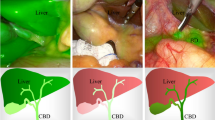
Abstract
Background
Ideal visualization of fluorescent cholangiography during laparoscopic cholecystectomy is when maximum fluorescence into biliary ducts and absent signal into liver parenchyma, defined as “signal to background ratio” (SBR), is obtained. Such condition is mainly dependent by indocyanine green (ICG) dose and timing. The aim of this study was to identify the ideal ICG dose to obtain the best possible intraoperative visualization of the extra-hepatic biliary tree.
Methods
The first part of the study was used to define a range of small weight-based ICG dosages using the mathematical function bisection method. During the second part of the study, the midpoint dose of the identified range, was tested in 50 consecutive cholecystectomies using a laser-based fluorescence laparoscopic camera (SynergyID system by Arthrex, Naples, FL, USA). Timing administration was set at 1 h before surgery, since this is the most common situation in clinical practice. Fluorescence intensity of bile ducts and liver parenchyma were assessed both subjectively, by blinded operative surgeon, as well as objectively, using an image analysis software (Fiji plugin), before and after Calot’s triangle dissection.
Results
Fourteen patients were included in the first part of the study and ICG dose between 0.01191406 and 0.0119873 mg/kg was identified. The second part confirmed previous results after testing the dosage equal to 0.0119 mg/kg (midpoint of the defined range) in 50 consecutive cholecystectomies. Cystic duct was identified in 66 and 100% of cases before and after dissection of Calot’s triangle respectively. On the other hand, common bile duct was identified in 82 and 92% before and after dissection respectively. Subjective and objective SBRs confirmed the benefit of the identified ICG dose.
Conclusion
ICG dose calculated by 0.0119 mg/kg administered one hour before surgery allows an ideal intraoperative visualization of the extra-hepatic biliary tree.
Registration number: ISRCTN10190039.
Graphical abstract




Similar content being viewed by others
References
Gollan J, Bulkley PL, Diehl A (1993) NIH consensus conference: gallstones and laparoscopic cholecystectomy. JAMA 269(8):1018–1024
Deziel DJ, Millikan KW, Economou SG, Doolas A, Ko ST, Airan MC (1993) Complications of laparoscopic cholecystectomy: a national survey of 4,292 hospitals and an analysis of 77,604 cases. Am J Surg 165(1):9–14
Flum DR, Dellinger EP, Cheadle A, Chan L, Koepsell T (2003) Intraoperative cholangiography and risk of common bile duct injury during cholecystectomy. JAMA 289:1639–1644
Strasberg SM, Hertl M, Soper NJ (1995) An analysis of the problem of biliary injury during laparoscopic cholecystectomy. J Am Coll Surg 180:101–125
Pucher PH, Brunt LM, Fanelli RD, Asbun HJ, Aggarwal R (2015) SAGES expert Delphi consensus: critical factors for safe surgical practice in laparoscopic cholecystectomy. Surg Endosc 29:3074–3085
Aziz O, Ashrafian H, Jones C, Harling L, Kumar S, Garas G, Holme T, Darzi A, Zacharakis E, Athanasiou T (2014) Laparoscopic ultrasonography versus intra-operative cholangiogram for the detection of common bile duct stones during laparoscopic cholecystectomy: a meta-analysis of diagnostic accuracy. Int J Surg 12:712–719
Phillips EH, Carroll BJ, Fallas MJ (1993) Laparoscopically guided cholecystectomy: a detailed report of the first 453 cases performed by one surgical team. Am Surg 59:235–242 (PMID: 8489085)
Ishizawa T, Bandai Y, Ijichi M, Kaneko J, Hasegawa K, Kokudo N (2010) Fluorescent cholangiography illuminating the biliary tree during laparoscopic cholecystectomy. Br J Surg 97(9):1369–1377
Dip F, Roy M, Lo Menzo E, Simpfendorfer C, Szomstein S, Rosenthal RJ (2015) Routine use of fluorescent incisionless cholangiography as a new imaging modality during laparoscopic cholecystectomy. Surg Endosc 29:1621–1626
Dip F, LoMenzo E, Sarotto L, Phillips E, Todeschini H, Nahmod M, Alle L, Schneider S, Kaja L, Boni L, Ferraina P, Carus T, Kokudo N, Ishizawa T, Walsh M, Simpfendorfer C, Mayank R, White K, Rosenthal RJ (2019) Randomized trial of nearinfrared incisionless fluorescent cholangiography. Ann Surg 270(6):992–999
Verbeek FPR, Schaafsma BE, Tummers QR, van der Vorst JR, van der Made WJ, Baeten CI, Bonsing BA, Frangioni JV, van de Velde CJ, Vahrmeijer AL, Swijnenburg RJ (2014) Optimization of near-infrared fluorescence cholangiography for open and laparoscopic surgery. Surg Endosc 28:1076–1082
Boogerd LSF, Handgraaf HJM, Huurman VAL, Lam HD, Mieog JSD, van der Made WJ, van de Velde CJH, Vahrmeijer AL (2017) The best approach for laparoscopic fluorescence cholangiography: overview of the literature and optimization of dose and dosing time. Surg Innov 24(4):386–396
Tsutsui N, Yoshida M, Nakagawa H, Ito E, Iwase R, Suzuki N, Imakita T, Ohdaira H, Kitajima M, Yanaga K, Suzuki Y (2018) Optimal timing of preoperative indocyanine green administration for fluorescent cholangiography during laparoscopic cholecystectomy using the PINPOINT® endoscopic fluorescence imaging system. Asian J Endosc Surg 11(3):199–205
Matsumura M, Kawaguchi Y, Kobayashi Y, Kobayashi K, Ishizawa T, Akamatsu N, Kaneko J, Arita J, Kokudo N, Hasegawa K (2021) Indocyanine green administration a day before surgery may increase bile duct detectability on fluorescence cholangiography during laparoscopic cholecystectomy. J Hepatobiliary Pancreat Sci 28(2):202–210
Zarrinpar A, Dutson EP, Mobley C, Busuttil RW, Lewis CE, Tillou A, Cheaito A, Hines OJ, Agopian VG, Hiyama DT (2016) Intraoperative laparoscopic near-infrared fluorescence cholangiography to facilitate anatomical identification: when to give indocyanine green and how much. Surg Innov 23(4):360–365
Zarate Rodriguez JG, Hammill CW (2021) Micro-dosing of indocyanine green for intraoperative fluorescence cholangiography. Surg Technol Int 20(38):98–101
Chen Q, Zhou R, Weng J, Lai Y, Liu H, Kuang J, Zhang S, Wu Z, Wang W, Gu W (2021) Extrahepatic biliary tract visualization using near-infrared fluorescence imaging with indocyanine green: optimization of dose and dosing time. Surg Endosc 35(10):5573–5582
Pujol Cano N, Molina Romero FX, Palma Zamora E, Bonnin Pascual J, Coll Sastre M, Gonzàlez-Argenté FX (2021) Near-infrared fluorescence cholangiography at a very low dose of indocyanine green: a prospective experimental study. HPB 23(3):S700
Mathew G, Agha R, for the STROCSS Group (2021) STROCSS 2021: strengthening the reporting of cohort, cross-sectional and case-control studies in surgery. Int J Surg 96:106165
Dip FD, Asbun D, Rosales-Velderrain A, Lo Menzo E, Simpfendorfer CH, Szomstein S, Rosenthal RJ (2014) Cost analysis and effectiveness comparing the routine use of intraoperative fluorescent cholangiography with fluoroscopic cholangiogram in patients undergoing laparoscopic cholecystectomy. Surg Endosc 28:1838–1843
van den Bos J, Wieringa FP, Bouvy ND, Stassen LPS (2018) Optimizing the image of fluorescence cholangiography using ICG: a systematic review and ex vivo experiments. Surg Endosc 32(12):4820–4832
Burden RL, Faires JD (1985) 2.1 The bisection algorithm, numerical analysis, 3rd edn. PWS Publishers
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, Preibisch S, Rueden C, Saalfeld S, Schmid B, Tinevez JY, White DJ, Hartenstein V, Eliceiri K, Tomancak P, Cardona A (2012) Fiji: an open-source platform for biological-image analysis. Nat Method 9(7):676–682
Kono Y, Ishizawa T, Tani K, Harada N, Kaneko J, Saiura A, Bandai Y, Kokudo N (2015) Techniques of fluorescence cholangiography during laparoscopic cholecystectomy for better delineation of the bile duct anatomy. Medicine 94(25):e1005
Perring CS, Barnes SJ, Verrall M, Hill RET (2004) Using automated digital image analysis to provide quantitative petrographic data on olivine–phyric basalts. Comput Geosci 30(2):183–195
Mi MY, Betensky RA (2013) An analysis of adaptive design variations on the sequential parallel comparison design for clinical trials. Clin Trials 10(2):207–215. https://doi.org/10.1177/1740774512468806
Goor DA, Ebert PA (1972) Anomalies of the biliary tree: report of a repair of an accessory bile duct and review of the literature. Arch Surg 104:302–309
Calatayud D, Milone L, Elli EF, Giulianotti PC (2012) ICG fluorescence identification of a small aberrant biliary canaliculus during robotic cholecystectomy. Liver Int 32:602
Wakabayashi G, Iwashita Y, Hibi T, Takada T, Strasberg SM, Asbun HJ, Endo I, Umezawa A, Asai K, Suzuki K, Mori Y, Okamoto K, Pitt HA, Han HS, Hwang TL, Yoon YS, Yoon DS, Choi IS, Huang WS, Giménez ME, Garden OJ, Gouma DJ, Belli G, Dervenis C, Jagannath P, Chan ACW, Lau WY, Liu KH, Su CH, Misawa T, Nakamura M, Horiguchi A, Tagaya N, Fujioka S, Higuchi R, Shikata S, Noguchi Y, Ukai T, Yokoe M, Cherqui D, Honda G, Sugioka A, de Santibañes E, Supe AN, Tokumura H, Kimura T, Yoshida M, Mayumi T, Kitano S, Inomata M, Hirata K, Sumiyama Y, Inui K, Yamamoto M (2018) Tokyo Guidelines 2018: surgical management of acute cholecystitis: safe steps in laparoscopic cholecystectomy for acute cholecystitis (with videos). J Hepatobiliary Pancreat Sci 25(1):73–86
Conrad C, Wakabayashi G, Asbun HJ, Dallemagne B, Demartines N, Diana M, Fuks D, Giménez ME, Goumard C, Kaneko H, Memeo R, Resende A, Scatton O, Schneck AS, Soubrane O, Tanabe M, van den Bos J, Weiss H, Yamamoto M, Marescaux J, Pessaux P (2017) IRCAD recommendation on safe laparoscopic cholecystectomy. J Hepatobiliary Pancreat Sci 24(11):603–615. https://doi.org/10.1002/jhbp.491
Sgaramella LI, Gurrado A, Pasculli A, de Angelis N, Memeo R, Prete FP, Berti S, Ceccarelli G, Rigamonti M, Badessi FGA, Solari N, Milone M, Catena F, Scabini S, Vittore F, Perrone G, de Werra C, Cafiero F, Testini M (2021) SYoN Italian collaborative group: the critical view of safety during laparoscopic cholecystectomy: Strasberg yes or no? An Italian multicenter study. Surg Endosc 35(7):3698–3708
Acknowledgements
We would like to express our warm thanks to Lorenzo Bianconi and Emanuele Martini for their help in image analysis and statistical analysis, and to Matilde Bongio for scientific illustration.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Prof Luigi Boni reports consultancy fees, unrelated to this study, on the behalf of Arthrex. Dr Ludovica Baldari, Dr Hayato Kurihara and Prof Elisa Cassinotti have no conflicts of interest or financial ties to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Baldari, L., Boni, L., Kurihara, H. et al. Identification of the ideal weight-based indocyanine green dose for fluorescent cholangiography. Surg Endosc 37, 7616–7624 (2023). https://doi.org/10.1007/s00464-023-10280-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-023-10280-x