Abstract
Many elderly people under long-term care suffer from malnutrition caused by dysphagia, frequently leading to sarcopenia. Our hypothesis is that sarcopenia may compromise oral function, resulting in dysphagia. The objectives of this study were to evaluate sarcopenia of the lingual muscles by measuring the tongue thickness, and elucidate its relationship with nutritional status. We examined 104 elderly subjects (mean age = 80.3 ± 7.9 years). Anthropometric data, such as triceps skinfold thickness and midarm muscle area (AMA), were obtained. The tongue thickness of the central part was determined using ultrasonography. Measurement was performed twice and the mean value was obtained. The relationship between tongue thickness and nutritional status was analyzed by Pearson’s correlation coefficient and Spearman’s rank correlation coefficient. AMA and age were identified by multiple-regression analysis as factors influencing tongue thickness. The results of this study suggest that malnutrition may induce sarcopenia not only in the skeletal muscles but also in the tongue.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The tongue plays an important role in feeding and swallowing function. Feinberg et al. [1] reported that bolus misdirection due to dysfunction and abnormality was more frequent at the oral stage alone or at both the oral and pharyngeal stages than at the pharyngeal stage alone. Dysfunction and abnormality of the tongue might also be a reason for dysphagia, since problems at the oral stage are one of the reasons for dysphagia. Many elderly people under long-term care suffer from malnutrition caused by dysphagia and frequently develop sarcopenia because of malnutrition [2]. Sarcopenia is defined as loss of muscular mass, strength, and physical performance. Sarcopenia caused by aging is also affected by the levels of anabolic hormones, which may suppress appetite or lead to a reduction of protein synthesis, resulting in worsening of the condition [3, 4] and subsequent restriction of physical activities in the elderly.
Elderly people frequently suffer from eating malfunction and malnutrition [5, 6]. Fewer occluding pairs of teeth decrease chewing function and increase chewing difficulty [7]. Therefore, chewing ability may contribute to the regulation of nutritional status in the elderly, as reported previously [8]. Subsequently, chewing ability is associated with not only oral health status but also with the physical constitution of the elderly [8]. Low tongue pressure reflects dysphagic tongue movement and cough [9]. Moreover, a decline of oral muscle strength as well as fewer occluding teeth may cause malfunction of feeding; therefore, we presume that malnutrition may worsen in dysphagic patients. Our hypothesis is that sarcopenia may occur in the tongue as well as in other tissues. In other words, we speculated that muscle volume may relate to tongue sarcopenia rather than to body size. If so, sarcopenia of the lingual muscles would compromise oral function in the elderly. Once atrophy of the tongue occurs, people may start to develop malnutrition because of dysphagia. In most cases, the meal texture of these people becomes softer, requiring less power of tongue movement. Consequently, tongue atrophy may be promoted. The objectives of this study were to evaluate sarcopenia of the lingual muscles by measuring the tongue thickness and to elucidate its relationship with nutritional status.
Subjects and Methods
We studied 104 elderly subjects (32 men and 72 women, mean age = 80.3 ± 7.9 years). All maintained occlusal support with either natural dentition or dentures. Neither paralysis nor atrophy of the tongue was observed. The anthropometric data of triceps skinfold thickness (TSF), midarm muscle area (AMA), body weight (BW), and height (HT) were measured to evaluate nutritional status [8, 10].
Anthropometric measurements were conducted as follows: Mid-upper-arm circumference (MAC) was measured on the left arm with a tape measure. TSF was measured with Harpenden Skinfold Calipers over the triceps muscle at the midway point between the acromion and the olecranon process. AMA was calculated from MAC and TSF values based on a previously reported formula [11]. The mean of the twice-repeated measurements was taken as the true value. Tongue thickness was measured using ultrasonography (Nemio 17, SSA-550A, Toshiba Medical Systems, Tokyo, Japan). A fixation device to retain a 3.75-MHz convex probe (contact face size = 12 × 70 mm) in an appropriate position was employed to obtain accurate images, as shown in Fig. 1. To assure stable image acquisition, the probe was firmly fixed to the subject’s lower jaw by wrapping a belt around the head. The subjects were asked to remain seated in an upright position. They were also instructed to swallow their saliva often and to set the tongue at the resting position. Then, ultrasonic measurements were carried out.
The measurement points were determined on the upper and lower surfaces of the lingual muscles in the center of the plane perpendicular to the Frankfurt horizontal plane in a frontal section, as shown in Fig. 2 [12]. This perpendicular plane went through the distal surfaces of the mandibular second premolars on both sides. The measurement point on the coronal plane is shown in Fig. 3. The vertical distance was measured from the surface of the mylohyoid muscle to the tongue dorsum. Figure 4 shows an image of a frontal section of the tongue on ultrasonography. Measurements were performed twice in freeze-frame when the tongue was restored to the resting position after swallowing saliva, and the mean values were obtained. To determine the reliability of the tongue thickness measurement, the two-way mixed-effects model of the intraclass correlation coefficient (ICC) (1,2) was used. The ICC values were above 0.75, indicating good reliability; values of 0.9 and above are reportedly even more reliable for ensuring the validity and reproducibility of clinical measurements [13]. The ICC (1,2) value for the intrarater reliability of tongue thickness measurement was 0.856 (95 % CI: 0.741–0.924).
The relationship between tongue thickness and nutritional status was analyzed using Pearson’s correlation coefficient and Spearman’s rank correlation coefficients using the software SPSS v16 (SPSS, Inc., Chicago, IL).
This study was approved by the Ethics Committee of The Nippon Dental University, School of Life Dentistry at Tokyo, Dental Hospital. Before starting measurements, the purpose and the protocol were explained to the subjects and/or their guardians in order to obtain their consent.
Results
Baseline Characteristics of Subjects
Table 1 gives the baseline characteristics of our subjects. TSF = 11.4 ± 4.6 mm, AMA = 34.9 ± 7.6 cm2, HT = 151.2 ± 8.8 cm, BW = 48.9 ± 8.8 kg, and tongue thickness = 46.9 ± 5.5 mm.
Correlation Coefficients Between Tongue Thickness and Other Variables
Table 2 gives the correlation coefficients between tongue thickness and the other variables examined. Tongue thickness correlated with age (r = − 0.393, P < 0.001), TSF (r = 0.225, P < 0.05), AMA (r = 0.424, P < 0.001), HT (r = 0.312, P < 0.01), and BW (r = 0.434, P < 0.001).
Stepwise Multiple Regression Analysis
Table 3 shows the results of a stepwise multiple regression analysis conducted to identify the factor most strongly influencing tongue thickness. The multiple correlation coefficient (R) was 0.492 and the adjusted coefficient of determination (R 2) was 0.227.
Discussion
Masticatory movement is governed by the coordinated functions of oral organs: teeth, jaw, cheek, lips, and tongue. Among them, the tongue plays an important role in mastication and swallowing since it transports food to the molars, initiates mastication, mixes foods with saliva, and propels a food bolus into the pharynx. Furthermore, the swallowing reflex occurs because the tongue and the soft palate close at the region of the fauces. Many elderly people under long-term care develop malnutrition because of a decline in masticatory and swallowing functions as described above. Improvement in swallowing is considered the most effective way to treat dysphagia because oral dysfunction is also strongly associated with dysphagia [1]. Therefore, evaluating tongue dysfunction or abnormality may be an essential diagnostic procedure for dysphagia. There are many methods for evaluating tongue function, i.e., measuring the strength [14–17] and speed and location of movement [18]. The strength of the tongue has been evaluated by measuring the maximum tongue pressure against the palate [14, 15]. There are some reports that tongue function in the elderly declines with age [14, 15, 19, 20]. However, the effects of malnutrition on tongue volume in the elderly are still unknown. In our study we used ultrasonography to measure tongue thickness. Ultrasonography is widely used for functional analysis of dysphagia and is also reported to be very practical for anatomical analysis [21]. Furthermore, ultrasonography has enormous potential for visualizing the tongue in clinical research because it is noninvasive and it is easy to perform repeated examinations.
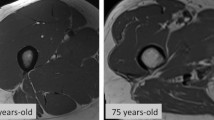
The age-associated loss of both muscle mass and strength, termed sarcopenia, is highly relevant to nursing home residents [22]. It was reported that tongue sarcopenia was observed more frequently in aged rats than in control rats [23, 24]. However, the relationship between tongue sarcopenia and aging in humans is obscure. The absence of occlusal support affects tongue movement and oral function [14, 25, 26]. In this study we employed subjects with posterior occlusal dentition of their natural teeth or dentures to eliminate confounding variables.
It has been suggested that TSF and AMA correlate with nutritional status [8, 10]. TSF represents fat volume and AMA the muscle volume of the upper arm. Since there was a significant association between tongue thickness and nutritional status, tongue muscle volume may also be related to nutritional status.
Furthermore, it was suggested that sarcopenia may develop not only in skeletal muscles but also in the tongue. Hence, dysphagia, tongue disuse syndrome, or malnutrition may affect tongue thickness, with subsequent worsening of malnutrition. Moreover, Saito et al. [27] reported that in rats, the structures of tongue muscles (genioglossus and geniohyoid) may be affected by fat deposition in myofibers. Determination of the fat fraction may be required in our future studies on tongue sarcopenia [28].
It was suspected that tongue thickness correlates with mandibular length. In this regard, an animal study [29] showed the relationship between tongue thickness and mandibular length from infancy through childhood, whereas no such relationship was identified in a human study [30]. However, in the present study we demonstrated a significant relationship between tongue thickness and AMA (an index indicating muscle mass) and age by applying multiple regression analysis. Neither HT, a marker of bone in humans, nor BW (a similar marker) was found to correlate with tongue thickness, suggesting that general muscle volume and/or age alone may affect this feature of the tongue.
Atrophy of the tongue may not be the only reason for reduced tongue function and inability to maintain nutritional status. However, Kikutani et al. [31] reported that oral functional training to maintain and/or improve feeding function is very efficient for improving the nutritional condition. It was reported that muscle is replaced by fat or fibrous tissues with aging [32], implying that tongue exercise might restore muscle tissue. Robbins et al. [32] and Yeates et al. [33] also reported that exercising the tongue prevented general sarcopenia. Therefore, effective measures or protocols to prevent malnutrition, which involve tongue exercise or rehabilitation, may be necessary to improve tongue disuse syndrome. For this purpose, our method of monitoring tongue thickness by ultrasonography may provide information for a tongue exercise protocol or treatment plan. We will study further the relationship between tongue pressure and tongue thickness in a future investigation.
Conclusion
The findings of this study suggest that tongue thickness is related to nutritional status in the elderly.
References
Feinberg MJ, Ekberg O. Videofluoroscopy in elderly patients with aspiration: importance of evaluating both oral and pharyngeal stages of deglutition. AJR Am J Roentgenol. 1991;156:293–6.
Rosenberg IH, Roubenoff R. Stalking sarcopenia. Ann Intern Med. 1995;123:727–8.
Greenlund LJ, Nair KS. Sarcopenia—consequences, mechanisms, and potential therapies. Mech Ageing Dev. 2003;124:287–99.
Roubenoff R. Sarcopenia: effects on body composition and function. J Gerontol A Biol Sci Med Sci. 2003;58:1012–7.
Ney DM, Weiss JM, Kind AJ, Robbins J. Senescent swallowing: impact, strategies, and interventions. Nutr Clin Pract. 2009;24:395–413.
White GN, O’Rourke F, Ong BS, Cordato DJ, Chan DK. Dysphagia: causes, assessment, treatment, and management. Geriatrics. 2008;63:15–20.
Leake JL. An index of chewing ability. J Public Health Dent. 1990;50:262–7.
Okada K, Enoki H, Izawa S, Iguchi A, Kuzuya M. Association between masticatory performance and anthropometric measurements and nutritional status in the elderly. Geriatr Gerontol Int. 2010;10:56–63.
Yoshida M, Kikutani T, Tsuga K, Utanohara Y, Hayashi R, Akagawa Y. Decreased tongue pressure reflects symptom of dysphagia. Dysphagia. 2006;21:61–5.
Chazot C, Laurent G, Charra B, Blanc C, VoVan C, Jean G, et al. Malnutrition in long-term haemodialysis survivors. Nephrol Dial Transpl. 2001;16:61–9.
Heymsfield SB, McManus C, Smith J, Stevens V, Nixon DW. Anthropometric measurement of muscle mass: revised equations for calculating bone-free arm muscle area. Am J Clin Nutr. 1982;36:680–90.
Okayama H, Tamura F, Kikutani T, Kayanaka H, Katagiri H, Nishiwaki K. Effects of a palatal augmentation prosthesis on lingual function in postoperative patients with oral cancer: coronal section analysis by ultrasonography. Odontology. 2008;96:26–31.
Portney LG, Watkins MP. Foundations of clinical research: applications to practice. Englewood Cliffs: Prentice-Hall; 2000.
Kikutani T, Tamura F, Nishiwaki K, Kodama M, Suda M, Fukui T, et al. Oral motor function and masticatory performance in the community-dwelling elderly. Odontology. 2009;97:38–42.
Hayashi R, Tsuga K, Hosokawa R, Yoshida M, Sato Y, Akagawa Y. A novel handy probe for tongue pressure measurement. Int J Prosthodont. 2002;15:385–8.
Ziegler W. Task-related factors in oral motor control: speech and oral diadochokinesis in dysarthria and apraxia of speech. Brain Lang. 2002;80:556–75.
Utanohara Y, Hayashi R, Yoshikawa M, Yoshida M, Tsuga K, Akagawa Y. Standard values of maximum tongue pressure taken using newly developed disposable tongue pressure measurement device. Dysphagia. 2008;23:286–90.
Klawe JJ, Tafil-Klawe M. Age-related response of the genioglossus muscle EMG-activity to hypoxia in humans. J Physiol Pharmacol. 2003;54(Suppl 1):14–9.
Peng CL, Miethke RR, Pong SJ, Lin CT. Investigation of tongue movements during swallowing with M-mode ultrasonography. J Orofac Orthop. 2007;68:17–25.
Ardakani FE. Evaluation of swallowing patterns of the tongue using real-time B-mode sonography. J Contemp Dent Pract. 2006;7:67–74.
Ajaj W, Goyen M, Herrmann B, Massing S, Goehde S, Lauenstein T, et al. Measuring tongue volumes and visualizing the chewing and swallowing process using real-time TrueFISP imaging—initial clinical experience in healthy volunteers and patients with acromegaly. Eur Radiol. 2005;15:913–8.
Bauer J, Kaiser M, Sieber CC. Sarcopenia in nursing home residents. J Am Med Dir Assoc. 2008;9:545–51.
Ota F, Connor NP, Konopacki R. Alterations in contractile properties of tongue muscles in old rats. Ann Otol Rhinol Laryngol. 2005;114:799–803.
Schwarz EC, Thompson JM, Connor NP, Behan M. The effects of aging on hypoglossal motoneurons in rats. Dysphagia. 2009;24:40–8.
Tamura F, Suzuki S. Effects of edentulism on lingual functions during swallowing. J Disabil Oral Health. 2004;5:83–7.
Tamura F, Mizukami M, Ayano R, Mukai Y. Analysis of feeding function and jaw stability in bedridden elderly. Dysphagia. 2002;17:235–41.
Saito T, Yamane A, Kaneko S, Ogawa T, Ikawa T, Saito K, et al. Changes in the lingual muscles of obese rats induced by high-fat diet feeding. Arch Oral Biol. 2010;55:803–8.
Humbert IA, Reeder SB, Porcaro EJ, Kays SA, Brittain JH, Robbins J. Simultaneous estimation of tongue volume and fat fraction using IDEAL-FSE. J Magn Reson Imaging. 2008;28:504–8.
Liu ZJ, Shcherbatyy V, Gu G, Perkins JA. Effects of tongue volume reduction on craniofacial growth: a longitudinal study on orofacial skeletons and dental arches. Arch Oral Biol. 2008;53:991–1001.
Siebert JR. A morphometric study of normal and abnormal fetal to childhood tongue size. Arch Oral Biol. 1985;30:433–40.
Kikutani T, Enomoto R, Tamura F, Oyaizu K, Suzuki A, Inaba S. Effects of oral functional training for nutritional improvement in Japanese older people requiring long-term care. Gerodontology. 2006;23:93–8.
Robbins J, Gangnon RE, Theis SM, Kays SA, Hewitt AL, Hind JA. The effects of lingual exercise on swallowing in older adults. J Am Geriatr Soc. 2005;53:1483–9.
Yeates EM, Molfenter SM, Steele CM. Improvements in tongue strength and pressure-generation precision following a tongue-pressure training protocol in older individuals with dysphagia: three case reports. Clin Interv Aging. 2008;3:735–47.
Acknowledgments
The authors thank Drs. Tetsuo Hanagata and Akira Ozawa at Yamanashi Dental Association, Drs. Yasumasa Akagawa and Kazuhiro Tsuga at Hiroshima University, and Dr. Misaka Kimura at Kyoto Prefectural University of Medicine for helpful discussions. This study was supported in part by a Research Grant for Longevity Science (H19-2) from of the Ministry of Health, Labour and Welfare, Japan.
Conflict of interest
The authors have no conflicts of interest to declare.
Open Access
This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Tamura, F., Kikutani, T., Tohara, T. et al. Tongue Thickness Relates to Nutritional Status in the Elderly. Dysphagia 27, 556–561 (2012). https://doi.org/10.1007/s00455-012-9407-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00455-012-9407-z