Abstract
The dynamic interactions among nutrients, algae and grazers were tested in a 2 × 3 factorial microcosm experiment that manipulated grazers (Daphnia present or absent) and algal composition (single species cultures and mixtures of an undefended and a digestion-resistant green alga). The experiment was run for 25 days in 10-L carboys under mesotrophic conditions that quickly led to strong phosphorus limitation of algal growth (TP ≅ 0.5 μM, N:P 40:1). Four-day Daphnia juvenile growth assays tested for Daphnia P-limitation and nutrient-dependent or grazer-induced algal defenses. The maximal algal growth rate of undefended Ankistrodesmus (mean ± SE for three replicate microcosms; 0.92 ± 0.02 day−1) was higher than for defended Oocystis (0.62 ± 0.03 day−1), but by day 6, algal growth was strongly P-limited in all six treatments (molar C:P ratio >900). The P-deficient algae were poor quality resources in all three algal treatments. However, Daphnia population growth, reproduction, and survival were much lower in the digestion-resistant treatment even though growth assays provided evidence for Daphnia P-limitation in only the undefended and mixed treatments. Growth assays provided little or no support for simple threshold element ratio (TER) models that fail to consider algae defenses that result in viable gut passage. Our results show that strong P-limitation of algal growth enhances the defenses of a digestion-resistant alga, favoring high abundance of well-defended algae and energy limitation of zooplankton growth.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Stoichiometric constraints can reduce food quality for many aquatic and terrestrial herbivores (Elser et al. 2000; Sterner and Elser 2002). Daphnia and phosphorus have received much attention in part because of their importance in lakes, and in part because daphniids have high requirements for P compared to other crustacean zooplankton. Threshold element ratio (TER) models predict the resource C:nutrient (usually N or P) ratio at which the grazer’s growth shifts from energy to nutrient limitation, taking into account the grazer’s nutrient content, ingestion rate, assimilation efficiencies (AEs), respiration costs, and nutrient excretion. The predicted molar C:P TER for Daphnia ranges from about 200 at high food to 230 at low food concentration (Anderson and Hessen 2005). TER models have been developed using laboratory data for high quality, undefended green algae. Thus, TER models include food quantity but, thus far, have not accounted for algal defenses that reduce ingestion rate or AE.
Deficiencies in essential fatty acids (EFAs) and algal digestion defenses have been proposed as alternatives to Daphnia P-limitation (reviewed by Sterner and Schulz 1998; Moe et al. 2005). Evidence that algae grown under nutrient limitation produce less EFAs supported an hypothesized interaction between P limitation and EFA limitation in Daphnia (Müller-Navarra 1995). However, while essential fatty acids are important for zooplankton growth (reviewed in Ravet et al. 2010), EFA supplements do not stimulate the growth of Daphnia feeding on strongly P-deficient green algae (Boersma 2000; Plath and Boersma 2001), while supplements of phosphate and phosphorus-rich but EFA-deficient cyanobacteria do (DeMott 1998).
Certain green algae that are high quality, undefended resources when grown without nutrient limitation develop thicker cell walls that resist digestion when grown under P-limitation (Van Donk and Hessen 1993; Van Donk et al. 1997). Since an increased proportion of cells survive gut passage, digestion resistance acts as a defense. We describe such algae as having “nutrient-dependent defenses” to distinguish them from constitutive defenses that are expressed in nutrient sufficient cultures (reviewed by Sterner 1989) and defenses that are induced by a grazer’s chemical or physical presence (reviewed by Van Donk et al. 2011). The terrestrial plant literature provides many examples in which constitutive defenses against herbivores are strengthened following leaf damage or are modulated by drought, nutrient limitation, or other stressors (reviewed by Stamp 2003). Which algae strengthen their defenses when nutrients are limiting and how strongly do these nutrient-dependent defenses vary between algal species? As far as we know, no study has tested whether planktonic algae with constitutive digestion defenses under nutrient-sufficient conditions show nutrient-dependent defenses under P-limitation.
One important goal for stoichiometric theory and TER models, in particular, is to predict when grazer growth will be P-limited in nature. The surprisingly few direct tests of Daphnia P-limitation in nature have provided mixed results. The best evidence for Daphnia P-limitation in nature comes from experimental testing in oligotrophic Canadian Shield lakes (Elser et al. 2001; Makino et al. 2002) and hypereutrophic Dutch lakes (DeMott et al. 2001). The importance of Daphnia P-limitation in the Dutch lakes gains further support from a 9-year study of between-year variation in seston C:P ratios and Daphnia abundance (DeMott and Gulati 1999; Van Donk et al. 2008). As predicted by TER models, high resource C:P ratios were associated with Daphnia P-limitation and low Daphnia abundance in both the Canadian and Dutch lakes. Several studies, however, provide no evidence for Daphnia P-limitation despite resources at or above predicted threshold C:P ratios, including research in temperate (DeMott and Tessier 2002; DeMott et al. 2004) and tropical (Fileto et al. 2007) lakes and polar ponds (Van Geest et al. 2007).
DeMott and Tessier (2002) used growth assays and isotope assimilation experiments to show that energy limitation due to digestion defenses accounted for the strong food quality limitation of Daphnia in deep, stratified Michigan lakes, despite resource C:P ratios well above nominal TER values. In contrast with shallow Michigan lakes and the Dutch and Canadian lakes mentioned above, the deep Michigan lakes were inhabited by high densities of large-bodied Daphnia and contained high relative abundance of algae with constitutive digestion defenses (Tessier and Woodruff 2002). Similar gradients of constitutive digestion defenses have been shown when grazer abundance was manipulated in field mesocosms (e.g., Porter 1973; Vanni 1997; Kerfoot et al. 1988). We (DeMott and Tessier 2002) showed that the interaction between algal defenses and algal P-limitation can lead to energy limitation in Daphnia, but we did not consider that digestion defenses could be strengthened when algal growth is nutrient limited.
Here, we use a laboratory microcosm experiment to test the effectiveness of algal defenses and their potential interactions with stoichiometric constraints. We manipulated grazers (Daphnia present or absent) and algal composition (single species cultures and mixtures of a defended and an undefended green alga). Our defended alga was a strain of Oocystis with a gelatinous sheath and thick cell walls that provide moderate constitutive defenses against digestion (see below). Using an alga with moderate constitutive defenses allowed us to test whether defenses would be enhanced under P-limited growth or induced by Daphnia’s presence. As in the studies by Elser et al. (2001), DeMott et al. (2001), and DeMott and Tessier (2002), we used juvenile growth assays with phosphate supplements to test for Daphnia P-limitation. Comparison of juvenile growth assays with resources from the grazed and control (Daphnia-free) microcosms tested for grazer-induced defenses.
Microcosm experiments can provide realistic feedbacks between grazer density, resource abundance, and resource C:P ratios. Previous microcosm experiments with strong P-limitation of algal growth provided evidence for density-dependent facilitation in Daphnia (Sommer 1992; Nelson et al. 2001; Urabe et al. 2002). In this situation, food conditions improve when Daphnia populations reduce the abundance of poorly defended, P-limited algae, causing a decline in the resource C:P ratio and a switch from Daphnia P-limitation to energy limitation. Comparison of the dynamics of the two algal species in the mixed treatment should provide the clearest basis for assessing the effectiveness of algal defenses. According to the algal defenses hypothesis (DeMott and Tessier 2002), effective defenses could lead to the coexistence of energy-limited Daphnia with high abundance of P-deficient, well-defended Oocystis.
Materials and methods
Experimental design
The effectiveness of algal defenses and their interactions with stoichiometric constraints were tested in a microcosm experiment that manipulated Daphnia presence and algal composition (undefended Ankistrodesmus and digestion-resistant Oocystis in single species cultures or a mixture). The microcosms were randomly positioned in 3 replicate blocks of 6 (2 grazer × 3 algae treatments; 18 total). The experiment was run for 25-days under nutrient and light conditions that were expected to produce strong P limitation of algal growth. Transparent Nalgene (Rochester, New York, USA) polycarbonate carboys were filled with 10-L of Combo medium (Kilham et al. 1998) with 10 μg P-phosphate L−1 and an N:P ratio of 40:1. The experiment was run in a temperature-controlled room (20 °C) with fluorescent lights (16 h light: 8 h dark) positioned about 1.2 m above the water level (PAR intensity measured at water level with a Li Cor model LI-189 photometer; mean ± SD = 96 ± 7.5 μmol s−1 m−2). Each microcosm was mixed by gentle continuous aeration and was lifted and swirled daily to resuspend particles that had settled to the bottom.
The grazer was a clone of a Daphnia pulex × pulicaria hybrid (Tessier and Woodruff 2002). This clone was the most sensitive to P-limitation of 10 Daphnia taxa tested by DeMott and Pape (2004). Animals for stocking the 9 Daphnia microcosms were fed P-rich Ankistrodesmus in excess, were grown in 5 cohorts comprising animals born within a 24-h period (range 0–12 days old), and were counted out individually (63 animals including 13 adults per microcosm).
The digestion-resistant Oocystis, which typically grew in colonies of 1–4 cells, was isolated from the guts of field-collected zooplankton about 8 months before the start of the experiment (mesotrophic Crooked Lake, Whitley and Noble counties, Indiana, USA). The digestion defenses of this gelatinous green alga were confirmed by visual observations on viable gut passage (DeMott et al. 2010) and by a preliminary juvenile growth assay that gave a growth rate of 0.37 ± 0.02 day−1 with a high food concentration (1 mg C L−1) from a P-sufficient culture. Previous studies with the same Daphnia clone and similar conditions gave juvenile growth rates ranging from 0.10 to 0.46 day−1 for 4 digestion-resistant green algae and from 0.57 to 0.61 for 3 undefended green algae, including Ankistrodesmus (DeMott et al. 2010). The undefended green alga, Ankistrodesmus falcatus, which grows in single, needle-shaped cells, was also used for culturing Daphnia. Estimates of Daphnia AE using 32P-labeled cells revealed a small but statistically significant decrease in the digestibility Ankistrodesmus with strong P-limitation (Ferrao-Filho et al. 2007). Algae from batch cultures growing in P-rich WC medium (Guillard and Lorenzen 1972) were centrifuged, resuspended in P-free Combo medium, and added to the microcosms at a concentration of 0.2 mg C L−1. The mixed treatment microcosms received 0.1 mg C L−1 of each algal species.
Sampling, sample processing and data analysis
The initial (day 0) Daphnia population density was based on exact counts of animals stocked into each microcosm, while the initial estimates of particulate organic carbon (POC), particulate phosphorus (PP), and the resource C:P ratio were calculated from samples of the algal cultures added to the microcosms. Initial samples for estimates of particle volume and algal cell densities were collected from the microcosms with 25-mL glass pipettes.
Subsequent routine sampling used plexiglass tubes with an inner diameter of 56 mm and a volume of 400 mL (De Senerpont Domis et al. 2007). Before sampling, each microcosm was stirred with the sampling device to insure complete mixing. To reduce the chance of contamination, a separate sampling tube was used for each microcosm. Two tubes of water (800 mL) were collected from each microcosm on five sampling dates over 25 days, and the water was filtered through a 60-μm screen to collect Daphnia, which were preserved in a sucrose–formalin mixture. Sampling imposed a mortality rate of about 0.02 day−1 on the Daphnia and algal populations. The filtrate was used to estimate resource abundance (POC, PP, particulate volume, algal cell counts), the resource C:P ratio, and as a medium for juvenile growth assays. After sampling, Combo medium with phosphate was added to replace the volume sampled. Greater volumes were sampled for Daphnia on the penultimate (1.6 L) and final (3.2 L) sampling dates from microcosms with low grazer abundance.
All Daphnia in each sample were counted and measured using a dissecting microscope and Autoshape software (v.3.0; Comef). We determined clutch size as the number of parthenogenic eggs per adult female. The length of the smallest egg-bearing female was taken as the minimum size of the adult stage (1.50 mm).
POC, PP, and the C:P ratio were estimated from matter collected on the same glassfiber filter (GF/F, 25 mm diameter; Whatman, Maidstone, UK). Four small circles cut from each filter with a paper punch were analyzed for carbon as CO2 in a CN analyzer (Flash 2000; Interscience). The remaining filter was ashed for 30 min at 550 °C, dissolved in 10 mL of 2 % persulfate, and analyzed as orthophosphate according to Murphy and Riley (1962), using an autoanalyser (Quaatro; Seal Analytical, Beun de Ronde, Abcoude, The Netherlands). A number of spuriously low PP values (6 of 90) were replaced with the mean of the previous and following sampling dates.
The volume of algae, detritus, and bacteria (0–30 μm diameter) was estimated using a CASY model TTC (Shärfe System) electronic particle counter with a 100-μm aperture. Samples were collected and analyzed daily during the first 4 days and every second day thereafter. On days when tube samples were not collected, samples were collected with 25-mL glass pipettes. The particle counter apparently sensed Oocystis cells individually, and thus did not provide useful data on Oocystis colony size.
Algal densities were estimated from samples preserved in acidified Lugol’s solution and counted at ×200 using an inverted microscope. Estimates for each sample and species were based on counts of at least 100 colonies or cells and 8 fields of view. The effect of Daphnia presence on Oocystis size was tested by measuring the diameter of Oocystis colonies from the 12 microcosms in the Oocystis and mixed treatments on days 3 and 20. Photographs were taken at ×400 of 6–12 random fields from each sample. Diameter (mean of length and width of ovoid colonies) was estimated for each colony in clear focus (range 37–95 colonies per sample) using Cell D Software (v.1.2; Olympus).
Daphnia juvenile growth assays
Since P-deficient algae take up phosphate from solution within minutes, short-term growth assays with phosphate supplements provide direct tests of Daphnia P-limitation. P supplements typically restore much but not all of the reduced growth of Daphnia fed undefended algae cultured under P-deficient conditions (DeMott 1998; Boersma 2000; Plath and Boersma 2001). Thus, we tested the intensity and mechanism of food limitation by growing newborn Daphnia for 4 days in resources from the microcosms with and without phosphate supplements (0.5 μmol L−1). All growth assays were run using cohorts of Daphnia born within the previous 24 h to mothers fed high concentrations of P-sufficient Ankistrodesmus, and were run in the temperature-controlled room (20 °C) and in the dark to prevent algal growth and to limit potential changes in algal biochemistry or morphology. Since Daphnia feeding on P-deficient resources show declines in body P-content and produce young with reduced P-content (DeMott et al. 1998), single generation growth essays underestimate stoichiometric constraints on Daphnia growth (Frost et al. 2010).
Growth assays started on days 6, 11, 15, and 20 were run with resources from the three Daphnia treatments and the ungrazed Oocystis treatment. Thus, on each of the four dates, we started an experiment with 8 treatments (2 phosphorus × 4 algal treatments). In addition, on each of these 4 days, and on day 0, we ran treatments with Ankistrodesmus and Oocystis from cultures growing exponentially in P-rich WC medium at a concentration of 1.0 mg C L−1. These positive controls tested whether animals would show the expected high growth rates with P-rich resources. The growth assays were run with 3 Daphnia in 100 mL of medium and the medium was changed after 2 days. On days when the medium was not changed (days 1 and 3), particles that had settled to the bottom were resuspended with a glass pipette. Water collected from each microcosm was stored in a beaker in the dark for use in one replicate of a growth assay and the corresponding P-enriched beaker. Thus, the bioassays reflected resource variation between the replicate microcosms.
Lipid reserves can provide clues to whether Daphnia growth is limited by P or energy. Animals experiencing P-limitation assimilate carbon in excess of energy requirements and may accumulate substantial lipid reserves despite low growth (Sterner et al. 1992). In contrast, energy limitation and starvation lead to low lipid reserves (Tessier et al. 1983). At the end of each growth assay, a visual lipid–ovary index was estimated for each individual on a scale from 0 (no lipid reserves) to 3 (maximal lipids; Tessier et al. 1983). The animals were then dried and weighed to the nearest μg. The growth rate (g day−1) was calculated as the difference between the natural logarithm of the final and the initial dry mass, divided by the experiment duration (4 days). P-differentials were calculated by subtracting the growth rate for a control beaker from growth for the beaker with added phosphate from the same microcosm (DeMott and Tessier 2002).
A final series of growth assay experiments was designed to test mechanisms for the poor quality of P-deficient resources. Phosphate additions should reduce the algal C:P ratio below potentially limiting levels within minutes. However, we hypothesized that phosphate, light, and sufficient time for cell division would be needed to overcome the nutrient-dependent digestion defenses of thickened cell walls and gelatinous sheaths. Preparation for this experiment was started on day 25 using composite samples from the strongly P-deficient, ungrazed Ankistrodesmus and Oocystis microcosms. Each algal species was grown in 3 flasks under 3 sets of conditions for 3 days. In the P-deficient treatment, 1 L of algae was kept in the light without added phosphate. In second and third treatments, 800 mL of microcosm resources were diluted with 200 mL of P-rich WC algal medium and incubated in the light or in the dark, respectively. The growth assays included five treatments for each algal species: (1) P-sufficient laboratory culture control, (2) added WC medium incubated in light, (3) added WC medium incubated in the dark, (4) P-deficient treatment +0.5 μmol phosphate L−1, and (5) P-deficient treatment. These growth assays followed the same methods as the previous experiments except that the medium was changed daily and the food concentration was diluted to 1 mg C L−1. Limiting the food concentration insured that the algal C:P ratio would be reduced below nominal TER values (<200) in treatments with added phosphate and WC medium. The phosphate supplement in treatment 4 was added immediately after the daily medium change. Samples were filtered from the 6 stock culture flasks to estimate the resource C:P ratios.
Statistical analysis
The effects of algal treatment on Daphnia dynamics were analyzed by one-way ANOVA while the effects of grazers on their resources were analyzed by two-way ANOVA (grazer × algal species). A Daphnia × algae species interaction provides evidence of differences between Ankistrodesmus and Oocystis in their susceptibility to Daphnia grazing. Pairwise comparisons were based on the Holm–Sidak method. The parameter value from each microcosm was the mean from day 6 (when the first zooplankton samples were taken) until the end of the experiment (day 25). For juvenile growth assays, each P-differential was testing using the t distribution to determine whether the 95 % confidence interval overlapped with zero. With the exceptions of exponential rates of individual and population growth, data were transformed to equalize variances (log transformation of measurements; arcsin transformation of proportions).
Results
Daphnia dynamics
Algal species composition strongly affected Daphnia population dynamics, including population density, clutch size, and the proportion of adult females (three one-way ANOVAs, F = 13.2–128, each p < 0.01). Daphnia dynamics reflected similarities between the Ankistrodesmus and mixed treatments (3 pairwise contrasts, each p > 0.05) and strong divergences between the Oocystis treatment and the other two treatments (6 contrasts, each p < 0.01). As described below, Daphnia population growth, reproduction, and survival were much lower in the Oocystis treatment.
Daphnia populations increased in all three treatments during the first 6 days of the experiment (Fig. 1a). However, from day 6 to day 20, Daphnia in the Oocystis treatment declined sharply, while populations in both the Ankistrodesmus and mixed treatments showed gradual increases and slow declines over the same period.
Effects of three algal treatments on a Daphnia population density, b clutch size, and c proportion of adults during the 25 day experiment. Algal treatments include Ankistrodesmus (circles), Oocystis (squares), or a mixture (triangles). Data are mean ± SE for 3 replicate mesocosms. p values are from one-way ANOVAs. Note log scale in (a)
The divergences in Daphnia abundance between the Oocystis treatment and the other two treatments were corroborated by data on reproduction. Clutch size in the Oocystis treatment declined from 2.5 ± 0.9 on day 6 to near-zero values from days 11–20 (Fig. 1b). Clutch size in the other two treatments declined from high, nearly identical levels on day 6 to near-zero values on days 20 and 25.
Declines in the proportion of adults early in the experiment can be attributed to reproduction and poor conditions for juvenile growth (Fig. 1c). However, between days 11 and 15, the proportion of adults in the Oocystis treatment increased from 0.31 ± 0.028 to 1.00, as body length increased from 1.09 ± 0.06 to 1.90 ± 0.06 mm. The rapid increases in both body size and the proportion of adults during this period of near-zero reproduction (Fig. 1b) and population decline (Fig. 1a) were probably due to nearly complete juvenile mortality, rather than rapid juvenile growth.
Resource dynamics
Particle volume (0–30 μm) was closely correlated with POC measured from the same sample (log–log plot; r 2 = 0.97; data not shown). Both algal species showed maximal exponential growth in biovolume from days 1 to 3 in their respective controls (log particle volume versus time; Ankistrodesmus, r 2 > 0.99; Oocystis, r 2 = 0.92–0.98). The maximal exponential growth rate of Ankistrodesmus (0.93 ± 0.01 day−1) was greater than that of Oocystis (0.63 ± 0.01 day−1; t = 16.8, p < 0.001).
Particulate organic carbon (POC) was reduced in the Daphnia treatment (F = 484, p < 0.001) and the Daphnia effect differed between algal treatments (Daphnia × algae interaction: F = 78; p < 0.001; Fig. 2a). Moreover, POC was lower in the Ankistrodesmus treatment than the other two treatments (2 contrasts, each p < 0.02). Resources remained abundant in the mixed and Oocystis treatments. Thus, the sharp decline in Daphnia abundance in the Oocystis treatment occurred despite consistently high food abundance (range 1.4–4.7 mg C L−1 from day 6 to final day).
Effects of Daphnia presence (filled symbols) or absence (open symbols) and three algal treatments on a particulate organic carbon, and b molar C:P ratio. Algal treatments include Ankistrodesmus (circles), Oocystis (squares), or a mixture (triangles). Data are mean ± SE for replicate microcosms. Error bars were removed from (b) for clarity. Note log scales
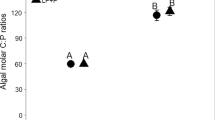
All treatments showed sharp increases in the C:P ratio over the first 6 days to values far above the nominal TER for Daphnia P-limitation (Fig. 2b). On day 6, the mean C:P ratio ranged from 910 to 1,000 in the Daphnia treatments and from 1,110 to 1,700 in the ungrazed controls. From day 6 to the end of the experiment, the resource C:P ratio was lower in the Daphnia treatment (F = 64.5, p < 0.001) and the Daphnia effect differed between algae treatments (Daphnia × algae interaction; F = 25.0, p < 0.001). Pairwise comparisons show significant reductions of the C:P ratio in the Daphnia treatment for the Ankistrodesmus and mixed treatments relative to their respective controls (both p < 0.001), but not in the Oocystis treatment (p = 0.33). With the exception of the last sampling date in the Ankistrodesmus treatment (mean 284 ± 26), the C:P ratio remained well above the nominal threshold for Daphnia P-limitation in all treatments from day 6 until the end of the experiment (C:P >600; Fig. 2b).
Consistent with the POC data, algal cell counts demonstrate very strong differences between Ankistrodesmus and Oocystis in their vulnerability to suppression by Daphnia grazing (Fig. 3). Separate two-way ANOVAs showed strong Daphnia effects and Daphnia × algae interactions in both the single species (Fig. 3a; Daphnia effect, F = 247; interaction, F = 66.1; both, p < 0.001) and mixed treatments (Fig. 3b; Daphnia effect, F = 128; interaction, F = 34.1; both, p < 0.001). In both the single species and mixed treatments, Ankistrodesmus showed the highest cell counts in the control but the lowest cell counts in the Daphnia treatment (Fig. 3). By the last sampling date, Ankistrodesmus had declined in the Daphnia treatment to values <1 % of its controls in both the single species and mixed treatments, whereas Oocystis in the Daphnia treatments were 29–51 % of control values on the same date (Fig. 3). On the last sampling date, Ankistrodesmus comprised 82.5 ± 3.5 % of the algae in the mixed ungrazed treatment, but only 2.5 ± 0.9 % of the algae in the mixed Daphnia treatment.
We directly compared the performance of the two algal species by calculating mean population growth rates (r, day−1) over the interval from day 6 to the end of the experiment. Over the final 19 days of the experiment, Ankistrodesmus declined sharply in the mixed algae Daphnia treatment (r = −0.24 ± 0.032 day−1) while Oocystis remained abundant and stable in the same microcosms (r = −0.005 ± 0.016 day−1; p < 0.001). In contrast, both populations of both species were relatively stable in the controls, although Ankistrodesmus had a higher growth rate than Oocystis, and this difference was significant in the single species treatment (p < 0.02).
Measurements of Oocystis diameter on days 3 and 20 show that the presence of Daphnia favored larger colony size, and this effect increased over time (two-way ANOVA: Daphnia effect, F = 74.1, p < 0.001; Daphnia × day interaction, F = 12.9, p = 0.002). The magnitude of this grazer effect was, however, small (mean diameters of 12.0 ± 0.25 and 15.2 ± 0.31 μm in the control and Daphnia treatments, respectively, on day 20).
Daphnia juvenile growth assays
Juvenile growth assays provide information on the intensity and mechanism of Daphnia food limitation that supports and complements our analysis of the microcosm populations. An initial experiment with algae from the P-sufficient cultures used to inoculate the microcosms confirmed that juvenile growth is very high with Ankistrodesmus, intermediate with the algal mixture, and lowest but still moderately high with Oocystis (day 0; Fig. 4). Experiments with P-sufficient laboratory cultures starting on days 6, 11, 15, and 20 showed similar high growth rates for Ankistrodesmus (range 0.57–0.61 day−1) and moderately high rates for Oocystis (0.42–0.52 day−1; data not shown).
The effects of mesocosm resources and phosphate supplements on Daphnia juvenile growth (circles) and lipid-ovary index (bars). The day 0 trials used P-sufficient laboratory cultures (laboratory culture). Subsequent experiments used mesocosm resources from the a grazed Ankistrodesmus, b grazed mixture, c grazed Oocystis and d ungrazed Oocystis microcosms. Responses to phosphate supplements (solid circles) relative to controls (open circles) are shown by asterisks (*) for p < 0.05. Growth data are mean ± SE for resources from 3 replicate beakers. The lipid ovary index was scored on a scale of 0–3 and shows overall mean ± SE for both the control and supplemented trials. The value of the lipid-ovary index is zero for experiments starting on days 6 and 11 in (c) and days 6, 11, and 15 in (d)
Weight loss (i.e., negative growth) in the control grazed Oocystis bioassay begun on day 6 (Fig. 4c) corresponds with the beginning of the sharp decline in Daphnia abundance in the Oocystis microcosms, while substantially higher growth in the Ankistrodesmus and mixed control bioassays (Fig. 4a, b) corresponds to the period of slow Daphnia increase in the corresponding microcosms (Fig. 1a). Since algae were abundant in the microcosms in all three algal treatments from days 6–11 (Fig. 2a; POC range 1.2–4.7 C L−1), sharply reduced growth in both the microcosms and the growth assays can be attributed to reduced food quality, not low quantity.
In four growth assay experiments (excluding day 0), the overall mean Daphnia control growth rate was 0.10 ± 0.06 day−1 for resources from the grazed Oocystis microcosms and 0.05 ± 0.04 day−1 for the ungrazed (control) microcosms (Fig. 4c, d). Thus, there is no evidence that the strong defenses of P-deficient Oocystis were induced by Daphnia’s presence.
Responses to phosphate supplements suggest that P-limitation reduced Daphnia growth in both Ankistrodesmus and mixed treatments in the growth bioassays begun on day 6 (Fig. 4a, b). P-differentials were positive for the Ankistrodesmus and mixed treatments for the final 3 experiments, but were not statistically significant, even though the resource C:P ratio ranged from 980 to 600.
Daphnia showed no statistically significant responses to P supplements in any of the trials with resources from the grazed or ungrazed Oocystis microcosms (Fig. 4c, d). Moreover, the mean values for mesocosm resources were higher than the mean P-supplement values in 4 of the 8 comparisons. Resource C:P ratios remained above 900 in all of the Oocystis microcosms.
Based on POC and PP estimates from the microcosms, and assuming complete uptake of the phosphate supplements, the resource C:P ratios were reduced (ranges of means for experiments) from 285–1,000 (control) to 32–310 (phosphate supplement) for Ankistrodesmus; from 615–1,040 to 180–350 for the mixed treatment; from 960–1,830 to 215–490 for grazed Oocystis and from 940–1,360 to 290–545 for control Oocystis. Thus, due to the extreme algal P-limitation, the phosphate supplements strongly reduced but did not eliminate the potential for Daphnia P-limitation.
The lipid–ovary index provides further evidence of differences in the mechanism of food limitation between the Oocystis and the Ankistrodesmus and mixed treatments. For resources from both the grazed and ungrazed Oocystis microcosms, zero lipid values from experiments begun on days 6 and 11 suggest extreme energy limitation (Fig. 4c, d). In contrast, maximal lipid values for the Ankistrodesmus and mixed treatments on the same dates are consistent with P-limitation (Fig. 4a, b). The shift in the Ankistrodesmus assays from maximal to moderate lipid reserves between days 15 and 20 occurred as the food concentration decline from 1.3 to 0.6 mg C L−1 and the resource C:P ratio declined from 970 to 600.
Growth assays using resources from the ungrazed Ankistrodesmus and Oocystis microcosms at the end of the experiment provide further evidence for differences in the mechanism of poor food quality for P-limited cultures of these two taxa. First, growing P-deficient cultures in the light for 3 days with P-rich WC algal medium fully restored food quality for both algal species (laboratory culture vs. WC light; Fig. 5). Comparisons between the two WC treatments (light vs. dark) support our hypothesis that phosphorus, light, and enough time for cell division are needed to reduce the nutrient-dependent defenses of Oocystis to the baseline constitutive level (Fig. 5b). In contrast to Oocystis, growing Ankistrodesmus with the P-rich WC medium in the dark for 3 days improved Daphnia growth to almost the same level as the laboratory culture control, supporting the P-limitation hypothesis (Fig. 5a). However, daily phosphate supplements in the dark (+PO −34 ) only partially ameliorated the poor food quality of strongly P-deficient Ankistrodesmus (P-), suggesting that unknown factors may also play a role in poor food quality. As in the previous growth assays (Fig. 4), short-term supplementation of P-deficient Oocystis with phosphate did not improve Daphnia growth.
Daphnia juvenile growth assays with 1.0 mg C L−1 of algae from P-sufficient laboratory cultures (laboratory culture) and from the ungrazed microcosms of a Ankistrodesmus and b Oocystis. Resources from microcosms were incubated for 3 days in the light without nutrients or with WC algal medium in light (WC light) or dark (WC dark). The mesocosm resources without WC medium were supplemented with phosphate each day of the growth experiment (+PO −34 ) or left untreated (P−). Treatments not connected by lines are significantly different (Holm–Sidak procedure, p < 0.05). The C:P ratio (shown above each bar) was calculated from the concentration of added phosphate (numbers in parentheses) or measured from 3 replicate filters
Daphnia feeding in all five Ankistrodesmus treatments were rich in lipids suggesting that energy limitation was weak or absent in each treatment (Table 1). In contrast, Daphnia feeding on Oocystis showed moderate lipid levels when feeding on cultures rapidly growth in the light (laboratory and WC + light) and low lipids when feeding on the P-deficient culture (P−) or P-enriched cultures kept in the dark (WC dark and +PO −34 ). Thus, as in the previous series of growth assays (Fig. 4), Daphnia feeding on P-deficient Oocystis showed no evidence of P-limitation.
We conducted 47 juvenile growth assays with P-rich laboratory cultures and P-deficient and phosphate supplemented resources from the Ankistrodesmus and Oocystis microcosms. Based on median values, Ankistrodesmus was a better resource than Oocystis with both P-rich laboratory cultures (0.58 vs. 0.40 day−1) and P-deficient (0.13 vs. 0.10 day−1) microcosm resources. However, the two resources differed qualitatively in grazer responses to phosphate supplements in the dark (medians: Ankistrodesmus, 0.29 day−1; Oocystis, 0.06 day−1). Direct P-limitation explains part of the poor quality of P-Ankistrodesmus (36 % of the difference between medians of P-rich laboratory cultures and P-deficient microcosms resources), while there is no evidence of P-limitation for juvenile Daphnia feeding on strongly P-deficient Oocystis (−13 % of the difference between P-rich laboratory cultures and P-deficient microcosm resources).
Discussion
This study provides evidence that P-limitation enhances the defenses of a digestion-resistant alga, favoring high abundance of strongly defended algae and energy limitation of Daphnia growth. Although earlier studies showed nutrient-dependent digestion defenses in green algae that were undefended, high quality resources under nutrient-sufficient conditions (Van Donk and Hessen 1993; Van Donk et al. 1997), this study is the first to show that P-limitation enhances the defenses of an alga with constitutive digestion defenses. Algal dynamics provide a marked contrast between strong defenses in P-deficient Oocystis and weak, ineffective defenses in P-limited Ankistrodesmus. Thus, we found very strong differences between two species of green algae in the degree of defense enhancement under P-limitation.
Daphnia population dynamics in the microcosms indicate that food quality was much lower in the Oocystis treatment. Juvenile growth assays indicate that our strain of Oocystis is a good, although suboptimal, resource when grown under P-sufficient conditions (median growth assay 0.40 day−1), but a very poor quality food when P-deficient (0.10 day−1). Using the same Daphnia clone, DeMott et al. (2010) found that juvenile growth rate was tightly correlated with juvenile carbon assimilation efficiency (AE; r 2 = 0.96) among seven taxa of green algae that varied in digestion defenses. Viable gut passage in the digestion-resistant taxa implies that the cell wall is not breached and that neither C nor P is assimilated. Mean Daphnia juvenile growth rate on high concentrations of the P-sufficient algae ranged from 0.57 to 0.61 for three undefended taxa and from 0.10 to 0.46 day−1 for four digestion-resistant taxa, including two strains of Oocystis. Thus, the strain of Oocystis used in the microcosms, when grown under a range of nutrient conditions, spans nearly the full range in food quality of the four digestion-resistant algae studied by DeMott et al. (2010).
Using the AE versus growth relationship from DeMott et al. (2010), juvenile Daphnia feeding on the P-sufficient Oocystis in the growth assays are predicted to assimilate carbon with 41 % efficiency, while the same strain under P-deficient conditions would be assimilated with only 7 % efficiency. Clearly, such a low AE is consistent with a high frequency of viable gut passage and a strongly enhanced digestion defense. Thus, application of TER models to algae with strong, nutrient-dependent digestion defenses may require C and P assimilation efficiencies <0.1.
Although we did not directly verify energy limitation in Daphnia feeding on Oocystis, our results are contrary to three important alternatives to the nutrient-dependent digestion-resistance hypothesis. First and most important, juvenile growth assays with phosphate supplements provide no evidence for even weak P-limitation of Daphnia feeding on strongly P-deficient Oocystis from the microcosms. Since phosphate supplements can saturate C:P ratios of P-deficient algae within minutes, growth assays with phosphate supplements are the “gold standard” for demonstrating zooplankton P-limitation (DeMott 1998; Plath and Boersma 2001). We hypothesized that light, added phosphate, and sufficient time for cell division would be needed to reduce the nutrient-dependent digestion defenses of P-deficient Oocystis back to the baseline constitutive level, and this hypothesis was supported in our final series of growth assays. The lack of evidence for P-limitation in Daphnia feeding on Oocystis is strengthened by the positive growth responses for Daphnia feeding on P-deficient Ankistrodesmus with phosphate supplements. The very low lipid reserves of Daphnia feeding on P-deficient Oocystis are also consistent with strong energy limitation and contrast with the high lipid reserves of P-limited Daphnia feeding on high concentrations of P-deficient Ankistrodesmus. The phosphate supplements substantially reduced resource C:P ratios, in some, but not all cases, to below nominal TER values. On the other hand, while population dynamics in the microcosms included transgenerational effects of P-deficient resources (Frost et al. 2010), most growth assays, including ours, did not.
Since P-deficient Oocystis from both the grazed and control microcosms were very poor resources, we conclude that algal P-limitation, rather than a grazer-induced defense, was responsible for the extremely poor quality of the P-deficient algae in the grazed Oocystis microcosms. We cannot rule out the possibility, however, that Oocystis might show an induced digestion defense when grown in the presence of grazers under P-sufficient conditions. Kampe et al. (2007) showed that the presence of Daphnia caused a shift toward large, inedible colonies in the gelatinous green alga Sphaerocystis. We also found that Daphnia caused an increase in Oocystis colony size. However, the increase from 12 to 15 μm in diameter for Oocystis compares to an increase from 8–20 to 30–200 μm observed in Sphaerocystis. Based on a gut content study, DeMott (1995) found that small (1 mm) Daphnia readily ingested a spherical gelatinous green alga (Eurdorina) up to 40 μm in diameter, while large (2 mm) Daphnia showed an upper limit of about 80 μm. Thus, for Daphnia feeding on P-deficient Oocystis, our results are contrary to Daphnia P-limitation, while we show that Oocystis from both the grazed and ungrazed mesocosms were readily ingested even by small Daphnia. Although the P-deficient Oocystis was clearly a poor resource for both adults and juveniles, the evidence for nearly complete juvenile mortality in the Oocystis treatment is consistent with the juvenile bottleneck hypothesis (DeMott et al. 2010).
Laboratory studies testing for Daphnia P-limitation typically have used P-sufficient and P-deficient cultures of a single, readily ingested and digested, undefended green alga. This approach controls for taxonomic variation while maximizing the chance of finding evidence for zooplankton P-limitation. However, as our study shows, the results may be misleading, because natural algal assemblages include many species that vary in maximal growth rate, competitive ability, and defenses against grazers. While there are no comparable laboratory studies, our results are very similar to those of DeMott and Tessier (2002), who studied deep, stratified Michigan lakes with high densities of large-bodied Daphnia that coexisted with high abundance of P-limited digestion-resistant algae during summer (Tessier and Woodruff 2002).
Throughout the microcosm experiment, Daphnia abundance and reproduction were very similar in the mixed and the Ankistrodesmus treatments. This suggests that Daphnia mainly responded to the abundance of Ankistrodesmus and that Oocystis in the mixed treatment had very little positive or negative, interfering effect. Notably, Daphnia in both the mixed and Ankistrodesmus treatments showed evidence of similar, very strong energy limitation by the end of the experiment, when Ankistrodesmus had declined to low densities in the grazed Ankistrodesmus and the grazed mixed treatment mesocosms. Thus, in all three grazed treatments, the mesocosms approached equilibria with strongly energy-limited Daphnia. Ironically, by the end of the experiment, algal P-limitation was strongest in the Oocystis and mixed treatments where it acted to enhance Oocystis’ defenses. The rapid declines of Ankistrodesmus in the single species and mixed treatments clearly depended on high Daphnia density and strong grazing.
With only a few exceptions, stoichiometric models have assumed homogeneous, undefended resources (reviewed by Andersen et al. 2004; Moe et al. 2005). One effect of including inedible (Hall et al. 2006) or digestion-resistant (Hall et al. 2007) algae in stoichiometrically explicit food chains is to elevate and prolong algal nutrient limitation. This occurs because inedible and well-defended algae remain abundant despite intense grazing. Supporting these predictions, algal densities and resource C:P ratios in the Daphnia microcosms were highest in the Oocystis treatment and lowest in the Ankistrodesmus treatment. The theoretical models also predict that predation on grazers increases grazer and algal nutrient limitation by reducing grazing intensity (Hall et al. 2007). Since Hall et al. (2007) assumed that digestion resistance is a fixed trait, our finding that digestion resistance is enhanced by nutrient limitation adds a new dimension for theoretical analysis and empirical study.
In agreement with other empirical studies (reviewed by Agrawal 1998), our results suggest a tradeoff between algal defenses and maximal growth rate that is consistent with theory on how algal defenses help account for the dynamic outcome of interactions between multi-species assemblages of phytoplankton and their grazers (Grover 1995; Grover and Holt 1998). Stronger evidence for an evolutionary tradeoff would require comparisons between closely related taxa, such as different strains of Oocystis (Yoshida et al. 2004). Under our experimental conditions, maximal growth rate was only a factor during the first few days, before the onset of severe nutrient limitation. However, phytoplankton can experience prolonged periods of near-maximal cell division when intense grazing keeps populations at low levels. Such conditions may favor fast-growing undefended algae over slower-growing, well-defended species (Grover and Holt 1998; Sarnelle 2005). Low food abundance and larger grazer body size also reduce the effectiveness of digestion defenses by slowing gut passage (DeMott et al. 2010). Thus, understanding the costs and benefits of defense is very important for predicting when well-defended algae will dominate phytoplankton assemblages. Our results show that stoichiometric theory needs to be better integrated with theory on algal defenses. Such a theory will need to consider stoichiometric constraints together with the costs of defense, the enhancement of digestion defenses with nutrient limitation, and the effects of food concentration and grazer body size in modulating the effectiveness of digestion defenses.
References
Agrawal AA (1998) Algal defenses, grazers and their interactions in aquatic trophic cascades. Acta Oecol 19:331–337
Andersen T, Elser JJ, Hessen DO (2004) Stoichiometry and population dynamics. Ecol Lett 7:884–900
Anderson TR, Hessen DO (2005) Threshold elemental ratios for carbon versus phosphorus limitation in Daphnia. Freshw Biol 50:2063–2075
Boersma M (2000) The nutritional quality of P-limited alga for Daphnia. Limnol Oceanogr 45:1157–1161
De Senerpont Domis LN, Mooij WM, Huisman J (2007) Climate-induced shifts in an experimental phytoplankton community: a mechanistic approach. Hydrobiologia 584:403–413
DeMott WR (1995) The influence of prey hardness on Daphnia’s selectivity for large prey. Hydrobiologia 307:127–138
DeMott WR (1998) Utilization of a cyanobacterium and a phosphorous-deficient green alga as complementary resources by daphnids. Ecology 79:2463–2481
DeMott WR, Gulati RD (1999) Phosphorus limitation in Daphnia: evidence from a long term study of three hypereutrophic Dutch lakes. Limnol Oceanogr 44:1557–1564
DeMott WR, Pape BJ (2004) Stoichiometry in an ecological context: testing for links between Daphnia P-content, growth rate and habitat preference. Oecologia 142:20–27
DeMott WR, Tessier AJ (2002) Stoichiometric constraints vs. algal defenses: testing mechanisms of zooplankton food limitation. Ecology 83:3426–3433
DeMott WR, Gulati RD, Siewertsen K (1998) Effects of phosphorus-deficient diets on the carbon and phosphorus balance of Daphnia magna. Limnol Oceanogr 43:1147–1161
DeMott WR, Gulati RD, Van Donk E (2001) Effects of dietary phosphorus deficiency on the abundance, phosphorus balance and growth of Daphnia cucullata in three hypereutrophic Dutch lakes. Limnol Oceanogr 46:1871–1880
DeMott WR, Edington JR, Tessier AJ (2004) Testing zooplankton food limitation across gradients of depth and productivity in small stratified lakes. Limnol Oceanogr 49:1408–1416
DeMott WR, McKinney EN, Tessier AJ (2010) Ontogeny of digestion in Daphnia: implications for the effectiveness of algal defenses. Ecology 91:540–548
Elser JJ, Fagan WF, Denno RF et al (2000) Nutritional constraints in terrestrial and freshwater food webs. Nature 408:578–580
Elser JJ, Hayakawa K, Urabe J (2001) Nutrient limitation reduces food quality for zooplankton: Daphnia response to seston phosphorus enrichment. Ecology 82:898–903
Ferrao-Filho AS, Tessier AJ, DeMott WR (2007) Sensitivity of herbivorous zooplankton to phosphorus-deficient diets: testing stoichiometric theory and the growth rate hypothesis. Limnol Oceanogr 52:40–415
Fileto C, Arcifa MS, Silva LHS (2007) Testing mineral quality for tropical cladocerans from a shallow Brazilian lake. Acta Limnol Bras 19:257–271
Frost PC, Ebert D, Larson JH, Marcus MA, Wagner ND, Zalewski A (2010) Transgenerational effects of poor elemental food quality on Daphnia magna. Oecologia 162:865–872
Grover JP (1995) Competition, herbivory, and enrichment: nutrient-based models for edible and inedible plants. Am Nat 145:746–774
Grover JP, Holt RD (1998) Disentangling resource and apparent competition: realistic models for plant-herbivore communities. J Theor Biol 191:353–376
Guillard RR, Lorenzen CJ (1972) Yellow-green algae with chlorophyllide C. J Phycol 8:10–14
Hall SR, Leibold MA, Lytle DA, Smith VH (2006) Inedible producers in food webs: controls on stoichiometric food quality and composition of grazers. Am Nat 167:628–637
Hall SR, Shurin JB, Diehl S, Nisbet RM (2007) Food quality, nutrient limitation of secondary production and the strength of trophic cascades. Oikos 116:1128–1143
Kampe H, König-Rinke M, Petzoldt T, Benndorf J (2007) Direct effects of Daphnia-grazing, not infochemicals, mediate a shift towards large inedible colonies of the gelatinous green alga Sphaerocystis schroeteri. Limnologica 37:137–145
Kerfoot WC, Levitan C, DeMott WR (1988) Daphnia-phytoplankton interactions: density dependent shifts in resource quality. Ecology 69:1806–1825
Kilham SS, Kreeger DA, Lynn SG, Goulden CE, Herrera L (1998) COMBO: a defined freshwater culture medium for algae and zooplankton. Hydrobiologia 377:147–159
Makino W, Urabe J, Elser JJ (2002) Evidence of phosphorus-limited individual and population growth of Daphnia in a Canadian shield lake. Oikos 96:197–205
Moe SJ, Stelzer RS, Forman MR, Harpole WS, Daufresne T, Yoshida T (2005) Recent advances in ecological stoichiometry: insights for population and community ecology. Oikos 109:29–39
Müller-Navarra D (1995) Biochemical versus mineral limitation in Daphnia. Limnol Oceanogr 40:1209–1214
Murphy J, Riley JP (1962) A modified single solution for determination of phosphate in natural waters. Anal Chim Acta 27:31–36
Nelson WA, McCauley E, Wrona FJ (2001) Multiple dynamics in a single predator-prey system: experimental effects of food quality. Proc R Soc Lond B 268:1223–1230
Plath K, Boersma M (2001) Mineral limitation of zooplankton: stoichiometric constraints and optimal foraging. Ecology 82:1260–1269
Porter KG (1973) Selective grazing and differential digestion of algae by zooplankton. Nature 244:179–180
Ravet JL, Brett MT, Arhonditsis GB (2010) The effects of seston lipids on zooplankton fatty acid composition in Lake Washington, Washington, USA. Ecology 91:180–190
Sarnelle O (2005) Daphnia as keystone predators: effects on phytoplankton diversity and grazing resistance. J Plankton Res 27:1229–1238
Sommer U (1992) Phosphorus-limited Daphnia: intraspecific facilitation instead of competition. Limnol Oceanogr 37:966–973
Stamp N (2003) Out of the quagmire of plant defense hypotheses. Q Rev Biol 78:23–55
Sterner RW (1989) The role of grazers in phytoplankton succession. In: Sommer U (ed) Plankton ecology: succession in plankton communities. Springer, Berlin, pp 107–170
Sterner RW, Elser JJ (2002) Ecological stoichiometry: the biology of elements from molecules to the biosphere. Princeton University Press, Princeton
Sterner RW, Schulz KL (1998) Zooplankton nutrition: recent progress and a reality check. Aquat Ecol 32:261–279
Sterner RW, Hagenmeier DD, Smith RF, Smith WL (1992) Lipid-ovary indices in food limited Daphnia. J Plankton Res 14:1449–1460
Tessier AJ, Woodruff P (2002) Cryptic trophic cascade along a gradient of lake size. Ecology 83:1263–1270
Tessier AJ, Henry LL, Goulden CE, Durand MW (1983) Starvation in Daphnia: energy reserves and reproductive allocation. Limnol Oceanogr 28:667–676
Urabe J, Elser JJ, Kyle M, Yoshida TS, Kawabata Z (2002) Herbivorous animals can mitigate unfavorable ratios of energy and material supplies by enhancing nutrient recycling. Ecol Lett 5:177–185
Van Donk E, Hessen DO (1993) Grazing resistance in nutrient-stressed phytoplankton. Oecologia 93:508–511
Van Donk E, Lürling M, Hessen DO, Lokhorst GM (1997) Altered cell wall morphology in nutrient-deficient phytoplankton and its impact on grazers. Limnol Oceanogr 42:357–364
Van Donk E, Hessen DO, Verschoor AM, Gulati RD (2008) Re-oligotrophication by phosphorus reduction and effects on seston quality in lakes. Limnologica 38:189–202
Van Donk E, Ianora A, Vos M (2011) Induced defenses in marine and freshwater phytoplankton: a review. Hydrobiologia 668:3–19
Van Geest GJ, Spierenburg P, Van Donk E, Hessen DO (2007) Daphnia growth rates in arctic ponds: limitation by nutrients or carbon? Polar Biol 30:235–242
Vanni MJ (1997) Effects of nutrients and zooplankton size on the structure of a phytoplankton community. Ecology 68:624–635
Yoshida T, Hairston NG Jr, Ellner SP (2004) Evolutionary trade-off between defense against grazing and competitive ability in a simple unicellular alga, Chlorella vulgaris. Proc R Soc Lond B 271:1947–1953
Acknowledgments
We thank Nico Helmsing, Suzanne Wiezer and Peg DeMott for technical assistance. A.J. Tessier, S.R. Hall and three anonymous reviewers provided helpful comments on drafts of the manuscript. W.R.D. was supported by a sabbatical from Indiana–Purdue University and a fellowship from the Royal Dutch Academy of Sciences. This research was carried out in compliance with the laws of The Netherlands.
Open Access
This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Ulrich Sommer.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
DeMott, W.R., Van Donk, E. Strong interactions between stoichiometric constraints and algal defenses: evidence from population dynamics of Daphnia and algae in phosphorus-limited microcosms. Oecologia 171, 175–186 (2013). https://doi.org/10.1007/s00442-012-2404-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-012-2404-y