses AM, Frymoyer PA, Favus MJ, Hoepner JA, Scheinman SJ (1995) Characterization of carrier females and affected males with X-linked recessive nephrolithiasis. J Am Soc Nephrol 5:1451–1461
Waldegger S, Jentsch TJ (2000) From tonus to tonicity: physiology of CLC chloride channels. J Am Soc Nephrol 11:1331–1339
Dutzler R (2004) Structural basis for ion conduction and gating in ClC chloride channels. FEBS Lett 564:229–233
Gunther W, Luchow A, Cluzeaud F, Vandewalle A, Jentsch TJ (1998) ClC-5, the chloride channel mutated in Dent’s disease, colocalizes with the proton pump in endocytotically active kidney cells. Proc Natl Acad Sci U S A 95:8075–8080
Devuyst O, Christie PT, Courtoy PJ, Beauwens R, Thakker RV (1999) Intra-renal and subcellular distribution of the human chloride channel, CLC-5, reveals a pathophysiological basis for Dent’s disease. Hum Mol Genet 8:247–257
Hara-Chikuma M, Wang Y, Guggino SE, Guggino WB, Verkman AS (2005) Impaired acidification in early endosomes of ClC-5 deficient proximal tubule. Biochem Biophys Res Commun 329:941–946
Picollo A, Pusch M (2005) Chloride/proton antiporter activity of mammalian CLC proteins ClC-4 and ClC-5. Nature 436:420–423
Scheel O, Zdebik AA, Lourdel S, Jentsch TJ (2005) Voltage-dependent electrogenic chloride/proton exchange by endosomal CLC proteins. Nature 436:424–427
Novarino G, Weinert S, Rickheit G, Jentsch TJ (2010) Endosomal chloride-proton exchange rather than chloride conductance is crucial for renal endocytosis. Science 328:1398–1401
Friedrich T, Breiderhoff T, Jentsch TJ (1999) Mutational analysis demonstrates that ClC-4 and ClC-5 directly mediate plasma membrane currents. J Biol Chem 274:896–902
Hryciw DH, Ekberg J, Pollock CA, Poronnik P (2006) ClC-5: a chloride channel with multiple roles in renal tubular albumin uptake. Int J Biochem Cell Biol 38:1036–1042
Piwon N, Gunther W, Schwake M, Bosl MR, Jentsch TJ (2000) ClC-5 Cl--channel disruption impairs endocytosis in a mouse model for Dent’s disease. Nature 408:369–373
Wang SS, Devuyst O, Courtoy PJ, Wang XT, Wang H, Wang Y, Thakker RV, Guggino S, Guggino WB (2000) Mice lacking renal chloride channel, CLC-5, are a model for Dent’s disease, a nephrolithiasis disorder associated with defective receptor-mediated endocytosis. Hum Mol Genet 9:2937–2945
Christensen EI, Devuyst O, Dom G, Nielsen R, Van der Smissen P, Verroust P, Leruth M, Guggino WB, Courtoy PJ (2003) Loss of chloride channel ClC-5 impairs endocytosis by defective trafficking of megalin and cubilin in kidney proximal tubules. Proc Natl Acad Sci U S A 100:8472–8477
Silva IV, Cebotaru V, Wang H, Wang XT, Wang SS, Guo G, Devuyst O, Thakker RV, Guggino WB, Guggino SE (2003) The ClC-5 knockout mouse model of Dent’s disease has renal hypercalciuria and increased bone turnover. J Bone Miner Res 18:615–623
Leheste JR, Melsen F, Wellner M, Jansen P, Schlichting U, Renner-Muller I, Andreassen TT, Wolf E, Bachmann S, Nykjaer A, Willnow TE (2003) Hypocalcemia and osteopathy in mice with kidney-specific megalin gene defect. FASEB J 17:247–249
Devuyst O, Thakker RV (2010) Dent’s disease. Orphanet J Rare Dis 5:28
Claverie-Martin F, Ramos-Trujillo E, Garcia-Nieto V (2011) Dent’s disease: clinical features and molecular basis. Pediatr Nephrol 26:693–704
Hoopes RR Jr, Raja KM, Koich A, Hueber P, Reid R, Knohl SJ, Scheinman SJ (2004) Evidence for genetic heterogeneity in Dent’s disease. Kidney Int 65:1615–1620
Hoopes RR Jr, Shrimpton AE, Knohl SJ, Hueber P, Hoppe B, Matyus J, Simckes A, Tasic V, Toenshoff B, Suchy SF, Nussbaum RL, Scheinman SJ (2005) Dent Disease with mutations in OCRL1. Am J Hum Genet 76:260–267
Ghanekar Y, Lowe M (2005) Signalling for secretion. Nat Cell Biol 7:851–853
Choudhury R, Diao A, Zhang F, Eisenberg E, Saint-Pol A, Williams C, Konstantakopoulos A, Lucocq J, Johannes L, Rabouille C, Greene LE, Lowe M (2005) Lowe syndrome protein OCRL1 interacts with clathrin and regulates protein trafficking between endosomes and the trans-Golgi network. Mol Biol Cell 16:3467–3479
Tosetto E, Addis M, Caridi G, Meloni C, Emma F, Vergine G, Stringini G, Papalia T, Barbano G, Ghiggeri GM, Ruggeri L, Miglietti N, D Angelo A, Melis MA, Anglani F (2009) Locus heterogeneity of Dent’s disease: OCRL1 and TMEM27 genes in patients with no CLCN5 mutations. Pediatr Nephrol 24:1967–1973
Hichri H, Rendu J, Monnier N, Coutton C, Dorseuil O, Poussou RV, Baujat G, Blanchard A, Nobili F, Ranchin B, Remesy M, Salomon R, Satre V, Lunardi J (2011) From Lowe syndrome to Dent disease: correlations between mutations of the OCRL1 gene and clinical and biochemical phenotypes. Hum Mutat 32:379–388
Copelovitch L, Nash MA, Kaplan BS (2007) Hypothesis: Dent disease is an underrecognized cause of focal glomerulosclerosis. Clin J Am Soc Nephrol 2:914–918
Frishberg Y, Dinour D, Belostotsky R, Becker-Cohen R, Rinat C, Feinstein S, Navon-Elkan P, Ben-Shalom E (2009) Dent’s disease manifesting as focal glomerulosclerosis: is it the tip of the iceberg? Pediatr Nephrol 24:2369–2373
Bokenkamp A, Bockenhauer D, Cheong HI, Hoppe B, Tasic V, Unwin R, Ludwig M (2009) Dent-2 disease: a mild variant of Lowe syndrome. J Pediatr 155:94–99
Davey PG, Gosling P (1982) Beta 2-microglobulin instability in pathological urine. Clin Chem 28:1330–1333
Suzuki S, Suzuki J, Kume K, Yoshida K, Suyama H, Kawasaki Y, Nozawa R, Suzuki H, Fujiki T, Kamiyama S, Suzuki A (1999) Poor renal accumulation of 99mTc-DMSA in idiopathic tubular proteinuria. Nephron 81:49–54
Cebotaru V, Kaul S, Devuyst O, Cai H, Racusen L, Guggino WB, Guggino SE (2005) High citrate diet delays progression of renal insufficiency in the ClC-5 knockout mouse model of Dent’s disease. Kidney Int 68:642–652
Michelis MF, Drash AL, Linarelli LG, De Rubertis FR, Davis BB (1972) Decreased bicarbonate threshold and renal magnesium wasting in a sibship with distal renal tubular acidosis. (Evaluation of the pathophysiological role of parathyroid hormone). Metabolism 21:905–920
Weber S, Schneider L, Peters M, Misselwitz J, Ronnefarth G, Boswald M, Bonzel KE, Seeman T, Sulakova T, Kuwertz-Broking E, Gregoric A, Palcoux JB, Tasic V, Manz F, Scharer K, Seyberth HW, Konrad M (2001) Novel paracellin-1 mutations in 25 families with familial hypomagnesemia with hypercalciuria and nephrocalcinosis. J Am Soc Nephrol 12:1872–1881
Faguer S, Chauveau D, Cintas P, Tack I, Cointault O, Rostaing L, Vargas-Poussou R, Ribes D (2011) Renal, ocular, and neuromuscular involvements in patients with CLDN19 mutations. Clin J Am Soc Nephrol 6:355–360
Godron A, Harambat J, Boccio V, Mensire A, May A, Rigothier C, Couzi L, Barrou B, Godin M, Chauveau D, Faguer S, Vallet M, Cochat P, Eckart P, Guest G, Guigonis V, Houillier P, Blanchard A, Jeunemaitre X, Vargas-Poussou R (2012) Familial hypomagnesemia with hypercalciuria and nephrocalcinosis: phenotype-genotype correlation and outcome in 32 patients with CLDN16 or CLDN19 mutations. Clin J Am Soc Nephrol 7:801–809
Rodriguez-Soriano J, Vallo A, Garcia-Fuentes M (1987) Hypomagnesaemia of hereditary renal origin. Pediatr Nephrol 1:465–472
Simon DB, Lu Y, Choate KA, Velazquez H, Al-Sabban E, Praga M, Casari G, Bettinelli A, Colussi G, Rodriguez-Soriano J, McCredie D, Milford D, Sanjad S, Lifton RP (1999) Paracellin-1, a renal tight junction protein required for paracellular Mg2+ resorption. Science 285:103–106
Konrad M, Schaller A, Seelow D, Pandey AV, Waldegger S, Lesslauer A, Vitzthum H, Suzuki Y, Luk JM, Becker C, Schlingmann KP, Schmid M, Rodriguez-Soriano J, Ariceta G, Cano F, Enriquez R, Juppner H, Bakkaloglu SA, Hediger MA, Gallati S, Neuhauss SC, Nurnberg P, Weber S (2006) Mutations in the tight-junction gene claudin 19 (CLDN19) are associated with renal magnesium wasting, renal failure, and severe ocular involvement. Am J Hum Genet 79:949–957
Li J, Ananthapanyasut W, Yu AS (2011) Claudins in renal physiology and disease. Pediatr Nephrol 26:2133–2142
Blanchard A, Jeunemaitre X, Coudol P, Dechaux M, Froissart M, May A, Demontis R, Fournier A, Paillard M, Houillier P (2001) Paracellin-1 is critical for magnesium and calcium reabsorption in the human thick ascending limb of Henle. Kidney Int 59:2206–2215
Haisch L, Almeida JR, Abreu da Silva PR, Schlingmann KP, Konrad M (2011) The role of tight junctions in paracellular ion transport in the renal tubule: lessons learned from a rare inherited tubular disorder. Am J Kidney Dis 57:320–330
Kiuchi-Saishin Y, Gotoh S, Furuse M, Takasuga A, Tano Y, Tsukita S (2002) Differential expression patterns of claudins, tight junction membrane proteins, in mouse nephron segments. J Am Soc Nephrol 13:875–886
Hou J, Paul DL, Goodenough DA (2005) Paracellin-1 and the modulation of ion selectivity of tight junctions. J Cell Sci 118:5109–5118
Hou J, Renigunta A, Konrad M, Gomes AS, Schneeberger EE, Paul DL, Waldegger S, Goodenough DA (2008) Claudin-16 and claudin-19 interact and form a cation-selective tight junction complex. J Clin Invest 118:619–628
Hou J, Renigunta A, Gomes AS, Hou M, Paul DL, Waldegger S, Goodenough DA (2009) Claudin-16 and claudin-19 interaction is required for their assembly into tight junctions and for renal reabsorption of magnesium. Proc Natl Acad Sci U S A 106:15350–15355
Hou J, Shan Q, Wang T, Gomes AS, Yan Q, Paul DL, Bleich M, Goodenough DA (2007) Transgenic RNAi depletion of claudin-16 and the renal handling of magnesium. J Biol Chem 282:17114–17122
Kausalya PJ, Amasheh S, Gunzel D, Wurps H, Muller D, Fromm M, Hunziker W (2006) Disease-associated mutations affect intracellular traffic and paracellular Mg2+ transport function of Claudin-16. J Clin Invest 116:878–891
Hampson G, Konrad MA, Scoble J (2008) Familial hypomagnesaemia with hypercalciuria and nephrocalcinosis (FHHNC): compound heterozygous mutation in the claudin 16 (CLDN16) gene. BMC Nephrol 9:12
Stechman MJ, Loh NY, Thakker RV (2009) Genetic causes of hypercalciuric nephrolithiasis. Pediatr Nephrol 24:2321–2332
Praga M, Vara J, Gonzalez-Parra E, Andres A, Alamo C, Araque A, Ortiz A, Rodicio JL (1995) Familial hypomagnesemia with hypercalciuria and nephrocalcinosis. Kidney Int 47:1419–1425
Zimmermann B, Plank C, Konrad M, Stohr W, Gravou-Apostolatou C, Rascher W, Dotsch J (2006) Hydrochlorothiazide in CLDN16 mutation. Nephrol Dial Transplant 21:2127–2132
Lieske JC, Spargo BH, Toback FG (1992) Endocytosis of calcium oxalate crystals and proliferation of renal tubular epithelial cells in a patient with type 1 primary hyperoxaluria. J Urol 148:1517–1519
Muda AO, Barsotti P, Rinaldi S, Rizzoni G, Faraggiana T (1997) Renal pathology in hyperoxaluria. In: Sessa A, Conte F, Meroni M, Battini G (eds) Hereditary kidney diseases. Karger, Basel, pp 160–166
van Woerden CS, Groothoff JW, Wanders RJ, Davin JC, Wijburg FA (2003) Primary hyperoxaluria type 1 in The Netherlands: prevalence and outcome. Nephrol Dial Transplant 18:273–279
Cochat P, Liutkus A, Fargue S, Basmaison O, Ranchin B, Rolland MO (2006) Primary hyperoxaluria type 1: still challenging! Pediatr Nephrol 21:1075–1081
Coulter-Mackie MB (2005) Preliminary evidence for ethnic differences in primary hyperoxaluria type 1 genotype. Am J Nephrol 25:264–268
Danpure CJ (2005) Molecular etiology of primary hyperoxaluria type 1: new directions for treatment. Am J Nephrol 25:303–310
Milliner DS (2005) The primary hyperoxalurias: an algorithm for diagnosis. Am J Nephrol 25:154–160
Belostotsky R, Seboun E, Idelson GH, Milliner DS, Becker-Cohen R, Rinat C, Monico CG, Feinstein S, Ben-Shalom E, Magen D, Weissman I, Charon C, Frishberg Y (2010) Mutations in DHDPSL are responsible for primary hyperoxaluria type III. Am J Hum Genet 87:392–399
Monico CG, Rossetti S, Belostotsky R, Cogal AG, Herges RM, Seide BM, Olson JB, Bergstrahl EJ, Williams HJ, Haley WE, Frishberg Y, Milliner DS (2011) Primary hyperoxaluria type III gene HOGA1 (formerly DHDPSL) as a possible risk factor for idiopathic calcium oxalate urolithiasis. Clin J Am Soc Nephrol 6:2289–2295
Riedel TJ, Johnson LC, Knight J, Hantgan RR, Holmes RP, Lowther WT (2011) Structural and biochemical studies of human 4-hydroxy-2-oxoglutarate aldolase: implications for hydroxyproline metabolism in primary hyperoxaluria. PLoS One 6:e26021
Monico CG, Rossetti S, Olson JB, Milliner DS (2005) Pyridoxine effect in type I primary hyperoxaluria is associated with the most common mutant allele. Kidney Int 67:1704–1709
Monico CG, Rossetti S, Schwanz HA, Olson JB, Lundquist PA, Dawson DB, Harris PC, Milliner DS (2007) Comprehensive mutation screening in 55 probands with type 1 primary hyperoxaluria shows feasibility of a gene-based diagnosis. J Amer Soc Nephrol 18:1905–1914
Lieske JC, Monico CG, Holmes WS, Bergstralh EJ, Slezak JM, Rohlinger AL, Olson JB, Milliner DS (2005) International registry for primary hyperoxaluria. Am J Nephrol 25:290–296
Harambat J, Fargue S, Acquaviva C, Gagnadoux MF, Janssen F, Liutkus A, Mourani C, Macher MA, Abramowicz D, Legendre C, Durrbach A, Tsimaratos M, Nivet H, Girardin E, Schott AM, Rolland MO, Cochat P (2010) Genotype-phenotype correlation in primary hyperoxaluria type 1: the p.Gly170Arg AGXT mutation is associated with a better outcome. Kidney Int 77:443–449
Frishberg Y, Rinat C, Shalata A, Khatib I, Feinstein S, Becker-Cohen R, Weismann I, Wanders RJ, Rumsby G, Roels F, Mandel H (2005) Intra-familial clinical heterogeneity: absence of genotype-phenotype correlation in primary hyperoxaluria type 1 in Israel. Am J Nephrol 25:269–275
Hoppe B, Langman CB (2003) A United States survey on diagnosis, treatment, and outcome of primary hyperoxaluria. Pediatr Nephrol 18:986–991
Milliner D (2006) Treatment of the primary hyperoxalurias: a new chapter. Kidney Int 70:1198–1200
Leumann E, Hoppe B, Neuhaus T (1993) Management of primary hyperoxaluria: efficacy of oral citrate administration. Pediatr Nephrol 7:207–211
Hoppe B, Groothoff JW, Hulton SA, Cochat P, Niaudet P, Kemper MJ, Deschenes G, Unwin R, Milliner D (2011) Efficacy and safety of Oxalobacter formigenes to reduce urinary oxalate in primary hyperoxaluria. Nephrol Dial Transplant 26:3609–3615
Monico CG, Olson JB, Milliner DS (2005) Implications of genotype and enzyme phenotype in pyridoxine response of patients with type I primary hyperoxaluria. Am J Nephrol 25:183–188
Illies F, Bonzel KE, Wingen AM, Latta K, Hoyer PF (2006) Clearance and removal of oxalate in children on intensified dialysis for primary hyperoxaluria type 1. Kidney Int 70:1642–1648
Bergstralh EJ, Monico CG, Lieske JC, Herges RM, Langman CB, Hoppe B, Milliner DS (2010) Transplantation outcomes in primary hyperoxaluria. Am J Transplant 10:2493–2501
Millan MT, Berquist WE, So SK, Sarwal MM, Wayman KI, Cox KL, Filler G, Salvatierra O Jr, Esquivel CO (2003) One hundred percent patient and kidney allograft survival with simultaneous liver and kidney transplantation in infants with primary hyperoxaluria: a single-center experience. Transplantation 76:1458–1463
Silva M, Silva CH, Iulek J, Thiemann OH (2004) Three-dimensional structure of human adenine phosphoribosyltransferase and its relation to DHA-urolithiasis. Biochemistry 43:7663–7671
Matos V, van Melle G, Boulat O, Markert M, Bachmann C, Guignard JP (1997) Urinary phosphate/creatinine, calcium/creatinine, and magnesium/creatinine ratios in a healthy pediatric population. J Pediatr 131:252–257
Hoppe B, Kemper MJ (2010) Diagnostic examination of the child with urolithiasis or nephrocalcinosis. Pediatr Nephrol 25:403–413
Rumsby G (1998) Experience in prenatal diagnosis of primary hyperoxaluria type 1. J Nephrol 11(Suppl 1):13–14
Barratt TM, Kasidas GP, Murdoch I, Rose GA (1991) Urinary oxalate and glycolate excretion and plasma oxalate concentration. Arch Dis Child 66:501–503
von Schnakenburg C, Byrd DJ, Latta K, Reusz GS, Graf D, Brodehl J (1994) Determination of oxalate excretion in spot urines of healthy children by ion chromatography. Eur J Clin Chem Clin Biochem 32:27–29
Gibbs DA, Watts RW (1969) The variation of urinary oxalate excretion with age. J Lab Clin Med 73:901–908
Morgenstern BZ, Milliner DS, Murphy ME, Simmons PS, Moyer TP, Wilson DM, Smith LH (1993) Urinary oxalate and glycolate excretion patterns in the first year of life: a longitudinal study. J Pediatr 123:248–251
Campfield T, Braden G (1989) Urinary oxalate excretion by very low birth weight infants receiving parenteral nutrition. Pediatrics 84:860–863
Milliner DS, Eickholt JT, Bergstralh EJ, Wilson DM, Smith LH (1994) Results of long-term treatment with orthophosphate and pyridoxine in patients with primary hyperoxaluria. N Engl J Med 331:1553–1558
Morgan SH, Purkiss P, Watts RW, Mansell MA (1987) Oxalate dynamics in chronic renal failure. Comparison with normal subjects and patients with primary hyperoxaluria. Nephron 46:253–257
Acknowledgements
The authors gratefully acknowledge the support of the Rare Kidney Stone Consortium (U54KD083908), a part of the National Institutes of Health (NIH) Rare Diseases Clinical Research Network (RDCRN), funded by the NIDDK, and the NIH Office of Rare Diseases Research (ORDR) and the Mayo Clinic O’Brien Urology Research Center (P50 DK083007) funded by the NIDDK. We thank Rachel Miller of the Mayo Clinic Renal Function Laboratory for photomicrographs of cystine, calcium oxalate, and calcium phosphate crystals, and Hrafnhildur L. Runolfsdottir, a medical student at the University of Iceland, Reykjavik, Iceland, for generating the images of urinary DHA crystals. The authors also thank members of the Rare Kidney Stone Consortium for critical feedback during construction of the diagnostic algorithms (Figs. 3, 4, 5, 6 and 7).
Conflict of interest statement
Dr. Goldfarb is a consultant for Takeda.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Edvardsson, V.O., Goldfarb, D.S., Lieske, J.C. et al. Hereditary causes of kidney stones and chronic kidney disease. Pediatr Nephrol 28, 1923–1942 (2013). https://doi.org/10.1007/s00467-012-2329-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-012-2329-z