Abstract
Background
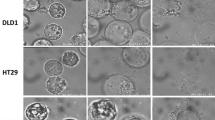
The purpose of the study was to investigate the proliferation and migration capability of human gastrointestinal stromal tumor line GIST-T1 after exposure to different pressures and times of CO2 pneumoperitoneum.
Methods
We established simulated CO2 pneumoperitoneum environment in vitro and divided the human GIST cell GIST-T1 into open control group, 8 mmHg CO2 pneumoperitoneum treatment group and 15 mmHg CO2 pneumoperitoneum treatment group. Each group was divided into two subgroups respectively cultured for 1 h and 3 h. pH value of cell culture, cell growth curve, and cell cycle distribution of each group was measured. By application of scratch healing tests and Transwell chamber experiments, mobility ratio and number of cells through 8 µm membranes were measured to assess the migration ability of cells in each group after intervention.
Results
Cell culture pH value of each subgroup in CO2 group decreased significantly after exposed in CO2 pneumoperitoneum (P < 0.01). The proliferation of GIST-T1 cells in 15 mmHg CO2 group was significantly inhibited early (1–2 days) (P < 0.05) and the proliferation of GIST-T1 cells in 8 mmHg CO2 1 h subgroup and 15 mmHg CO2 1 h subgroup was increased significantly late (4–6 days) (P < 0.05) after the interventions of CO2 pneumoperitoneum. The percentage of cells in G0–G1 phase increased, the percentage of S phase cells decreased (P < 0.01) in 1-h subgroup and 3-h subgroup of 15 mmHg CO2 group 24 h after exposure to CO2. The percentage of cells in S phase increased in 1-h subgroup of 8 mmHg CO2 group and decreased in 3-h subgroup of 15 mmHg CO2 group 72 h after exposure to CO2. In the Transwell chamber experiment, the cell number through 8-µm membrane increased significantly (P < 0.01) in 3-h subgroup of CO2 group compared to that in 3-h subgroup of control group.
Conclusions
The routine pressure and duration of CO2 pneumoperitoneum used in clinic did not promote the proliferation of gastrointestinal stromal tumors, but had a potential risk of increasing postoperative recurrence and distant metastasis.






Similar content being viewed by others
References
Dulucq JL, Wintringer P, Stabilini C, Solinas L, Perissat J, Mahajna A (2005) Laparoscopic and open gastric resections for malignant lesions: a prospective comparative study. Surg Endosc 19:933–938
Shehzad K, Mohiuddin K, Nizami S, Sharma H, Khan IM, Memon B, Memon MA (2007) Current status of minimal access surgery for gastric cancer. Surg Oncol 16:85–98
Ströhlein MA, Grützner KU, Jauch KW, Heiss MM (2008) Comparison of laparoscopic vs. open access surgery in patients with rectal cancer: a prospective analysis. Dis Colon Rectum 51:385–391
Theophilus M, Platell C, Spilsbury K (2014) Long-term survival following laparoscopic and open colectomy for colon cancer: a meta-analysis of randomized controlled trials. Colorectal Dis 16:O75–O81
Whelan RL (2001) Laparotomy, laparoscopy, cancer, and beyond. Surg Endosc 15:110–115
Lécuru F, Agostini A, Camatte S, Robin F, Aggerbeck M, Jaïss JP, Vildé F, Taurelle R (2001) Impact of pneumoperitoneum on visceral metastasis rate and survival. Results in two ovarian cancer models in rats. BJOG 108:733–737
Gutt CN, Bruttel T, Brier C, Paolucci V, Encke A (1998) CO2 pneumoperitoneum inhibits in vitro proliferation of human carcinoma cells. Langenbecks Arch Chir Suppl Kongressbd 115:535–540
Canis M, Botchorishvili R, Wattiez A, Mage G, Pouly JL, Bruhat MA (1998) Tumor growth and dissemination after laparotomy and CO2 pneumoperitoneum: a rat ovarian cancer model. Obstet Gynecol 92:104–108
Zerey M, Burns JM, Kercher KW, Kuwada TS, Heniford BT (2006) Minimally invasive management of colon cancer. Surg Innov 13:5–15
Datar M, Khanna R (2012) Inpatient burden of gastrointestinal stromal tumors in the United States. J Gastrointest Oncol 3:335–341
Patel S (2013) Exploring novel therapeutic targets in GIST: focus on the PI3K/Akt/mTOR pathway. Curr Oncol Rep 15:386–395
Greenson JK (2003) Gastrointestinal stromal tumors and other mesenchymal lesions of the gut. Mod Pathol 16:366–375
Rubin BP, Singer S, Tsao C, Duensing A, Lux ML, Ruiz R, Hibbard MK, Chen CJ, Xiao S, Tuveson DA, Demetri GD, Fletcher CD, Fletcher JA (2001) KIT activation is a ubiquitous feature of gastrointestinal stromal tumors. Cancer Res 61:8118–8121
Agaimy A, Wünsch PH, Hofstaedter F, Blaszyk H, Rümmele P, Gaumann A, Dietmaier W, Hartmann A (2007) Minute gastric sclerosing stromal tumors (GIST tumorlets) are common in adults and frequently show c-KIT mutations. Am J Surg Pathol 31:113–120
Taylor ML, Metcalfe DD (2000) Kit signal transduction. Hematol Oncol Clin North Am 14:517–535
Miettinen M, Lasota J (2006) Gastrointestinal stromal tumors: review on morphology, molecular pathology, prognosis, and differential diagnosis. Arch Pathol Lab Med 130:1466–1478
Lennartsson J, Rönnstrand L (2006) The stem cell factor receptor/c-Kit as a drug target in cancer. Curr Cancer Drug Targets 6:65–75
DeMatteo RP, Lewis JJ, Leung D, Mudan SS, Woodruff JM, Brennan MF (2000) Two hundred gastrointestinal stromal tumors: recurrence patterns and prognostic factors for survival. Ann Surg 231:51–58
Goh BK, Chow PK, Yap WM, Kesavan SM, Song IC, Paul PG, Ooi BS, Chung YF, Wong WK (2008) Which is the optimal risk stratification system for surgically treated localized primary GIST? Comparison of three contemporary prognostic criteria in 171 tumors and a proposal for a modified Armed Forces Institute of Pathology risk criteria. Ann Surg Oncol 15:2153–2163
Nguyen SQ, Divino CM, Wang JL, Dikman SH (2006) Laparoscopic management of gastrointestinal stromal tumors. Surg Endosc 20:713–716
Novitsky YW, Kercher KW, Sing RF, Heniford BT (2006) Long-term outcomes of laparoscopic resection of gastric gastrointestinal stromal tumors. Ann Surg 243:738–745
Sexton JA, Pierce RA, Halpin VJ, Eagon JC, Hawkins WG, Linehan DC, Brunt LM, Frisella MM, Matthews BD (2008) Laparoscopic gastric resection for gastrointestinal stromal tumors. Surg Endosc 22:2583–2587
Otani Y, Furukawa T, Yoshida M, Saikawa Y, Wada N, Ueda M, Kubota T, Mukai M, Kameyama K (2006) Operative indications for relatively small (2–5 cm) gastrointestinal stromal tumor of the stomach based on analysis of 60 operated cases. Surgery 139:484–492
Sasaki A, Koeda K, Obuchi T, Nakajima J, Nishizuka S, Terashima M, Wakabayashi G (2010) Tailored laparoscopic resection for suspected gastric gastrointestinal stromal tumors. Surgery 147:516–520
Zheng MH (2011) Choice and technical problems of digestive tract reconstruction in laparoscopic gastrointestinal surgery. Zhonghua Wei Chang Wai Ke Za Zhi 14:399–402
Veldkamp R, Kuhry E, Hop WC, Jeekel J, Kazemier G, Bonjer HJ, Haglind E, Påhlman L, Cuesta MA, Msika S, Morino M, Lacy AM, COlon cancer Laparoscopic or Open Resection Study Group (COLOR) (2005) Laparoscopic surgery versus open surgery for colon cancer: short-term outcomes of a randomised trial. Lancet Oncol 6:477–484
Jayne DG, Guillou PJ, Thorpe H, Quirke P, Copeland J, Smith AM, Heath RM, Brown JM, UK MRC CLASICC Trial Group (2007) Randomized trial of laparoscopic-assisted resection of colorectal carcinoma: 3-year results of the UK MRC CLASICC Trial Group. J Clin Oncol 25:3061–3068
Song KY, Kim JJ, Kim SN, Park CH (2007) Staging laparoscopy for advanced gastric cancer: is it also useful for the group which has an aggressive surgical strategy? World J Surg 31:1223–1228
Nakagawa S, Nashimoto A, Yabusaki H (2007) Role of staging laparoscopy with peritoneal lavage cytology in the treatment of locally advanced gastric cancer. Gastric Cancer 10:29–34
Mochiki E, Kamiyama Y, Aihara R, Nakabayashi T, Asao T, Kuwano H (2005) Laparoscopic assisted distal gastrectomy for early gastric cancer: five years’ experience. Surgery 137:317–322
Shimizu S, Noshiro H, Nagai E, Uchiyama A, Tanaka M (2003) Laparoscopic gastric surgery in a Japanese institution: analysis of the initial 100 procedures. J Am Coll Surg 197:372–378
Döbrönte Z, Wittmann T, Karácsony G (1978) Rapid development of malignant metastases in the abdominal wall after laparoscopy. Endoscopy 10:127–130
Are C, Talamini MA (2005) Laparoscopy and malignancy. J Laparoendosc Adv Surg Tech A 15:38–47
Jacobi CA, Wenger F, Sabat R, Volk T, Ordemann J, Müller JM (1998) The impact of laparoscopy with carbon dioxide versus helium on immunologic function and tumor growth in a rat model. Dig Surg 15:110–116
Joensuu H, Hohenberger P, Corless CL (2013) Gastrointestinal stromal tumour. Lancet 382:973–983
Goh BK, Chow PK, Kesavan S, Yap WM, Wong WK (2008) Outcome after surgical treatment of suspected gastrointestinal stromal tumors involving the duodenum: is limited resection appropriate? J Surg Oncol 97:388–391
Wu PC, Langerman A, Ryan CW, Hart J, Swiger S, Posner MC (2003) Surgical treatment of gastrointestinal stromal tumors in the imatinib (STI-571) era. Surgery 134:656–665
Roberts PJ, Eisenberg B (2002) Clinical presentation of gastrointestinal stromal tumors and treatment of operable disease. Eur J Cancer 38:S37–S38
Rossi CR, Mocellin S, Mencarelli R, Foletto M, Pilati P, Nitti D, Lise M (2003) Gastrointestinal stromal tumors: from a surgical to a molecular approach. Int J Cancer 107:171–176
McMahon AJ, Baxter JN, Kenny G, O’Dwyer PJ (1993) Ventilatory and blood gas changes during laparoscopic and open cholecystectomy. Br J Surg 80:1252–1254
Volz J, Köster S, Weiss M, Schmidt R, Urbaschek R, Melchert F, Albrecht M (1996) Pathophysiologic features of a pneumoperitoneum at laparoscopy: a swine model. Am J Obstet Gynecol 174:132–140
Kuntz C, Wunsch A, Bödeker C, Bay F, Rosch R, Windeler J, Herfarth C (2000) Effect of pressure and gas type on intraabdominal, subcutaneous, and blood pH in laparoscopy. Surg Endosc 14:367–371
Jesch NK, Vieten G, Tschernig T, Schroedel W, Ure BM (2005) Mini-laparotomy and full laparotomy, but not laparoscopy, alter hepatic macrophage populations in a rat model. Surg Endosc 19:804–810
Kos M, Kuebler JF, Jesch NK, Vieten G, Bax NM, van der Zee DC, Busche R, Ure BM (2006) Carbon dioxide differentially affects the cytokine release of macrophage subpopulations exclusively via alteration of extracellular pH. Surg Endosc 20:570–576
Takiguchi S, Matsuura N, Hamada Y, Taniguchi E, Sekimoto M, Tsujinaka M, Shiozaki H, Monden M, Ohashi S (2000) Influence of CO2 pneumoperitoneum during laparoscopic surgery on cancer cell growth. Surg Endosc 14:41–44
Smidt VJ, Singh DM, Hurteau JA, Hurd WW (2001) Effect of carbon dioxide on human ovarian carcinoma cell growth. Am J Obstet Gynecol 185:1314–1317
Lee SW, Gleason N, Blanco I, Asi ZK, Whelan RL (2002) Higher colon cancer tumor proliferative index and lower tumor cell death rate in mice undergoing laparotomy versus insufflation. Surg Endosc 16:36–39
Wildbrett P, Oh A, Naundorf D, Volk T, Jacobi CA (2003) Impact of laparoscopic gases on peritoneal microenvironment and essential parameters of cell function. Surg Endosc 17:78–82
Molinas CR, Campo R, Elkelani OA, Binda MM, Carmeliet P, Koninckx PR (2003) Role of hypoxia inducible factors 1alpha and 2alpha in basal adhesion formation and in carbon dioxide pneumoperitoneum-enhanced adhesion formation after laparoscopic surgery in transgenic mice. Fertil Steril 80:795–802
Zhu YP, Feng YJ, Li HM (2003) Adaptation of ovarian cancer cell line SKOV-3ipl cells to hypoxia and its correlation with VEGF and Bcl-2 protein expressions. China Oncol 13:207–210,214
Huang KG, Wang CJ, Chang TC, Liou JD, Hsueh S, Lai CH, Huang LW (2003) Management of port-site metastasis after laparoscopic surgery for ovarian cancer. Am J Obstet Gynecol 189:16–21
Cavina E, Goletti O, Molea N, Buccianti P, Chiarugi M, Boni G, Lazzeri E, Bianchi R (1998) Trocar site tumor recurrences. May pneumoperitoneum be responsible? Surg Endosc 12:1294–1296
Song L, Yin RT (2009) The study of Laparoscopy and port-site metastasis of malignancy. Mod Prev Med 36:1791–1793
Hao YX, Zhong H, Zhang C, Qian F, Rao Y, Yu PW (2008) Effects of different CO2 pressure pneumoperitoneum on the migration and cytoskeleton in gastric cancer cells. Zhonghua Wei Chang Wai Ke Za Zhi 11:454–457
Hu ZH, Peng QL, Li QQ (2004) Effect of hypoxia on invasion and migration of lung carcinoma cells and its molecular basis. Chin J Pathophysiol 20:973–975
Wong K, Rubenthiran U, Jothy S (2003) Motility of colon cancer cells: modulation by CD44 isoform expression. Exp Mol Pathol 75:124–130
Lee TH, Avraham HK, Jiang S, Avraham S (2003) Vascular endothelial growth factor modulates the transendothelial migration of MDA-MB-231 breast cancer cells through regulation of brain microvascular endothelial cell permeability. J Biol Chem 278:5277–5284
Sieuwerts AM, Klijn JG, Foekens JA (1997) Assessment of the invasive potential of human gynecological tumor cell lines with the in vitro Boyden chamber assay: influences of the ability of cells to migrate through the filter membrane. Clin Exp Metastasis 15:53–62
Funding
This study was not supported by any research grants.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Lijun Zheng, Donglei Zhou, Liesheng Lu, Zhongchen Liu, and Lin Fang have no conflicts of interest or financial ties to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zheng, L., Zhou, D., Lu, L. et al. Effects of CO2 pneumoperitoneum on proliferation, apoptosis, and migration of gastrointestinal stromal tumor cells. Surg Endosc 33, 3384–3395 (2019). https://doi.org/10.1007/s00464-018-06633-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-018-06633-6