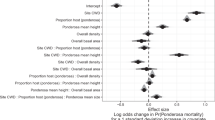
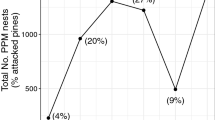
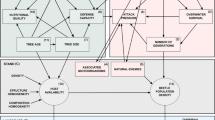
Abstract
Irruptive forest insects such as bark beetles undergo intermittent outbreaks that cause landscape-scale tree mortality. Despite their enormous economic and ecological impacts, we still have only limited understanding of the dynamics by which populations transition from normally stable endemic to irruptive densities. We investigated density-dependent changes in mountain pine beetle reliance on stressed hosts, host selection, spatial configuration of attacks, and the interaction of host selection and spatial configuration by performing a complete census of lodgepole pine across six stands and 6 years. In addition, we compared the dynamics of mountain pine beetle with those of other bark beetles. We found that as population size increased, reliance on stressed trees decreased and new attacks shifted to larger trees with thicker phloem and higher growth rates that can support higher offspring production. Moreover, the spatial configuration of beetle-attacked trees shifted from random to spatially aggregated. Further, we found evidence that beetle utilization of larger trees was related to aggregation behavior as the size of tree attacked was positively correlated at 10–25 m, within the effective distance of pheromone-mediated signaling. In contrast, non-irruptive bark beetle species did not exhibit such density-dependent spatial aggregation at the stand scale or switches in host selection behavior. These results identify how density-dependent linkages between spatial configuration and host utilization can converge to drive population transitions from endemic to irruptive phases. Specifically, a combination of stand-level spatial aggregation, behavioral shifts, and higher quality of attainable hosts defines a critical threshold beyond which continual population growth becomes self-driving.








Similar content being viewed by others
References
Allen CD, Breshears DD, McDowell NG (2015) On underestimation of global vulnerability to tree mortality and forest die-off from hotter drought in the Anthropocene. Ecosphere 6:art129. https://doi.org/10.1890/ES15-00203.1
Amman GD (1969) Mountain pine beetle emergence in relation to depth of lodgepole pine bark. US Department of Agriculture, Forest Service, Intermountain Forest and Range Experiment Station, Ogden, UT, USA
Amman GD, Cole WE (1983) Mountain pine beetle dynamics in lodgepole pine forests. US department of agriculture, forest service, intermountain forest and range experiment station
Anderegg WRL, Hicke JA, Fisher RA et al (2015) Tree mortality from drought, insects, and their interactions in a changing climate. New Phytol 208:674–683. https://doi.org/10.1111/nph.13477
Aukema BH, Carroll AL, Zheng Y et al (2008) Movement of outbreak populations of mountain pine beetle: influences of spatiotemporal patterns and climate. Ecography 31:348–358. https://doi.org/10.1111/j.0906-7590.2007.05453.x
Aukema BH, Carroll AL, Zhu J et al (2006) Landscape level analysis of mountain pine beetle in British Columbia, Canada: spatiotemporal development and spatial synchrony within the present outbreak. Ecography 29:427–441. https://doi.org/10.1111/j.2006.0906-7590.04445.x
Aukema BH, Zhu J, Møller J, Rasmussen JG, Raffa KF (2010) Predisposition to bark beetle attack by root herbivores and associated pathogens: roles in forest decline gap formation and persistence of endemic bark beetle populations. Forest Ecol and Manage 259(3):374–382. https://doi.org/10.1016/j.foreco.2009.10.032
Avery TE, Burkhart HE (2002) Forest Measurements, 5th ed. McGraw-Hill, New York, USA
Ayres MP, Lombardero MJ (2000) Assessing the consequences of global change for forest disturbance from herbivores and pathogens. Sci Total Environ 262:263–286. https://doi.org/10.1016/S0048-9697(00)00528-3
Baddeley RA, Rubak E, Turner R (2015) Spatial Point Patterns: Methodology and applications with R. Chapman and hall/CRC press. Boca Raton FL, USA
Berryman AA (1987) The theory and classification of outbreaks. Insect outbreaks. Elsevier, pp 3–30
Berryman AA (1996) What causes population cycles of forest Lepidoptera? Trends Ecol Evol 11:28–32. https://doi.org/10.1016/0169-5347(96)81066-4
Beudert B, Bässler C, Thorn S, Noss R, Schröder B, Dieffenbach-Fries H, Foullois N, Müller J (2015) Bark beetles increase biodiversity while maintaining drinking water quality. Conserv Lett 8(4):272–281. https://doi.org/10.1111/conl.12153
Biedermann PHW, Müller J, Grégoire J-CC et al (2019) Bark beetle population dynamics in the Anthropocene: challenges and solutions. Trends Ecol Evol. https://doi.org/10.1016/j.tree.2019.06.002
Bjornstad ON, Grenfell BT (2001) Noisy clockwork: time series analysis of population fluctuations in animals. Science 293:638–643. https://doi.org/10.1126/science.1062226
Bleiker KP, O’Brien MR, Smith GD, Carroll AL (2014) Characterisation of attacks made by the mountain pine beetle (Coleoptera: Curculionidae) during its endemic population phase. Can Entomol 146:271–284. https://doi.org/10.4039/tce.2013.71
Blomquist GJ, Figueroa-Teran R, Aw M et al (2010) Pheromone production in bark beetles. Insect Biochem Mol Biol 40:699–712. https://doi.org/10.1016/j.ibmb.2010.07.013
Boone CK, Aukema BH, Bohlmann J et al (2011) Efficacy of tree defense physiology varies with bark beetle population density: a basis for positive feedback in eruptive species. Can J for Res 41:1174–1188. https://doi.org/10.1139/x11-041
Burke JL, Carroll AL (2017) Breeding matters: natal experience influences population State-dependent host acceptance by an eruptive insect herbivore. PLoS ONE 12:1–16. https://doi.org/10.1371/journal.pone.0172448
Carroll A, Aukema B, Raffa K et al (2006) Mountain pine beetle outbreak development: the endemic—incipient epidemic transition. Working Paper, MPBI, Natural Resources Canada, Canadian Forest Service, Victoria, Canada PO # 1.03:21
Chang W-Y, Lantz VA, Hennigar CR, MacLean DA (2012) Benefit-cost analysis of spruce budworm (Choristoneura fumiferana Clem.) control: incorporating market and non-market values. J Environ Manag 93:104–112. https://doi.org/10.1016/j.jenvman.2011.08.022
Clark PJ, Evans FC (1954) Distance to nearest neighbor as a measure of spatial relationships in populations. Ecol 35:445–453
Cobbold CA, Roland J, Lewis MA (2009) The impact of parasitoid emergence time on host-parasitoid population dynamics. Theor Popul Biol 75:201–215. https://doi.org/10.1016/j.tpb.2009.02.004
Cooke BJ, Carroll AL (2017) Predicting the risk of mountain pine beetle spread to eastern pine forests: considering uncertainty in uncertain times. For Ecol Manag 396:11–25. https://doi.org/10.1016/j.foreco.2017.04.008
Coulson RN, Hennier PB, Flamm RO, Rykiel EJ, Hu LC, Payne TL (1983) The role of lightning in the epidemiology of the Southern Pine Beetle1. Zeitschrift für Angewandte Entomologie 96(1–5):182–193. https://doi.org/10.1111/j.1439-0418.1983.tb03659.x
Diggle PJ (2013) Statistical analysis of spatial and spatio-temporal point patterns. Chapman and Hall, New York, USA
Dwyer G, Dushoff J, Yee SH (2004) The combined effects of pathogens and predators on insect outbreaks. Nature 430:341–345. https://doi.org/10.1038/nature02569
Gara RI, Coster JE (1968) Studies on the attack behavior of the southern pine beetle. III. Sequence of tree infestation within stands. Contrib Boyce Thompson Inst 24:77–86
Geiszler DR, Gara RI, Driver CH, Galucci VF, Martin RE (1980) Fire, fungi and beetle influences on a lodgepole pine ecosystem. Oecologia 46:239–243
Geiszler DR, Gara RI, Lidke WR (1984) Bark beetle infestations of lodgepole pine following fire in south-central Oregon. Zeitschrift für Angewandte Entomologie 98:389–394
Geiszler DR, Gara RI (1978) Mountain pine beetle attack dynamics in lodgepole pine. Theory and practice of mountain pine beetle management in lodgepole pine forests: symposium proceedings. University of Idaho, Forest, Wildlife and Range Experiment Station, Moscow, USA, pp 182–187
Gitau CW, Bashford R, Carnegie AJ, Gurr GM (2013) A review of semiochemicals associated with bark beetle (Coleoptera: Curculionidae: Scolytinae) pests of coniferous trees: a focus on beetle interactions with other pests and their associates. For Ecol Manag 297:1–14. https://doi.org/10.1016/j.foreco.2013.02.019
Graf M, Reid ML, Aukema BH, Lindgren BS (2012) Association of tree diameter with body size and lipid content of mountain pine beetles. The Can Entomol 144(3):467–477. https://doi.org/10.4039/tce.2012.38
Gregoire JC (1985) Host colonization strategies in dendroctonus: larval gregariousness vs. mass attack by adults? The role of the host in the population dynamics of forest insects. Victoria, BC, Canada, pp 147–154
Hadley KS, Veblen TT (1993) Stand response to western spruce budworm and douglas-fir bark beetle outbreaks Colorado front range. Can J Res 23(3):479–491. https://doi.org/10.1139/x93-066
Hart SJ, Veblen TT, Schneider D, Molotch NP (2017) Summer and winter drought drive the initiation and spread of spruce beetle outbreak. Ecology 98:2698–2707. https://doi.org/10.1002/ecy.1963
Hicke JA, Allen CD, Desai AR et al (2012) Effects of biotic disturbances on forest carbon cycling in the United States and Canada. Glob Change Biol 18:7–34. https://doi.org/10.1111/j.1365-2486.2011.02543.x
Hicke JA, Meddens AJH, Kolden CA (2016) Recent tree mortality in the western United States from bark beetles and forest fires. For Sci 62:141–153. https://doi.org/10.5849/forsci.15-086
Howe M, Mason CJ, Gratton C, Keefover–Ring K, Wallin K, Yanchuk A, Zhu J, Raffa KF (2020) Relationships between conifer constitutive and inducible defenses against bark beetles change across levels of biological and ecological scale. Oikos 129(7):1093–1107. https://doi.org/10.1111/oik.07242
Jepsen JU, Hagen SB, Ims RA, Yoccoz NG (2008) Climate change and outbreaks of the geometrids Operophtera brumata and Epirrita autumnata in subarctic birch forest: evidence of a recent outbreak range expansion. J Anim Ecol 77:257–264. https://doi.org/10.1111/j.1365-2656.2007.0
Kassambara A, Mundt F (2020) Factoextra: extract and visualize the results of multivariate data analyses. R Package Version 1.0.7
Kausrud K, Økland B, Skarpaas O et al (2012) Population dynamics in changing environments: the case of an eruptive forest pest species. Biol Rev 87:34–51. https://doi.org/10.1111/j.1469-185X.2011.00183.x
Kendall BE, Briggs CJ, Murdoch WW et al (1999) Why do populations cycle? A synthesis of statistical and mechanistic modeling approaches. Ecology 80:1789–1805. https://doi.org/10.1890/0012-9658(1999)080[1789:WDPCAS]2.0.CO;2
Klutsch JG, Beam RD, Jacobi WR, Negrón JF (2014) Bark beetles and dwarf mistletoe interact to alter downed woody material, canopy structure, and stand characteristics in northern Colorado ponderosa pine. For Ecol Manag 315:63–71. https://doi.org/10.1016/j.foreco.2013.12.024
Kolb TE (2016) Observed and anticipated impacts of drought on forest insects and diseases in the United States. For Ecol Manag 14:321–334
Koopmans JM (2011) Spatial and temporal analyses of bark beetle population dynamics in southern British Columbia: stand-level studies of the bole-infesting assemblage during eruptive transitions of mountain pine beetle, Dendroctonus ponderosae Hopkins. MSc Thesis, University of Northern British Columbia, Prince George, Canada
Kurz WA, Dymond CC, Stinson G et al (2008) Mountain pine beetle and forest carbon feedback to climate change. Nature 452:987–990. https://doi.org/10.1038/nature06777
Larsson S, Ekbom B, Bjorkman C (2000) Influence of plant quality on pine sawfly population dynamics. Oikos 89:440–450
Lavender D P (1991) Reforestation in British Columbia. In:Proceedings of the International Workshop on Large-Scale Reforestation. US Environmental Protection Agency, Office of Research and Development, pp 29–39
Liebhold AM, Tobin PC (2008) Population ecology of insect invasions and their management. Annu Rev Entomol. https://doi.org/10.1146/annurev.ento.52.110405.091401
Lindgren BS, Raffa KF (2013) Evolution of tree killing in bark beetles (Coleoptera: Curculionidae): trade-offs between the maddening crowds and a sticky situation. Can Entomol 145:471–495. https://doi.org/10.4039/tce.2013.27
Logan JA, Régnière J, Powell JA (2003) Assessing the impacts of global warming on forest pest dynamics. Front Ecol Environ 1:130–137. https://doi.org/10.1890/1540-9295(2003)001[0130:ATIOGW]2.0.CO;2
Logan JA, White P, Bentz BJ, Powell JA (1998) Model analysis of spatial patterns in mountain pine beetle outbreaks. Theor Popul Biol 53:236–255. https://doi.org/10.1006/tpbi.1997.1350
Ludecke D (2018) ggeffects: Tidy data frames of marginal effects from regression models. J Open Source Softw 3(26):772. https://doi.org/10.21105/joss.00772
Marini L, Ayres MP, Battisti A, Faccoli M (2012) Climate affects severity and altitudinal distribution of outbreaks in an eruptive bark beetle. Clim Change 115:327–341. https://doi.org/10.1007/s10584-012-0463-z
Marini L, Økland B, Jönsson AM et al (2017) Climate drivers of bark beetle outbreak dynamics in Norway spruce forests. Ecography 40:1426–1435. https://doi.org/10.1111/ecog.02769
Martinson SJ, Ylioja T, Sullivan B, Billings RF, Ayres MP (2013) Alternate attractors in the population dynamics of a tree-killing bark beetle. Popul Ecol 55(1):95–106. https://doi.org/10.1007/s10144-012-0357-y
Matasci G, Hermosilla T, Wulder MA, White JC, Coops NC, Hobart GW, Bolton DK, Tompalski P, Bater CW (2018) Three decades of forest structural dynamics over Canada’s forested ecosystems using Landsat time-series and lidar plots. Remote Sens Environ 216:697–714. https://doi.org/10.1016/j.rse.2018.07.024
McDowell NG, Allen CD, Anderson-Teixeira K et al (2020) Pervasive shifts in forest dynamics in a changing world. Science 368:eaaz9463. https://doi.org/10.1126/science.aaz9463
Meddens AJH, Hicke JA, Ferguson CA (2012) Spatiotemporal patterns of observed bark beetle-caused tree mortality in British Columbia and the western United States. Ecol Appl 22(7):1876–1891. https://doi.org/10.1890/11-1785.1
Mitchell RG, Preisler HK (1991) Analysis of spatial patterns of lodgepole pine attacked by outbreak populations of the mountain pine beetle. Forest Sci 37:1390–1408
Nebeker TE, Tisdale RA, Schmitz RF, Hobson KR (1995) Chemical and nutritional status of dwarf mistletoe, Armillaria root rot, and Comandra blister rust infected trees which may influence tree susceptibility to bark beetle attack. Can J Bot 73:360–369. https://doi.org/10.1139/b95-037
Negrón J, Lynch A, Willis, Mercado SJ (2014) Douglas-fir tussock moth- and douglas-fir beetle-caused mortality in a ponderosa pine/douglas-fir forest in the Colorado front range USA. Forest 5(12):3131–3146. https://doi.org/10.3390/f5123131
Økland B, Berryman A (2004) Resource dynamic plays a key role in regional fluctuations of the spruce bark beetles Ips typographus. Agric for Entomol 6:141–146. https://doi.org/10.1111/j.1461-9555.2004.00214.x
Økland B, Bjørnstad ON (2006) A resource-depletion model of forest insect outbreaks. Ecology 87:283–290. https://doi.org/10.1890/05-0135
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O'Hara RB, Simpson GL, Solymos P, Henry M, Stevens H, Szoecs E, Helene W (2020) Vegan: Community ecology package. R Package Version 2.5-7
Pebesma E (2018) Simple features for R: standardized support for spatial vector data. The R J 10(1):439. https://doi.org/10.32614/RJ-2018-009
Peltonen M, Liebhold AM, Bjørnstad ON, Williams DW (2002) Spatial synchrony in forest insect outbreaks: roles of regional stochasticity and dispersal. Ecology 83:3120–3129. https://doi.org/10.1890/0012-9658(2002)083[3120:SSIFIO]2.0.CO;2
Powell EN, Townsend PA, Raffa KF (2012) Wildfire provides refuge from local extinction but is an unlikely driver of outbreaks by mountain pine beetle. Ecol Monogr 82:69–84. https://doi.org/10.1890/11-0607.1
Powers JS, Sollins P, Harmon ME, Jones JA (1999) Plant-pest interactions in time and space: a Douglas-fir bark beetle outbreak as a case study. Lands Ecol 14:105–120. https://doi.org/10.1023/A:1008017711917
Preisler HK, Hicke JA, Ager AA, Hayes JL (2012) Climate and weather influences on spatial temporal patterns of mountain pine beetle populations in Washington and Oregon. Ecology 93:2421–2434. https://doi.org/10.1890/11-1412.1
Preisler HK, Mitchell RG (1993) Colonization patterns of the mountain pine beetle in thinned and unthinned lodgepole pine stands. Forest Sci 39:528–545
Raffa KF, Berryman AA (1983) The Role of host plant resistance in the colonization behavior and ecology of bark beetles (coleoptera: scolytidae). Ecol Monogr 53(1):27–49. https://doi.org/10.3390/f9090552
Raffa KF, Aukema BH, Erbilgin N et al (2005) Interactions among conifer terpenoids and bark beetles across multiple levels of scale: an attempt to understand links between population patterns and physiological processes. In: Romeo JT (ed) Chemical ecology and phytochemistry of forest ecosystems. Elsevier, pp 79–118
Raffa KF, Aukema BH, Bentz BJ et al (2008) Cross-scale drivers of natural disturbances prone to anthropogenic amplification: the dynamics of bark beetle eruptions. Bioscience 58:501–517. https://doi.org/10.1641/B580607
Rankin LJ, Borden JH (1991) Competitive interactions between the mountain pine beetle and the pine engraver in lodgepole pine. Can J for Res 21:1029–1036. https://doi.org/10.1139/x91-141
Roffey J, Popov G (1968) Environmental and behavioural processes in a desert locust outbreak. Nature 219:446–450. https://doi.org/10.1038/219446a0
Royama T (1992) Analytical population dynamics. Springer, Netherlands, Dordrecht
Safranyik L, Carroll AL (2006) The biology and epidemiology of the mountain pine beetle in lodgepole pine forests. The mountain pine beetle: a synthesis of its biology, management and impacts on lodgepole pine. Natural Resources Canada Candian Forest Service, Victoria, pp 3–66
Safranyik L, Carroll AL, Regniere J et al (2010) Potential for range expansion of mountain pine beetle into the boreal forest of North America. Can Entomol 142:415–442. https://doi.org/10.4039/n08-CPA01
Safranyik L, Shore TL, Linton DA, Rankin LJ (eds) (1999) Effects of induced competitive interactions with secondary bark beetle species on the establishment and survival of mountain pine beetle broods in lodgepole pine. Canadian Forest Service Pacific Forestry Centre, Victoria
Schaupp WC, Costello SL, Ciesla WM (2008) The history of Douglas-fir tussock moth in Colorado and Wyoming. US Department of Agriculture, Forest Service, Rocky Mountain Region, Lakewood, CO, USA
Schlather M, Ribeiro PJ Jr, Diggle PJ (2004) Detecting dependence between marks and locations of marked point processes. J R Stat Soc: Ser B (Stat Methodol) 66(1):79–93. https://doi.org/10.1046/j.1369-7412.2003.05343.x
Seidl R, Müller J, Hothorn T et al (2016) Small beetle, large-scale drivers: how regional and landscape factors affect outbreaks of the European spruce bark beetle. J Appl Ecol 53:530–540. https://doi.org/10.1111/1365-2664.12540
Seidl R, Schelhaas M-J, Rammer W, Verkerk PJ (2015) Increasing forest disturbances in Europe and their impact on carbon storage 12.
Seidl R, Thom D, Kautz M et al (2017) Forest disturbances under climate change. Nat Clim Change 7:395–402. https://doi.org/10.1038/nclimate3303
Smith GD, Carroll AL, Lindgren BS (2011) Facilitation in bark beetles: endemic mountain pine beetle gets a helping hand. Agric for Entomol 13:37–43. https://doi.org/10.1111/j.1461-9563.2010.00499.x
Sommerfeld A, Senf C, Buma B et al (2018) Patterns and drivers of recent disturbances across the temperate forest biome. Nat Commun. https://doi.org/10.1038/s41467-018-06788-9
Stephenson NL, Das AJ, Ampersee NJ, Bulaon BM, Yee JL, Edwards D (2019) Which trees die during drought? The key role of insect host‐tree selection. J Ecol 107(5):2383–2401. https://doi.org/10.1111/1365-2745.13176
Thom D, Rammer W, Seidl R (2017) Disturbances catalyze the adaptation of forest ecosystems to changing climate conditions. Glob Change Biol 23(1):269–282. https://doi.org/10.1111/gcb.13506
Trubin A, Mezei P, Zabihi K et al (2022) Northernmost European spruce bark beetle Ips typographus outbreak: modelling tree mortality using remote sensing and climate data. For Ecol Manag 505:119829. https://doi.org/10.1016/j.foreco.2021.119829
Turchin P (1999) Dynamical role of predators in population cycles of a forest insect: an experimental test. Science 285:1068–1071. https://doi.org/10.1126/science.285.5430.1068
Turner MG, Tinker DB, Romme WH, Kashian DM, Litton CM (2004) Landscape patterns of sapling density leaf area and aboveground net primary production in postfire lodgepole pine forests yellowstone national park (USA). Ecosystems 7(7):751–775. https://doi.org/10.1007/s10021-004-0011-4
Wallin KF, Raffa KF (2004) Feedback between individual host selection behavior and population dynamics in an eruptive herbivore. In: Kimberly F, Wallin, Kenneth F (eds) Raffa Published by : Wiley on behalf of the Ecological Society of America Stable URL : https://www.jstor. Ecological Monographs 74, pp 101–116
Ward SF, Aukema BH (2019) Anomalous outbreaks of an invasive defoliator and native bark beetle facilitated by warm temperatures changes in precipitation and interspecific interactions. Ecography 42(5):1068–1078. https://doi.org/10.1111/ecog.04239
Waring RH, Pitman GB (1985) Modifying lodgepole pine stands to change susceptibility to mountain pine beetle attack. Ecol 66:889–897
Weed AS, Ayres MP, Hicke JA (2013) Consequences of climate change for biotic disturbances in North American forests. Ecol Monogr 83:441–470
Wickham H, Averick M, Bryan J, Chang W, McGowan L, Francois R, Grolemund G, Hayes A, Henry L, Hester J, Kuhn M (2019) Welcome to the Tidyverse. J Open Source Softw 4:1686
Williams DW, Liebhold AM (2006) Herbivorous insects and global change: potential changes in the spatial distribution of forest defoliator outbreaks. J Biogeogr 22:665. https://doi.org/10.2307/2845968
Williams H, Hood S, Keyes CR, Egan JM, Negron J(2018) Subwatershed-Level lodgepole pine attributes associated with a mountain pine beetle outbreak. Forest 9(9):552. https://doi.org/10.3390/f9090552
Yan Z, Sun J, Don O, Zhang Z (2005) The red turpentine beetle, Dendroctonus valens LeConte (Scolytidae): an exotic invasive pest of pine in China. Biodivers & Conserv 14:1735–1760
Acknowledgements
This investigation comprised a very large and complex multi-year field study that would not have been possible without invaluable contributions from Douglas Linton, Tony Ibaraki, Greg Smith, Andrew Copeland, Dion Manastyrski and Fred Davis. Funding was generously provided to ALC by Natural Resources Canada—Mountain Pine Beetle Initiative. Further support was provided by the University of Wisconsin–Madison College of Agricultural and Life Sciences, Graduate School and Vilas-Sorenson Professorship. We thank Anthony R. Ives, (UW-Madison) and Guillherme Ludwig (University of Campinas) for helpful conversations on analyzing point patterns.
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
AC conceived, designed, and performed the experiment. MH analyzed the data. MH, KR, BA, CG and AC wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
We declare no conflicts of interest.
Additional information
Communicated by Amy Austin.
Supplementary Information
Below is the link to the electronic supplementary material.
442_2022_5129_MOESM1_ESM.png
Figure 1: Screeplots for principal components analysis, depicting A) the variability explained by each dimension; and the factors contributing to the B) 1st, C) 2nd, and D) 3rd dimensions (PNG 715 KB)
442_2022_5129_MOESM2_ESM.png
Figure 2: Walkthrough of hypothesis testing process given a small (200 x 200 m) window from stand A in 2005. The example tests whether the distribution of trees attacked by mountain pine beetle is more (aggregation) or less (inhibition) spatially correlated than randomly distributed points (Poisson) based on the underlying joint density of beetle attacks. Point color denotes bark beetle guild for threshold limited (i.e., mountain pine beetle; black) and lower-stem (yellow) beetles. Gradient color denotes intensity of the joint density and is depicted on different scales for each null hypothesis. Scales are not shown because the relative intensity is more important than the realized values (PNG 470 KB)
442_2022_5129_MOESM3_ESM.png
Figure 3: Alternative hypothesis testing for the spatial configuration of attacked trees based on A) a nearest-neighbor approach (Clark Evan’s Aggregation Index) and B) how our interpretations of observed L(r)-r functions compares. Colors denote bark beetle guild. Shapes in A correspond to significance assessed at the α=0.01 level where filled circles denote significant and open circles denote not significant. Shapes in B correspond to our interpretation of the observed L(r)-r functions, where filled circles denote significant aggregation, circles with crosses denote weak significant aggregation (i.e., the observed L(r)-r function barely exceeded the significance bands), open circles denote no significant aggregation, and X’s denote stand/year/guild combinations where there were fewer than 5 attacked trees. (PNG 186 KB)
442_2022_5129_MOESM4_ESM.docx
Supplemental Table 1: Criteria used to estimate the number of years since initial complete (A) or partial (B) attacks by bole-infesting bark beetles on lodgepole pine trees. Adopted from Carroll et al. (2006) (DOCX 20 KB)
442_2022_5129_MOESM5_ESM.docx
Supplemental Table 2: Distribution of prior stressors A) in each stand, and B) the number of trees with at least 0, 1, 2, 3, or 4 stressors (DOCX 17 KB)
442_2022_5129_MOESM6_ESM.docx
Supplemental Table 3: Observed L(r)-r functions for each guild/stand/year combination. Patterns not shown contained less than five attacked trees. Panels for each point pattern correspond to different null hypotheses of the underlying spatial distribution of available host trees. We provide our interpretation of whether each pattern exhibits significant aggregation or inhibition in the right-hand columns. (DOCX 6076 KB)
Rights and permissions
About this article
Cite this article
Howe, M., Raffa, K.F., Aukema, B.H. et al. Numbers matter: how irruptive bark beetles initiate transition to self-sustaining behavior during landscape-altering outbreaks. Oecologia 198, 681–698 (2022). https://doi.org/10.1007/s00442-022-05129-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-022-05129-4