Abstract
An adverse fetal environment leads to smaller kidneys, with fewer nephrons, which might predispose an individual to the development of kidney disease and hypertension in adult life. In a prospective cohort study among 1,072 children followed from early fetal life onward, we examined whether maternal smoking during pregnancy, as a significant adverse fetal exposure, is associated with fetal (third trimester of pregnancy, n = 1,031) and infant kidney volume (2 years of age, n = 538) measured by ultrasound. Analyses were adjusted for various potential confounders. Among mothers who continued smoking, we observed dose-dependent associations between the number of cigarettes smoked during pregnancy and kidney volume in fetal life. Smoking less than five cigarettes per day was associated with larger fetal combined kidney volume, while smoking more than ten cigarettes per day tended to be associated with smaller fetal combined kidney volume (p for trend: 0.002). This pattern was not significant for kidney volume at the age of 2 years. Our results suggest that smoking during pregnancy might affect kidney development in fetal life with a dose-dependent relationship. Further studies are needed to assess the underlying mechanisms and whether these differences in fetal kidney volume have postnatal consequences for kidney function and blood pressure.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The developmental plasticity hypothesis suggests that various adverse intra-uterine exposures lead to persistent fetal developmental adaptations. These adaptations may be beneficial in the short term, but may have adverse consequences in postnatal life [1]. Also, they may lead to smaller kidneys with a reduced number of nephrons, which in turn lead to glomerular hyperfiltration and sclerosis, predisposing the individual to renal damage and subsequent development of higher blood pressure, impaired kidney function and end-stage kidney disease in adulthood [2]. This hypothesis is supported by many studies showing the associations between low birth weight and cardiovascular disease and chronic renal failure [3–5]. Low birth weight is also associated with impaired renal growth, raised blood pressure and impaired renal function [5–9]. Thus far, the specific adverse fetal exposures and mechanisms underlying these associations are not known. Maternal smoking is a very important modifiable adverse fetal exposure in western countries and leads to a decrease of 150–200 g in the offspring’s birth weight [10]. Maternal smoking during pregnancy may also have both direct and indirect adverse effects on fetal kidney development. Several studies suggested that maternal smoking during pregnancy might also be associated with higher blood pressure in the offspring, independent of birth weight [11], which might be explained by adverse kidney development [12].
For the present study, we hypothesised that maternal smoking during pregnancy, as a specific adverse fetal exposure, affects early kidney growth. In a population-based prospective cohort study, among 1,072 mothers and children, we evaluated the associations between maternal smoking during pregnancy and kidney volume in fetal life and infancy, both in those mothers who smoked during the first trimester only and in those who continued smoking throughout pregnancy.
Patients and methods
Design
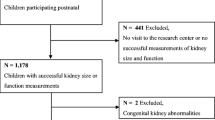
This study was embedded in the Generation R Study, a population-based prospective cohort study on fetal life to young adulthood in Rotterdam, The Netherlands [13, 14]. Detailed assessments were conducted in a subgroup of Dutch children and their parents. Mothers who were already participating during pregnancy were asked to participate in a subcohort for additional detailed renal and cardiovascular measurements. These women were all enrolled before a gestational age of 24 weeks. In total, 80% of these mothers were willing to participate in the subcohort. Data on smoking during pregnancy were available from prospectively collected questionnaires. For the present study, kidney measurements were performed at a gestational age of 30 weeks and at the postnatal age of 24 months. Written informed consent was obtained from all participants. The Medical Ethics Committee of the Erasmus Medical Center Rotterdam approved the study.
Population for analysis
In total 1,232 women were enrolled in the subgroup study. Twin pregnancies (n = 15) and pregnancies leading to perinatal death (n = 2) were excluded from the analysis, leading to 1,215 singleton live births. No renal or uterovesical anomalies other than mild pyelectasis over 10 mm (n = 3) were present in our study. Information about smoking during pregnancy was not available in 110 subjects. Of the remaining 1,105 mothers, fetal kidney characteristics were successfully measured in 1,031 subjects (93%). In total, 67% (n = 740) of the mothers and children with information about smoking during pregnancy participated in the follow-up study at the age of 2 years, with successful kidney measurements in 73% of these children (n = 538). Missing data were mostly due to crying and oppositional behaviour.
In total 1,072 (97%) had at least one complete kidney measurement.
Maternal smoking during pregnancy
Information about active smoking was obtained by postal questionnaires in the first, second and third trimester of pregnancy. Response rates for these questionnaires were 90%, 93% and 92% respectively [15]. Active maternal smoking at enrolment was assessed in the first questionnaire by asking whether the mother smoked in pregnancy (no, in the first trimester only, or whether she continued smoking during the first trimester). In the second and third-trimester questionnaires, mothers were asked whether they had smoked in the past 2 months (no, yes). Among the smoking mothers, the number of cigarettes was classified into different categories: less than five cigarettes per day; five to ten cigarettes per day; and more than ten cigarettes per day. Dose–response analyses for the first trimester only and for mothers who continued smoking were based on the first and third trimester questionnaires respectively.
Kidney measurements
All ultrasound examinations were performed using an ATL-Philips Model HDI 5000 (Seattle, WA, USA) equipped with a 5.0-MHz, high-frequency curved array transducer. Ultrasound examinations were carried out in a dedicated and well-equipped research centre in the third trimester of pregnancy. Fetal left and right kidney biometrics were assessed. In a sagittal plane, the maximum longitudinal kidney length was measured placing the callipers on the outer edges of the caudal and cranial side. Antero-posterior and transverse kidney diameter were measured perpendicular to each other, from the one outer edge to the other, in an axial plane. The cross-sectional area in which the kidney appeared symmetrically round at its maximum width was used. The images were sufficiently magnified to ensure optimal measurements [16]. Postnatally, two-dimensional ultrasounds of the kidneys were performed in children at the age of 24 months. The examination was carried out in a quiet room with the child quietly awake in a supine position. This position was standardised to prevent differences according to the position [16, 17]. The kidney was identified in the sagittal plane along its longitudinal axis. Measures of maximal bipolar kidney length, width and depth were obtained from both the left and the right kidney. Kidney width and depth were measured at the level of the kidney hilum [16, 17]. Both fetal and postnatal kidney volumes were calculated using the equation of an ellipsoid: volume (cm3) = 0.523 × length (mm) × width (mm) × depth (mm). Left and right kidney volumes were added for the combined kidney volume (cm3) [18].
Intra- and inter-observer reproducibility
Two well-trained, experienced sonographers performed all measurements. Quality checks were frequently carried out and feedback was provided to minimise inter-operator differences. We have previously shown that kidney characteristics can be reliably measured in children [19]. The intraobserver interclass correlation coefficients (ICC) ranged from 0.93 (left and right kidney width and right renal thickness) to 0.99 (left kidney length) and interobserver ICC ranged from 0.64 (right kidney thickness) to 0.90 (right kidney length), indicating good reproducibility.
Covariates
Maternal height was measured at enrolment. Information on maternal pre-pregnancy weight and maternal educational level was assessed using self-reported questionnaires. We defined maternal weight gain as the difference between weight at enrolment and weight in the third trimester of the pregnancy, as described before [20]. The educational level of the father and the family income were also assessed using questionnaires at enrolment. Total daily energy intake was established using semiquantitative food frequency questionnaires in the first trimester of the pregnancy. Exact gestational age was established using fetal biometry measured in the first trimester of the pregnancy [21]. Fetal biometrics including head circumference (HC), abdominal circumference (AC) and femur length (FL) were measured using standardised ultrasound procedures in the third trimester of the pregnancy and estimated fetal weight (EFW) was calculated using the formula by Hadlock: EFW (g) = 10 × (1.326 – 0.00326 × AC × FL + 0.0107 × HC + 0.0438 × AC + 0.158 × FL) [22]. Fetal biometrics was measured at the same visit as the fetal kidney measurement. Date of birth, birth weight and sex were obtained from the midwife and from the hospital registries. Postnatal height and weight were measured at the same visit as the postnatal kidney measurement.
Statistical methods
Differences in maternal and offspring characteristics among the non-smoking, the first trimester only and the continuing smokers were assessed using t tests and non-parametric tests for independent samples. Associations between maternal smoking, both first trimester only and continued smoking, and kidney volume were assessed using multiple linear regression models. We adjusted for covariates that changed the effect estimate of smoking during pregnancy on kidney volume by more than 10%, when added to the baseline model, including gender, age and estimated fetal weight. All regression models were adjusted for fetal sex, maternal height and weight before pregnancy, maternal weight gain during pregnancy, total daily caloric intake, maternal and paternal educational level, and household income. We further adjusted the analyses focussing on fetal kidney volume for estimated fetal weight in the third trimester of the pregnancy (30 weeks) and gestational age at the visit. The models focussing on postnatal kidney volume were additionally adjusted for age, weight and height at the visit. Systolic and diastolic blood pressure at intake, parity, maternal alcohol use during pregnancy and folic acid supplement use did not materially change the effect estimate.
Subsequently, using similar models we examined the associations between the number of cigarettes smoked and kidney volumes. Missing values in covariates ranged from 0 to 10%, except for paternal educational level (13.9%) and daily total energy intake (14.7%), and were imputed as the mean for continuous variables and a separate category for categorical variables. Since no differences in the main results were observed between data on complete and imputed analyses, we only present data on imputed analyses. Tests for trends were performed using multiple non-linear regression analyses, including a squared term of the smoking during pregnancy categories, using the non-smoking group as the reference group. All measures of association are presented with their 95% confidence intervals (95% CI). We considered a p value lower than 0.05 to be statistically significant. All statistical analyses were performed using the Statistical Package for the Social Sciences version 15.0 for Windows (SPSS, Chicago, IL, USA).
Results
Subject characteristics are presented in Table 1. The percentage of boys was 52.1%. The overall median maternal age was 31.8 (95% range: 23.5–37.7) years. The study group comprised 31 children who were born with a small size for gestational age (birth weight less than −2 SDS for gestational age), 37 children with a birth weight less than 2,500 g, and 41 children who were born preterm. Table 2 shows all kidney characteristics measured in the third trimester of pregnancy and at the postnatal age of 2 years.
Table 3 gives the adjusted associations between maternal smoking during pregnancy and fetal and postnatal combined volume. Overall, compared with non-smoking, no significant associations between first trimester only smoking, or continued smoking during pregnancy, and fetal kidney volume were observed. Smoking during the first trimester only, but not continued smoking, was positively associated with combined kidney volume at the age of 2 years. Among first trimester only smokers, we did not observe any consistent dose–response associations between the number of cigarettes smoked and the combined kidney volume in fetal life or at the age of 2 years (data not shown).
Among mothers who continued smoking during pregnancy, we observed a dose-dependent association between the number of cigarettes smoked during the third trimester and the fetal combined kidney volume (Table 3). Compared with non-smoking, smoking less than five cigarettes per day was associated with larger fetal combined kidney volume. Smoking more than ten cigarettes per day tended to be associated with smaller combined kidney volume. We observed non-significant associations between the number of cigarettes smoked during pregnancy and kidney volumes at the age of 2 years.
Figure 1 shows that the regression coefficients of multiple linear regressions on combined kidney volume and estimated fetal weight are affected separately by smoking throughout pregnancy. Estimated fetal weight proportionally decreased with the number of cigarettes smoked per day (p value for trend: 0.01). Combined kidney volume, however, was larger in offspring of mothers who smoked less than five cigarettes per day, and became smaller with increasing numbers of cigarettes smoked per day (p value for trend: 0.003). Figure 2 shows the associations between continued maternal smoking during pregnancy and postnatal combined kidney volume and body weight. No significant associations were observed.
Bars represent regression coefficients (95% CI) and reflect the difference in the standard deviation scores (SDS) of the fetal kidney volume and the estimated fetal weight for different numbers of cigarettes smoked continuously throughout pregnancy. All regression models were adjusted for fetal sex, gestational age at the visit, maternal height and weight before pregnancy, maternal weight gain during pregnancy, systolic and diastolic blood pressure at intake, parity, maternal alcohol use during pregnancy, folic acid supplement use, total daily caloric intake, maternal and paternal educational level, and household income. The model for estimated fetal weight was adjusted for the same covariates
Bars represent regression coefficients (95% CI) and reflect the difference in SDS of the kidney volume at 2 years of age and weight at 2 years of age for different numbers of cigarettes smoked continuously throughout pregnancy. All regression models were adjusted for sex, age at the visit, maternal height and weight before pregnancy, maternal weight gain during pregnancy, systolic and diastolic blood pressure at intake, parity, maternal alcohol use during pregnancy, folic acid supplement use, total daily caloric intake, maternal and paternal educational level, and household income. The model for weight at 2 years of age was adjusted for the same covariates
Discussion
Results from this population-based prospective cohort study from early fetal life onward suggest that first-trimester-only smoking is not consistently associated with kidney volume in fetal life and infancy, whereas continued smoking during pregnancy does affect kidney volume in fetal life. The effect size and direction depends on the number of cigarettes smoked. Smoking less than five cigarettes per day was associated with a larger fetal combined kidney volume. This association was not significant at the age of 2 years. Smoking more than ten cigarettes per day tended to be associated with a smaller combined kidney volume in both fetal and postnatal life.
Maternal smoking during pregnancy is an important modifiable adverse fetal exposure leading to various pregnancy complications in western countries [10, 23]. The recently demonstrated associations between maternal smoking during pregnancy and high blood pressure in the offspring suggest that fetal exposure to smoking has permanent renal and cardiovascular consequences [24]. We showed associations between continued smoking during pregnancy and fetal kidney volume, but not postnatal kidney volume. The effect of continued smoking during pregnancy was dependent on the number of cigarettes smoked. This suggests a differential effect of maternal smoking during pregnancy, depending on the specific period of exposure and the number of cigarettes smoked.
The main strength of our study is the prospective design from early fetal life onward and the size of the population-based cohort. Our analyses were based on more than 1,500 complete kidney ultrasounds. The size of the cohort enabled us to assess the associations between the number of cigarettes smoked during pregnancy and the kidney volume. The ultrasound measurements were carried out by two sonographers with good reproducibility.
Of all children with available data on maternal smoking during pregnancy, 67% participated in the follow-up measurements at the age of 2 years. Subjects who did not visit the research centre in postnatal life did not differ with regard to fetal kidney volume from the original sample. However, more mothers in this group continued smoking during pregnancy and mothers were less well educated than those in the original sample. This selective loss to follow-up might have led to the loss of power in the postnatal analyses, and possibly selection bias. Although we performed a meticulous adjustment, especially regarding socioeconomic status, residual confounding might still have been an issue because of the observational design of the study.
Another reason for the lack of associations in postnatal life might be that kidney measurements are more difficult at the age of 2 years, possibly introducing random error, which leads to a reduction in statistical power. One might also assume that some hypertrophy of nephrons is already ongoing in kidneys with lower nephron numbers (i.e. smaller fetal kidney volume) due to heavy smoking mothers, decreasing the difference in kidney volume over time. The increased kidney volume in mothers who smoked less than five cigarettes per day might have disappeared because of the return to the normal kidney growth curve in postnatal life, while the reduced body size catches up.
Using questionnaires for collecting data on smoking behaviour might have resulted in underestimation of the number of cigarettes smoked per day, since some individuals might not report their smoking behaviour accurately. We did not measure cotinine levels to check the smoking status in the mothers. Although assessing maternal smoking during pregnancy seems to be a valid method, misclassification may occur [25, 26]. Another limitation might be that the present study was performed in a healthy population-based cohort. The absolute numbers of subjects with low birth weight and preterm birth were small. Therefore, the generalisability of our results to preterm or low birth weight children is limited.
One other study among 34 subjects showed that maternal smoking during pregnancy leads to a different fetal kidney growth pattern compared with non-smoke-exposed subjects. This study suggested that reduction in kidney growth is present in the third trimester of pregnancy, resulting in relatively thinner kidneys [12]. This is in line with our finding that smoking more than ten cigarettes in the third trimester negatively affects the fetal and infant kidney volume. To our knowledge, no other studies have examined the associations between maternal smoking during pregnancy and kidney size.
The mechanisms underlying the associations between smoking during pregnancy and fetal and infant kidney volume found in this study are not known. Animal studies showed that nicotine has both a vasodilatory and a vasoconstrictive effect on the vasculature, depending on the dose [27–29]. A higher dose of nicotine was associated with vasoconstriction and might partly explain our findings of smaller kidney size when large numbers of cigarettes are smoked during pregnancy [29]. The larger kidneys among subjects exposed to less than five cigarettes per day could be explained by the vasodilatory effect of nicotine on the renal vasculature. Animal studies showed the vasodilatory effects of low-dose nicotine in the kidney as pre-constricted kidney vasculature [30, 31]. Maternal smoking during pregnancy affects fetal growth in different trimesters of pregnancy, with the largest effect in the third trimester. Results suggest that smoking during pregnancy preferentially affects peripheral tissues [25]. The vasodilatory effect of nicotine on the kidney vasculature might lead to increased kidney volume, despite the negative effect on the estimated fetal weight. However, further studies are needed to elucidate the vasoactive effects of nicotine in growth-restricted subjects.
Another possible mechanism could be the role of the renin–angiotensin system (RAS). It has been shown that an intact RAS is needed for normal kidney development and disruption is associated with a lower glomerular number, decreased renal function and increased adult blood pressure [32, 33]. It also has been shown that smoking during pregnancy alters the AT1/AT2 receptor ratio in renal tissue [34]. This could partly explain our findings that smoking more than ten cigarettes per day is associated with smaller fetal kidney volume. Further studies are needed to evaluate the effect of maternal smoking during pregnancy on RAS in humans.
The associations between low birth weight and hypertension, impaired renal function and end-stage renal disease in adulthood are well established [3, 6, 9, 35, 36]. Smaller kidneys with a reduced number of nephrons in low birth weight children might lead to hyperfiltration resulting in glomerular sclerosis [2, 37–40]. This may predispose the individual to renal damage and development of higher blood pressure, impaired kidney function and end-stage kidney disease in adulthood. This hypothesis is supported by animal studies that have shown that various adverse intrauterine environmental exposures, such as low protein intake, lead to fetal growth retardation and smaller kidney size with a lower nephron number [41–43]. Postmortem studies in humans showed that a lower nephron number is associated with both low birth weight and hypertension [37, 39, 44, 45].
Newborn kidney volume has been shown to be a surrogate of total nephron number. This was shown in 15 infants who died before 3 months of age, in whom an ultrasound was performed in the first 2 days of life. There was a strong and direct relationship between kidney mass and nephron number [46]. Since the nephron number varies between 250,000 and 2,000,000 per kidney and nephron development ceases after birth [44, 47], these findings suggest that early kidney development might be an underlying mechanism for the associations between maternal smoking during pregnancy and increased blood pressure in later life. Whether the differences in kidney volume due to smoking during pregnancy found in this study also correlate with nephron number needs to be studied.
Conclusion
Our results suggest that maternal smoking during pregnancy might be associated with altered kidney volume in late pregnancy, but not in infancy. The direction and size of the effect depends on the number of cigarettes smoked. These results should be considered as hypothesis-generating. Further studies are needed to identify the underlying mechanisms and to assess whether these changes in kidney size in early life are related to renal function and high blood pressure development in later life.
References
Gluckman PD, Hanson MA, Cooper C, Thornburg KL (2008) Effect of in utero and early-life conditions on adult health and disease. N Engl J Med 359:61–73
Brenner BM, Chertow GM (1994) Congenital oligonephropathy and the etiology of adult hypertension and progressive renal injury. Am J Kidney Dis 23:171–175
Huxley RR, Shiell AW, Law CM (2000) The role of size at birth and postnatal catch-up growth in determining systolic blood pressure: a systematic review of the literature. J Hypertens 18:815–831
Eriksson JG, Forsen T, Tuomilehto J, Osmond C, Barker DJ (2001) Early growth and coronary heart disease in later life: longitudinal study. BMJ 322:949–953
Lackland DT, Bendall HE, Osmond C, Egan BM, Barker DJ (2000) Low birth weights contribute to high rates of early-onset chronic renal failure in the Southeastern United States. Arch Intern Med 160:1472–1476
Hoy WE, Rees M, Kile E, Mathews JD, McCredie DA, Pugsley DJ, Wang Z (1998) Low birthweight and renal disease in Australian aborigines. Lancet 352:1826–1827
Nelson RG, Morgenstern H, Bennett PH (1998) Birth weight and renal disease in Pima Indians with type 2 diabetes mellitus. Am J Epidemiol 148:650–656
Keijzer-Veen MG, Schrevel M, Finken MJ, Dekker FW, Nauta J, Hille ET, Frolich M, van der Heijden BJ (2005) Microalbuminuria and lower glomerular filtration rate at young adult age in subjects born very premature and after intrauterine growth retardation. J Am Soc Nephrol 16:2762–2768
White SL, Perkovic V, Cass A, Chang CL, Poulter NR, Spector T, Haysom L, Craig JC, Salmi IA, Chadban SJ, Huxley RR (2009) Is low birth weight an antecedent of CKD in later life? A systematic review of observational studies. Am J Kidney Dis 54:248–261
Jaddoe VW, Troe EJ, Hofman A, Mackenbach JP, Moll HA, Steegers EA, Witteman JC (2008) Active and passive maternal smoking during pregnancy and the risks of low birthweight and preterm birth: the Generation R Study. Paediatr Perinat Epidemiol 22:162–171
Lawlor DA, Najman JM, Sterne J, Williams GM, Ebrahim S, Davey Smith G (2004) Associations of parental, birth, and early life characteristics with systolic blood pressure at 5 years of age: findings from the Mater-University study of pregnancy and its outcomes. Circulation 110:2417–2423
Lampl M, Kuzawa CW, Jeanty P (2005) Growth patterns of the heart and kidney suggest inter-organ collaboration in facultative fetal growth. Am J Hum Biol 17:178–194
Jaddoe VW, Bakker R, van Duijn CM, van der Heijden AJ, Lindemans J, Mackenbach JP, Moll HA, Steegers EA, Tiemeier H, Uitterlinden AG, Verhulst FC, Hofman A (2007) The Generation R Study Biobank: a resource for epidemiological studies in children and their parents. Eur J Epidemiol 22:917–923
Jaddoe VW, van Duijn CM, van der Heijden AJ, Mackenbach JP, Moll HA, Steegers EA, Tiemeier H, Uitterlinden AG, Verhulst FC, Hofman A (2010) The Generation R Study: design and cohort update 2010. Eur J Epidemiol 25:823–841
Jaddoe VW, van Duijn CM, van der Heijden AJ, Mackenbach JP, Moll HA, Steegers EA, Tiemeier H, Uitterlinden AG, Verhulst FC, Hofman A (2008) The Generation R Study: design and cohort update until the age of 4 years. Eur J Epidemiol 23:801–811
Jeanty P, Dramaix-Wilmet M, Elkhazen N, Hubinont C, van Regemorter N (1982) Measurements of fetal kidney growth on ultrasound. Radiology 144:159–162
Gupta AK, Anand NK, Lamba IM (1993) Ultrasound evaluation of kidney dimensions in neonates. Indian Pediatr 30:319–324
Geelhoed JJ, Taal HR, Steegers EA, Arends LR, Lequin M, Moll HA, Hofman A, van der Heijden AJ, Jaddoe VW (2010) Kidney growth curves in healthy children from the third trimester of pregnancy until the age of two years. The Generation R Study. Pediatr Nephrol 25:289–298
Geelhoed JJ, Kleyburg-Linkers VE, Snijders SP, Lequin M, Nauta J, Steegers EA, van der Heijden AJ, Jaddoe VW (2009) Reliability of renal ultrasound measurements in children. Pediatr Nephrol 24:1345–1353
Ay L, Kruithof CJ, Bakker R, Steegers EA, Witteman JC, Moll HA, Hofman A, Mackenbach JP, Hokken-Koelega AC, Jaddoe VW (2009) Maternal anthropometrics are associated with fetal size in different periods of pregnancy and at birth. The Generation R Study. BJOG 116:953–963
Verburg BO, Steegers EA, De Ridder M, Snijders RJ, Smith E, Hofman A, Moll HA, Jaddoe VW, Witteman JC (2008) New charts for ultrasound dating of pregnancy and assessment of fetal growth: longitudinal data from a population-based cohort study. Ultrasound Obstet Gynecol 31:388–396
Hadlock FP, Harrist RB, Carpenter RJ, Deter RL, Park SK (1984) Sonographic estimation of fetal weight. The value of femur length in addition to head and abdomen measurements. Radiology 150:535–540
Andres RL, Day MC (2000) Perinatal complications associated with maternal tobacco use. Semin Neonatol 5:231–241
Brion MJ, Leary SD, Lawlor DA, Smith GD, Ness AR (2008) Modifiable maternal exposures and offspring blood pressure: a review of epidemiological studies of maternal age, diet, and smoking. Pediatr Res 63:593–598
Jaddoe VW, Verburg BO, de Ridder MA, Hofman A, Mackenbach JP, Moll HA, Steegers EA, Witteman JC (2007) Maternal smoking and fetal growth characteristics in different periods of pregnancy: the generation R study. Am J Epidemiol 165:1207–1215
Klebanoff MA, Levine RJ, Morris CD, Hauth JC, Sibai BM, Ben Curet L, Catalano P, Wilkins DG (2001) Accuracy of self-reported cigarette smoking among pregnant women in the 1990s. Paediatr Perinat Epidemiol 15:140–143
Zhang W, Edvinsson L, Lee TJ (1998) Mechanism of nicotine-induced relaxation in the porcine basilar artery. J Pharmacol Exp Ther 284:790–797
Marano G, Ramirez A, Mori I, Ferrari AU (1999) Sympathectomy inhibits the vasoactive effects of nicotine in conscious rats. Cardiovasc Res 42:201–205
Wang R, Wang Z (2000) Three different vasoactive responses of rat tail artery to nicotine. Can J Physiol Pharmacol 78:20–28
Ay I, Tuncer M (1998) Vasodilatation induced by nicotine in the isolated perfused rat kidney. Gen Pharmacol 30:651–654
El-Mas MM, El-Gowilly SM, Gohar EY, Ghazal AR (2008) Pharmacological characterization of cellular mechanisms of the renal vasodilatory effect of nicotine in rats. Eur J Pharmacol 588:294–300
Guron G, Friberg P (2000) An intact renin-angiotensin system is a prerequisite for normal renal development. J Hypertens 18:123–137
Woods LL, Rasch R (1998) Perinatal ANG II programs adult blood pressure, glomerular number, and renal function in rats. Am J Physiol 275:R1593–R1599
Mao C, Wu J, Xiao D, Lv J, Ding Y, Xu Z, Zhang L (2009) The effect of fetal and neonatal nicotine exposure on renal development of AT(1) and AT(2) receptors. Reprod Toxicol 27:149–154
Curhan GC, Willett WC, Rimm EB, Spiegelman D, Ascherio AL, Stampfer MJ (1996) Birth weight and adult hypertension, diabetes mellitus, and obesity in US men. Circulation 94:3246–3250
Lawlor DA, Hubinette A, Tynelius P, Leon DA, Smith GD, Rasmussen F (2007) Associations of gestational age and intrauterine growth with systolic blood pressure in a family-based study of 386,485 men in 331,089 families. Circulation 115:562–568
Hinchliffe SA, Lynch MR, Sargent PH, Howard CV, Van Velzen D (1992) The effect of intrauterine growth retardation on the development of renal nephrons. Br J Obstet Gynaecol 99:296–301
Brenner BM, Lawler EV, Mackenzie HS (1996) The hyperfiltration theory: a paradigm shift in nephrology. Kidney Int 49:1774–1777
Manalich R, Reyes L, Herrera M, Melendi C, Fundora I (2000) Relationship between weight at birth and the number and size of renal glomeruli in humans: a histomorphometric study. Kidney Int 58:770–773
Silver LE, Decamps PJ, Korst LM, Platt LD, Castro LC (2003) Intrauterine growth restriction is accompanied by decreased renal volume in the human fetus. Am J Obstet Gynecol 188:1320–1325
Celsi G, Kistner A, Aizman R, Eklof AC, Ceccatelli S, de Santiago A, Jacobson SH (1998) Prenatal dexamethasone causes oligonephronia, sodium retention, and higher blood pressure in the offspring. Pediatr Res 44:317–322
Lelievre-Pegorier M, Vilar J, Ferrier ML, Moreau E, Freund N, Gilbert T, Merlet-Benichou C (1998) Mild vitamin A deficiency leads to inborn nephron deficit in the rat. Kidney Int 54:1455–1462
Langley-Evans SC, Welham SJ, Jackson AA (1999) Fetal exposure to a maternal low protein diet impairs nephrogenesis and promotes hypertension in the rat. Life Sci 64:965–974
Hughson M, Farris AB 3rd, Douglas-Denton R, Hoy WE, Bertram JF (2003) Glomerular number and size in autopsy kidneys: the relationship to birth weight. Kidney Int 63:2113–2122
Keller G, Zimmer G, Mall G, Ritz E, Amann K (2003) Nephron number in patients with primary hypertension. N Engl J Med 348:101–108
Zhang Z, Quinlan J, Hoy W, Hughson MD, Lemire M, Hudson T, Hueber PA, Benjamin A, Roy A, Pascuet E, Goodyer M, Raju C, Houghton F, Bertram J, Goodyer P (2008) A common RET variant is associated with reduced newborn kidney size and function. J Am Soc Nephrol 19:2027–2034
Nyengaard JR, Bendtsen TF (1992) Glomerular number and size in relation to age, kidney weight, and body surface in normal man. Anat Rec 232:194–201
Acknowledgements
The Generation R study is being conducted by the Erasmus Medical Center in close collaboration with the School of Law and Faculty of Social Sciences of Erasmus University Rotterdam; the Municipal Health Service Rotterdam area, Rotterdam; the Rotterdam Homecare Foundation, Rotterdam; and the Stichting Trombosedienst & Artsenlaboratorium Rijnmond, Rotterdam. We gratefully acknowledge the contributions of general practitioners, hospitals, midwives and pharmacies in Rotterdam.
Financial disclosure
The first phase of the Generation R Study has been made possible by financial support from the Erasmus Medical Center, Rotterdam; Erasmus University Rotterdam; and The Netherlands Organization for Health Research and Development (ZonMw). Additional support was provided by a grant from the Dutch Kidney Foundation (C08.2251).
Conflict of interest
All the authors declared that they have no competing interests.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Taal, H.R., Geelhoed, J.J.M., Steegers, E.A.P. et al. Maternal smoking during pregnancy and kidney volume in the offspring: the Generation R Study. Pediatr Nephrol 26, 1275–1283 (2011). https://doi.org/10.1007/s00467-011-1848-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-011-1848-3