Abstract
Low temperature in winter depresses rates of photosynthesis, which, in evergreen plants, can exacerbate imbalances between light absorption and photochemical light use. Damage that could result from increased excess light absorption is minimized by the conversion of excitation energy to heat in a process known as energy dissipation, which involves the de-epoxidized carotenoids of the xanthophyll cycle. Overwintering evergreens employ sustained forms of energy dissipation observable even after lengthy periods of dark acclimation. Whereas most studies of photoprotective energy dissipation examine one or a small number of species; here, we measured the levels of sustained thermal energy dissipation of seventy conifer taxa growing outdoors under common-garden conditions at the Red Butte Garden in Salt Lake City, Utah, U.S.A. (forty nine taxa were also sampled for needle pigment content). We observed an extremely wide range of wintertime engagement of sustained energy dissipation; the percentage decrease in dark-acclimated photosystem II quantum efficiency from summer to winter ranged from 6 to 95%. Of the many pigment-based parameters measured, the magnitude of the seasonal decrease in quantum efficiency was most closely associated with the seasonal increase in zeaxanthin content expressed on a total chlorophyll basis, which explained only slightly more than one-third of the variation. We did not find evidence for a consistent wintertime decrease in needle chlorophyll content. Thus, the prevailing mechanism for winter decreases in solar-induced fluorescence emitted by evergreen forests may be decreases in fluorescence quantum yield, and wintertime deployment of sustained energy dissipation likely underlies this effect.




Similar content being viewed by others
Data availability
Study data available via the Bowdoin Digital Commons.
References
Adams WW, Demmig-Adams B, Verhoeven AS, Barker DH (1995) ‘Photoinhibition’’ during winter stress: involvement of sustained xanthophyll cycle-dependent energy dissipation.’ Funct Plant Biol 22:261–276. https://doi.org/10.1071/PP9950261
Adams WW, Zarter CR, Ebbert V, Demmig-Adams B (2004) Photoprotective strategies of overwintering evergreens. AIBS Bull 54:41–49. https://doi.org/10.1641/0006-3568(2004)054[0041:PSOOE]2.0.CO;2
Alvarez-Uria P, Körner C (2007) Low temperature limits of root growth in deciduous and evergreen temperate tree species. Funct Ecol 21:211–218. https://doi.org/10.1111/j.1365-2435.2007.01231.x
American Conifer Society (2020) https://conifersociety.org/conifers/
Bag P, Chukhutsina V, Zhang Z, Paul S, Ivanov AG, Shutova T, Croce R, Holzwarth AR, Jansson S (2020) Direct energy transfer from photosystem II to photosystem I confers winter sustainability in Scots Pine. Nat Commun 11:1–13. https://doi.org/10.1038/s41467-020-20137-9
Bannister P, Neuner G (2001) Frost resistance and the distribution of conifers. In: Bigras FJ, Colombo S (eds) Conifer cold hardiness. Springer, Dordrecht, pp 3–21. https://doi.org/10.1007/978-94-015-9650-3_1
Bowling DR, Logan BA, Hufkens K, Aubrecht DM, Richardson AD, Burns SP, Anderegg WR, Blanken PD, Eiriksson DP (2018) Limitations to winter and spring photosynthesis of a Rocky Mountain subalpine forest. Agric Meteorol 252:241–255. https://doi.org/10.1016/j.agrformet.2018.01.025
Demmig-Adams B, Adams WW (1992) Photoprotection and other responses of plants to high light stress. Annu Rev Plant Biol 43:599–626. https://doi.org/10.1146/annurev.pp.43.060192.003123
Demmig-Adams B, Adams WW (2006) Photoprotection in an ecological context: the remarkable complexity of thermal energy dissipation. New Phytol 172:11–21. https://doi.org/10.1111/j.1469-8137.2006.01835.x
Ensminger I, Sveshnikov D, Campbell DA, Funk C, Jansson S, Lloyd J, Shibistova O, Öquist G (2004) Intermittent low temperatures constrain spring recovery of photosynthesis in boreal Scots pine forests. Glob Change Biol 10:995–1008. https://doi.org/10.1111/j.1365-2486.2004.00781.x
Esteban R, Barrutia O, Artetxe U, Fernández-Marín B, Hernández A, García-Plazaola JI (2015) Internal and external factors affecting photosynthetic pigment composition in plants: a meta-analytical approach. New Phytol 206:268–280. https://doi.org/10.1111/nph.13186
Foyer CH, Lelandais M, Kunert KJ (1994) Photooxidative stress in plants. Physiol Plant 92:696–717. https://doi.org/10.1111/j.1399-3054.1994.tb03042.x
Gamon JA, Huemmrich KF, Wong CYS, Ensminger I, Garrity S, Hollinger DY, Noormets A, Peñuelas J (2016) A remotely sensed pigment index reveals photosynthetic phenology in evergreen conifers. Proc Natl Acad Sci USA 113:13087–13092. https://doi.org/10.1073/pnas.1606162113
Hatcher PE (1990) Seasonal and age-related variation in the needle quality of five conifer species. Oecologia 85:200–212. https://doi.org/10.1007/BF00319402
Herben T, Nováková Z, Klimešová J, Hrouda L (2012) Species traits and plant performance: functional trade-offs in a large set of species in a botanical garden. J Ecol 100:1522–1533. https://doi.org/10.1111/j.1365-2745.2012.02018.x
Hochachka PW, Somero GN (1984) Biochemical Adaptation. Princeton University Press, Princeton
Jahns P, Holzwarth AR (2012) The role of the xanthophyll cycle and of lutein in photoprotection of photosystem II. BBA-Bioenergetics 1817:182–193. https://doi.org/10.1016/j.bbabio.2011.04.012
Kalberer SR, Wisniewski M, Arora R (2006) Deacclimation and reacclimation of cold-hardy plants: current understanding and emerging concepts. Plant Sci 171:3–16. https://doi.org/10.1016/j.plantsci.2006.02.013
Keenan RJ, Reams GA, Achard F, de Freitas JV, Grainger A, Lindquist E (2015) Dynamics of global forest area: Results from the FAO Global Forest Resources Assessment 2015. For Ecol Manage 352:9–20. https://doi.org/10.1016/j.foreco.2015.06.014
Körner C (2013) Growth controls photosynthesis–mostly. Nova Acta Leopoldina NF 114:273–283
Krieger-Liszkay A (2005) Singlet oxygen production in photosynthesis. J Exp Bot 56:337–346. https://doi.org/10.1093/jxb/erh237
Kromdijk J, Głowacka K, Leonelli L, Gabilly ST, Iwai M, Niyogi KK, Long SP (2016) Improving photosynthesis and crop productivity by accelerating recovery from photoprotection. Science 354:857–861. https://doi.org/10.1126/science.aai8878
Ledford HK, Niyogi KK (2005) Singlet oxygen and photo-oxidative stress management in plants and algae. Plant Cell Environ 28:1037–1045. https://doi.org/10.1111/j.1365-3040.2005.01374.x
Leverenz JW, Öquist G (1987) Quantum yields of photosynthesis at temperatures between−; 2° C and 35° C in a cold-tolerant C3 plant (Pinus sylvestris) during the course of one year. Plant Cell Environ 10:287–295. https://doi.org/10.1111/j.1365-3040.1987.tb01608.x
Little CHA (1970) Seasonal changes in carbohydrate and moisture content in needles of balsam fir (Abies balsamea). Can J Bot 48:2021–2028. https://doi.org/10.1139/b70-295
Logan BA (2006) Oxygen metabolism and stress physiology. In the structure and function of plastids. Kluwer Academic Publishers, Dorderecht, pp 539–553
Logan BA, Barker DH, Demmig-Adams B, Adams WW (1996) Acclimation of leaf carotenoid composition and ascorbate levels to gradients in the light environment within an Australian rainforest. Plant, Cell Environ 19:1083–1090. https://doi.org/10.1111/j.1365-3040.1996.tb00215.x
Logan BA, Grace SC, Adams WW, Demmig-Adams B (1998) Seasonal differences in xanthophyll cycle characteristics and antioxidants in Mahonia repens growing in different light environments. Oecologia 116:9–17. https://doi.org/10.1007/PL00013823
Logan BA, Demmig-Adams B, Adams WW (1999) Acclimation of photosynthesis to the environment. In: Singhal GS, Renger G, Sopory SK, Irrgang K-D, Govindjee I (eds) Concepts in photobiology: photosynthesis and photomorphogenesis. Narosa Publishing House, New Dehli, pp 477–512
Logan BA, Kornyeyev D, Hardison J, Holaday AS (2006) The role of antioxidant enzymes in photoprotection. Photosynth Res 88:119–132. https://doi.org/10.1007/s11120-006-9043-2
Magney TS, Bowling DR, Logan BA, Grossmann K, Stutz J, Blanken PD, Burns SP, Cheng R, Garcia MA, Kӧhler P, Lopez S, Parazoo N, Rackza B, Schimel D, Frankenberg C (2019) Mechanistic evidence for tracking the precise seasonality of photosynthesis with remotely sensed solar-induced fluorescence. Proc Natl Acad Sci USA 116:11640–11645. https://doi.org/10.1073/pnas.1900278116
Maxwell K, Johnson GN (2000) Chlorophyll fluorescence—a practical guide. J Exp Bot 51:659–668. https://doi.org/10.1093/jexbot/51.345.659
Míguez F, Fernández-Marín B, Becerril JM, García-Plazaola JI (2015) Activation of photoprotective winter photoinhibition in plants from different environments: a literature compilation and meta-analysis. Physiol Plant 155:414–423. https://doi.org/10.1111/ppl.12329
Motohka T, Nasahara KN, Oguma H, Tsuchida S (2010) Applicability of green-red vegetation index for remote sensing of vegetation phenology. Remote Sensing 2:2369–2387. https://doi.org/10.3390/rs2102369
Niklas KJ (1991) Biomechanical attributes of the leaves of pine species. Ann Bot 68:253–262. https://doi.org/10.1093/oxfordjournals.aob.a088251
Nippert JB, Duursma RA, Marshall JD (2004) Seasonal variation in photosynthetic capacity of montane conifers. Funct Ecol 18:876–886. https://doi.org/10.1111/j.0269-8463.2004.00909.x
Öquist G, Huner NP (2003) Photosynthesis of overwintering evergreen plants. Annu Rev Plant Biol 54:329–355. https://doi.org/10.1146/annurev.arplant.54.072402.115741
Primack RB, Miller-Rushing AJ (2009) The role of botanical gardens in climate change research. New Phytol 182:303–313. https://doi.org/10.1016/j.biocon.2009.03.016
R Core Team (2020) R: A language and environment for statistical computing, version 4.0.1. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/. Accessed 1 June 2020
Richardson AD, Jenkins JP, Braswell BH, Hollinger DY, Ollinger SV, Smith M-L (2007) Use of digital webcam images to track spring green-up in a deciduous broadleaf forest. Oecologia 152:323–334. https://doi.org/10.1007/s00442-006-0657-z
Rossi S, Deslauriers A, Anfodillo T, Carraro V (2007) Evidence of threshold temperatures for xylogenesis in conifers at high altitudes. Oecologia 152:1–12. https://doi.org/10.1007/s00442-006-0625-7
Sakai A, Larcher W (1987) Frost survival of plants. Springer, New York. https://doi.org/10.1007/978-3-642-71745-1
Seyednasrollah B, Bowling DR, Cheng R, Logan BA, Magney TS, Frankenberg C, Yang JC, Young AM, Hufkens K, Arain MA, Black TA, Blanken PD, Bracho R, Jassal R, Hollinger DY, Law BE, Nesic Z, Richardson AD (2020) Seasonal variation in the canopy color of temperate evergreen conifer forests. New Phytol. https://doi.org/10.1111/nph.17046
Sofronova VE, Dymova OV, Golovko TK, Chepalov VA, Petrov KA (2016) Adaptive changes in pigment complex of Pinus sylvestris needles upon cold acclimation. Russ J Plant Physiol 63:433–442. https://doi.org/10.1134/S1021443716040142
Sofronova VE, Antal TK, Dymova OV, Golovko TK (2018) Seasonal changes in primary photosynthetic events during low temperature adaptation of Pinus sylvestris in Central Yakutia. Russ J Plant Physiol 65:658–666. https://doi.org/10.1134/S1021443718050163
Steponkus PL (1984) Role of the plasma membrane in freezing injury and cold acclimation. Annu Rev Plant Physiol 35:543–584. https://doi.org/10.1146/annurev.pp.35.060184.002551
Strimbeck GR, Kjellsen TD, Schaberg PG, Murakami PF (2007) Cold in the common garden: comparative low-temperature tolerance of boreal and temperate conifer foliage. Trees 21:557–567
Strimbeck GR, Schaberg PG, Fossdal CG, Schröder WP, Kjellsen TD (2015) Extreme low temperature tolerance in woody plants. Front Plant Sci 6:884. https://doi.org/10.3389/fpls.2015.00884
Tranquillini W (2012) Physiological ecology of the alpine timberline. Springer, Berlin
Verhoeven A (2014) Sustained energy dissipation in winter evergreens. New Phytol 201:57–65. https://doi.org/10.1111/nph.12466
Verhoeven AS, Adams WW III, Demmig-Adams B (1999) The xanthophyll cycle and acclimation of Pinus ponderosa and Malva neglecta to winter stress. Oecologia 118:277–287. https://doi.org/10.1007/s004420050728
Wei T, Simko V, Levy M, Xie Y, Jin Y, Zemla J (2017) Package ‘corrplot.’ Statistician 56:e24
Wickham H (2016) ggplot2: elegant graphics for data analysis, version 3.3.2. Springer-Verlag, New York. ISBN 978-3-319-24277-4. https://ggplot2.tidyverse.org. Accessed 1 June 2020
Wong CYS, Gamon JA (2015a) The photochemical reflectance index provides an optical indicator of spring photosynthetic activation in evergreen conifers. New Phytol 206:196–208. https://doi.org/10.1111/nph.13251
Wong CYS, Gamon JA (2015b) Three causes of variation in the photochemical reflectance index (PRI) in evergreen conifers. New Phytol 206:187–195. https://doi.org/10.1111/nph.13159
Yang Q, Blanco NE, Hermida-Carrera C, Lehotai N, Hurry V, Strand Å (2020) Two dominant boreal conifers use contrasting mechanisms to reactivate photosynthesis in the spring. Nat Commun 11:1–12. https://doi.org/10.1038/s41467-019-13954-0
Acknowledgements
David Bowling and Barry Logan wish Russell Monson well on the occasion of his retirement and thank him for many years of mentoring, support and friendship. We thank Jason Baker and Marita Tewes at Red Butte Garden for permission to work in the garden and for logistical support.
Funding
This study was supported by the U. S. National Science Foundation Macrosystems and NEON-Enable Science Program (awards 1926090 and 1925992) and Division of Undergraduate Education (award 0088517). AW-M and DMB received travel support from the Grua-O’Connell Fund (Bowdoin College).
Author information
Authors and Affiliations
Contributions
DRB conceived of the study, which he designed with BAL. AW-M, MAG, DB, and AL performed chlorophyll fluorescence analyses. AW-M, DB, JSR and ES characterized needle chlorophyll and carotenoid composition. AW-M and CS led statistical analyses. AW-M conceived of and created figures. AW-M wrote the manuscript; all other authors provided editorial advice.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest or competing interests.
Additional information
Communicated by Kaoru Kitajima.
In this first-of-its-kind common-garden study, we report astonishing variation among seventy conifer taxa in their winter engagement of sustained energy dissipation, a key photoprotective mechanism.
Supplementary Information
Below is the link to the electronic supplementary material.
442_2021_5038_MOESM2_ESM.tiff
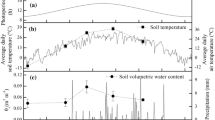
Supplementary file2 Temperature throughout period of winter measurement. Blue bars represent minimum and maximum temperature in 2019. Shown in gray bars are minimum and maximum temperature normals from 1981-2010. Days of measurement indicated by darker blue bars (TIFF 122510 KB)
442_2021_5038_MOESM3_ESM.tiff
Supplementary file3 Correlation matrix of parameters. Only relationships with p < 0.05 are represented with circles. Color denotes r2 as indicated, as does circle size. Abbreviations: A antheraxanthin; chl a chlorophyll a; chl b chlorophyll b; L lutein; N neoxanthin; PSII 200 and PSII 1600 photosystem II efficiency at 200 and 1600 µmol m-2 s-1, respectively; Z zeaxanthin. Pigment contents are on a needle fresh mass basis or, where indicated, on a total chlorophyll basis. n = 49 (pigment parameters) or 70 (parameters derived from chlorophyll fluorescence analysis) (TIFF 122510 KB)
442_2021_5038_MOESM4_ESM.tiff
Supplementary file4 Summer (pink) and winter (blue) needle pigment compositions on a fresh mass basis for individual taxa (genotype code [WT wildtype, C cultivar, D dwarf cultivar] and USDA numerical hardiness rating included). Abbreviations: A antheraxanthin; BC beta-carotene; chl a chlorophyll a; chl b chlorophyll b; L lutein; N neoxanthin; Z zeaxanthin. n = 49 (TIFF 134418 KB)
Rights and permissions
About this article
Cite this article
Walter-McNeill, A., Garcia, M.A., Logan, B.A. et al. Wide variation of winter-induced sustained thermal energy dissipation in conifers: a common-garden study. Oecologia 197, 589–598 (2021). https://doi.org/10.1007/s00442-021-05038-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-021-05038-y