Abstract
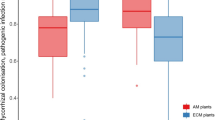
For plants, herbivory and interactions with their surrounding soil ecosystem are crucial factors influencing individual performance and plant-community composition. Until now, research has mostly focused on individual effects of herbivory or plant–soil feedbacks (PSFs) on plant growth and community composition, but few studies have explicitly investigated herbivory in the context of PSFs. These few studies, however, were performed under greenhouse conditions even though PSFs and herbivory may differ between greenhouse and field conditions. Therefore, we performed a field experiment in a grassland, testing the growth responses of three grass species that consistently differ in local abundance, on soils previously conditioned by these species. We tested these PSF effects for the three species both in the presence and in the absence of aboveground herbivores. Without herbivores, the two subdominant species suffered from negative PSF effects. However, in the presence of herbivores and on heterospecific soils, the same two species experienced a significant loss of shoot biomass, whereas, in contrast, enhanced root growth was observed on conspecific soils, resulting in overall neutral PSF effects. The dominant species was not damaged by herbivores and showed overall neutral PSF effects in the field with and without herbivores. Our study provides empirical evidence that negative PSF effects that exist under natural field conditions in grasslands can be overwhelmed by aboveground herbivory. Hence, potential PSF effects might not be detected in the field, because other abiotic and biotic interactions such as aboveground herbivory have stronger effects on plant performance and might therefore mask or override these PSF effects.



Similar content being viewed by others
References
Bardgett RD, Wardle DA, Yeates GW (1998) Linking above-ground and below-ground interactions: how plant responses to foliar herbivory influence soil organisms. Soil Biol Biochem 30:1867–1878. https://doi.org/10.1016/S0038-0717(98)00069-8
Bever JD (1994) Feedback between plants and their soil communities in an old field community. Ecology 75:1965–1977. https://doi.org/10.1111/j.1365-2745.2006.01104.x
Bever JD, Dickie IA, Facelli E, Facelli JM, Klironomos J, Moora M, Rillig MC, Stock WD, Tibbett M, Zobel M (2010) Rooting theories of plant community ecology in microbial interactions. Trends Ecol Evol 25:468–478. https://doi.org/10.1016/j.tree.2010.05.004
Bever JD, Mangan SA, Alexander HM (2015) Maintenance of plant species diversity by pathogens. Annu Rev Ecol Evol Syst 46:305–325. https://doi.org/10.1146/annurev-ecolsys-112414-054306
Bezemer TM, van Dam NM (2005) Linking aboveground and belowground interactions via induced plant defenses. Trends Ecol Evol 20:617–624. https://doi.org/10.1016/j.tree.2005.08.006
Bezemer TM, Lawson CS, Hedlund K, Edwards AR, Brook AJ, Igual JM, Mortimer SR, van der Putten WH (2006) Plant species and functional group effects on abiotic and microbial soil properties and plant–soil feedback responses in two grasslands. J Ecol 94:893–904. https://doi.org/10.1111/j.1365-2745.2006.01158.x
Bezemer TM, van der Putten WH, Martens H, van de Voorde TFJ, Mulder PPJ, Kostenko O (2013) Above- and below-ground herbivory effects on below-ground plant–fungus interactions and plant–soil feedback responses. J Ecol 101:325–333. https://doi.org/10.1111/1365-2745.12045
Bishop JG (2002) Early primary succession on Mount St. Helens: impact of insect herbivores on colonizing lupines. Ecology 83:191–202. https://doi.org/10.1890/0012-9658(2002)083[0191:EPSOMS]2.0.CO;2
Borgström P, Strengbom J, Viketoft M, Bommarco R (2016) Aboveground insect herbivory increases plant competitive asymmetry, while belowground herbivory mitigates the effect. PeerJ. https://doi.org/10.7717/peerj.1867
Branson DH, Sword GA (2009) Grasshopper herbivory affects native plant diversity and abundance in a grassland dominated by the exotic grass Agropyron cristatum. Restor Ecol 17:89–96. https://doi.org/10.1111/j.1526-100X.2007.00343.x
Brinkman EP, van der Putten WH, Bakker E-J, Verhoeven KJF (2010) Plant–soil feedback: experimental approaches, statistical analyses and ecological interpretations. J Ecol 98:1063–1073. https://doi.org/10.1111/j.1365-2745.2010.01695.x
Casper BB, Castelli JP (2007) Evaluating plant–soil feedbacks together with competition in a serpentine grassland. Ecol Lett 10:394–400. https://doi.org/10.1111/j.1461-0248.2007.01030.x
Cheli GH, Corley JC (2010) Efficient sampling of ground-dwelling arthropods using pitfall traps in arid steppes. Neotrop Entomol 39:912–917. https://doi.org/10.1590/S1519-566X2010000600010
Crawley MJ (1989) Insect herbivores and plant population dynamics. Annu Rev Entomol 34:531–564. https://doi.org/10.1146/annurev.en.34.010189.002531
Dierschke H (1997) Synopsis der Pflanzengesellschaften Deutschlands. Heft 3: Molinio- Arrhena- theretea (E1). Kulturgrasland und verwandte Vegetationstypen. Teil 1: Arrhenatheretalia. Wiesen und Weiden frischer Standorte. Göttingen
Ehrenfeld JG, Ravit B, Elgersma K (2005) Feedback in the plant–soil system. Annu Rev Environ Resour 30:75–115. https://doi.org/10.1146/annurev.energy.30.050504.144212
Engelkes T, Morriën E, Verhoeven KJF, Bezemer TM, Biere A, Harvey JA, McIntyre LM, Tamis WLM, van der Putten WH (2008) Successful range-expanding plants experience less above-ground and below-ground enemy impact. Nature 456:946–948. https://doi.org/10.1038/nature07474
Engelkes T, Meisner A, Morriën E, Kostenko O, van der Putten WH, Macel M (2016) Herbivory and dominance shifts among exotic and congeneric native plant species during plant community establishment. Oecologia 180:507–517. https://doi.org/10.1007/s00442-015-3472-6
Franzke A, Unsicker SB, Specht J, Köhler G, Weisser WW (2010) Being a generalist herbivore in a diverse world: how do diets from different grasslands influence food plant selection and fitness of the grasshopper Chorthippus parallelus? Ecol Entomol 35:126–138. https://doi.org/10.1111/j.1365-2311.2009.01168.x
Gardiner T, Pye M, Field R, Hill J (2002) The influence of sward height and vegetation composition in determining the habitat preferences of three Chorthippus species (Orthoptera: Acrididae) in Chelmsford, Essex, UK. J Orthoptera Res 11:207–213. http://www.jstor.org/stable/3503697
Gustafson DJ, Casper BB (2004) Nutrient addition affects AM fungal performance and expression of plant/fungal feedback in three serpentine grasses. Plant Soil 259:9–17. https://doi.org/10.1023/B:PLSO.0000020936.56786.a4
Heinz KM, Parrella MP, Newman JP (1992) Time-efficient use of yellow sticky traps in monitoring insect populations. J Econ Entomol 85:2263–2269. https://doi.org/10.1093/jee/85.6.2263
Heinze J, Bergmann J, Rillig MC, Joshi J (2015) Negative biotic soil-effects enhance biodiversity by restricting potentially dominant plant species in grasslands. Perspect Plant Ecol Evol Syst 17:227–235. https://doi.org/10.1016/j.ppees.2015.03.002
Heinze J, Sitte M, Schindhelm A, Wright J, Joshi J (2016) Plant–soil feedbacks: a comparative study on the relative importance of soil-feedbacks in the greenhouse vs. field. Oecologia 181:559–569. https://doi.org/10.1007/s00442-016-3591-8
Heinze J, Gensch S, Weber E, Joshi J (2017) Soil temperature modifies effects of soil biota on plant growth. J Plant Ecol. https://doi.org/10.1093/jpe/rtw097
Holland NJ, Cheng W, Crossley DA (1996) Herbivore-induced changes in plant carbon allocation assessment of below-ground C fluxes using carbon-14. Oecologia 107:87–94. https://doi.org/10.1007/BF00582238
Johnson SN, Erb M, Hartley SE (2016) Roots under attack: contrasting plant responses to below- and aboveground insect herbivory. New Phytol 210:413–418. https://doi.org/10.1111/nph.13807
Kaplan I, Halitschke R, Kessler A, Sardanelli S, Denno RF (2008) Constitutive and induced defenses to herbivory in above- and belowground plant tissues. Ecology 89:391–406. https://doi.org/10.1890/07-0471.1
Klironomos JN (2002) Feedback with soil biota contributes to plant rarity and invasiveness in communities. Nature 417:67–70. https://doi.org/10.1038/417067a
Klötzli F, Dietl W, Marti K, Schubiger-Bosshard C, Walther G-R (2010) Vegetation Europas—Das Offenland im vegetationskundlich-ökologischen Überblick. Ott Verlag, Bern, p 1192
Kneis D, Knoesche R, Bronstert A (2006) Analysis and simulation of nutrient retention and management for a lowland river-lake system. Hydrol Earth Syst Sci 10:575–588. http://www.hydrol-earth-syst-sci.net/10/575/2006/
Kos M, Tuijl MAB, de Roo J, Mulder PPJ, Bezemer TM (2015) Species-specific plant–soil feedback effects on above-ground plant–insect interactions. J Ecol 103:904–914. https://doi.org/10.1111/1365-2745.12402
Kostenko O, van de Voorde TFJ, Mulder PPJ, van der Putten WH, Bezemer TM (2012) Legacy effects of aboveground-belowground interactions. Ecol Lett 15:813–821. https://doi.org/10.1111/j.1461-0248.2012.01801.x
Kowarik I (2010) Biologische Invasionen. Neophyten und Neozoen in Mitteleuropa. Ulmer, Stuttgart
Kulmatiski A (2016) Factorial and, self vs. other’ plant soil feedback experiments produce similar predictions of plant growth in communities. Plant Soil 408:485–492. https://doi.org/10.1007/s11104-016-2946-6
Kulmatiski A, Kardol P (2008) Getting plant–soil feedbacks out of the greenhouse: experimental and conceptual approaches. In: Lüttge U, Beyschlag W, Murata J (eds) Progress in botany, vol 69. Springer, Heidelberg, pp 449–472. https://doi.org/10.1007/978-3-540-72954-9_18
Kulmatiski A, Beard KH, Stevens JR, Cobbold SM (2008) plant–soil feedbacks: a meta-analytic review. Ecol Lett 11:980–992. https://doi.org/10.1111/j.1461-0248.2008.01209.x
Luck RF, Shepard BM, Kenmore PE (1988) Experimental methods for evaluating arthropod natural enemies. Annu Rev Entomol 33:367–391. https://doi.org/10.1146/annurev.en.33.010188.002055
MacDonald AAM, Kotanen PM (2010) The effect of disturbance and enemy exclusion on performance of an invasive species, common ragweed, in its native range. Oecologia 162:977–986. https://doi.org/10.1007/s00442-009-1557-9
Massey FP, Ennos E, Hartley SE (2007) Grasses and the resource availability hypothesis: the importance of silica-based defences. J Ecol 95:414–424. https://doi.org/10.1111/j.1365-2745.2007.01223.x
Mattson WJ (1980) Herbivory in relation to plant nitrogen content. Annu Rev Ecol Syst 11:119–161. https://doi.org/10.1146/annurev.es.11.110180.001003
Mokany K, Raison RJ, Prokushkin AS (2006) Critical analysis of root:shoot ratios in terrestrial biomes. Glob Change Biol 12:84–96. https://doi.org/10.1111/j.1365-2486.2005.001043.x
Morriën E, Engelkes T, van der Putten WH (2011) Additive effects of aboveground polyphagus herbivores and soil feedback in native and range-expanding exotic plants. Ecology 92:1344–1352. https://doi.org/10.1890/10-1937.1
Olsen SR, Cole CV, Watanabe FS, Dean LA (1954) Estimation of available phosphorus in soils by extraction with sodium bicarbonate, vol 939. USDA circular. Gov. Printing Office, Washington, DC. pp 1–19
Pangesti N, Pineda A, Pieterse CMJ, Dicke M, van Loon JJA (2013) Two-way plant-mediated interactions between root-associated microbes and insects: from ecology to mechanisms. Front Plant Sci 4:1–11. https://doi.org/10.3389/fpls.2013.00414
Pearce JL, Schuurman D, Barber KN, Larrivee M, Vernier LA, McKee J, McKenney D (2005) Pitfall trap designs to maximize invertebrate captures and minimize captures of nontarget vertebrates. Can Entomol 137:233–250. https://doi.org/10.4039/n04-029
Pfisterer A, Diemer M, Schmid B (2003) Dietary shift and lowered biomass gain of a generalist herbivore in species-poor experimental plant communities. Oecologia 135:234–241. https://doi.org/10.1007/s00442-002-1169-0
Pfitzenmeyer CDC (1962) Arrhenatherum elatius (L.) J. &. C. Presl (A. avenaceum Beauv.). J Ecol 50:235–245
R Developmental Core Team (2014) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Reinhart KO, Rinella MJ (2016) A common soil handling technique can generate incorrect estimates of soil biota effects on plants. New Phytol 210:786–789. https://doi.org/10.1111/nph.13822
Russel FL, Rose KE, Louda SM (2010) Seed availability and insect herbivory limit recruitment and adult density of native tall thistle. Ecology 91:3081–3093. https://doi.org/10.1890/09-1101.1
Schädler M, Jung G, Auge H, Brandl R (2003) Palatability, decomposition and insect herbivory: patterns in a successional old-field plant community. Oikos 103:121–132. https://doi.org/10.1034/j.1600-0706.2003.12659.x
Schittko C, Wurst S (2014) Above- and belowground effects of plant–soil feedback from exotic Solidago canadensis on native Tanacetum vulgare. Biol Invasions 16:1465–1479. https://doi.org/10.1007/s10530-013-0584-y
Schittko C, Runge C, Strupp M, Wolff S, Wurst S (2016) No evidence that plant–soil feedback effects of native and invasive plant species under glasshouse conditions are reflected in the field. J Ecol 104:1243–1249. https://doi.org/10.1111/1365-2745.12603
Smith-Ramesh LM, Reynolds HL (2017) The next frontier of plant–soil feedback research: unraveling context dependence across biotic and abiotic gradients. J Veg Sci 28:484–494. https://doi.org/10.1111/jvs.12519
Southwood TRE, Henderson PA (2000) Ecological methods, 3rd edn. Blackwell Science, London
Stinchcombe JR, Rausher MD (2002) The evolution of tolerance to deer herbivory: modifications caused by the abundance of insect herbivores. Proc Biol Sci 269:1241–1246. https://doi.org/10.1098/rspb.2002.2015
Thompson DC, Gardner KT (1996) Importance of grasshopper defoliation period on southwestern blue grama-dominated rangeland. J Range Manag 49:494–498. https://doi.org/10.2307/4002288
Van der Heijden MGA, Bardgett RD, van Straalen NM (2008) The unseen majority: soilmicrobes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecol Lett 11:296–310. https://doi.org/10.1111/j.1461-0248.2007.01139.x
Van der Putten WH, Bardgett RD, Bever JD, Bezemer TM, Casper BB, Fukami T, Kardol P, Klironomos JN, Kulmatiski A, Schweitzer JA, Suding KN, van de Voorde TFJ, Wardle DA (2013) Plant–soil feedbacks: the past, the present and future challenges. J Ecol 101:265–276. https://doi.org/10.1111/1365-2745.12054
Van der Wal R, Madan N, van Lieshout S, Dormann C, Langvatn R, Albon SD (2000) Trading forage quality for quantity? Plant phenology and patch choice by Svalbard reindeer. Oecologia 123:108–115. https://doi.org/10.1007/s004420050995
Van Ruijven J, de Deyn GB, Raaijmakers CE, Berendse F, van der Putten WH (2005) Interactions between spatially separated herbivores indirectly alter plant diversity. Ecol Lett 8:30–37. https://doi.org/10.1111/j.1461-0248.2004.00688.x
Wardle DA, Bardgett RD, Klironomos JN, Setälä H, van der Putten WH, Wall DH (2004) Ecological linkages between aboveground and belowground biota. Science 304:1629–1633. https://doi.org/10.1126/science.1094875
Yang Y, Fang J, Ma W, Guo D, Mohammat A (2010) Large-scale pattern of biomass partitioning across China’s grasslands. Global Ecol Biogeogr 19:268–277. https://doi.org/10.1111/j.1466-8238.2009.00502.x
Zou Y, Feng J, Xue D, Sang W, Axmacher JC (2012) A comparison of terrestrial arthropod sampling methods. J Resour Ecol 3:174–182. https://doi.org/10.5814/j.issn.1674-764x.2012.02.010
Acknowledgements
We thank Frank Warschau and Mario Sitte for help in the field, Gabriele Gehrmann for help with the soil analysis, Ronald Wille and Stefan Saumweber for technical support for the measurements of abiotic conditions and the Botanical Garden Potsdam for their cooperation.
Author information
Authors and Affiliations
Contributions
JH designed the study, performed the experiment and analyzed the data. JH and JJ wrote the manuscript.
Corresponding author
Additional information
Communicated by Edith B. Allen.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Heinze, J., Joshi, J. Plant–soil feedback effects can be masked by aboveground herbivory under natural field conditions. Oecologia 186, 235–246 (2018). https://doi.org/10.1007/s00442-017-3997-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-017-3997-y