Abstract
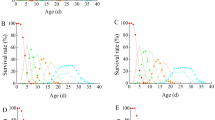
It has recently been suggested that the expression of parasite virulence depends on host population density, such that infected hosts have a higher sensitivity to density, and thus reach their carrying capacity earlier than uninfected hosts. In this scenario, parasite-induced reduction in fitness (i.e., virulence) increases with host density. We tested this hypothesis experimentally, using outdoor mesocosm populations of Daphnia magna infected by the microsporidian Octosporea bayeri. Contrary to the prediction, virulence was independent of host density. In a competition experiment with initial prevalence of 50%, O. bayeri reduced the competitive ability of infected Daphnia within the asexual growth phase independent of initial host population density. In an additional experiment we set up populations with 100% and 0% prevalence and followed their population dynamics over the whole season. Consistent with the competition experiment, we found no difference in population dynamics within the asexual growth phase of the host, suggesting that infected hosts are not more sensitive to density than uninfected hosts. The additional experiment, however, included more than the initial growth phase as did the competition experiment. Eventually, after 100 days, 100% infected populations assumed a reduced carrying capacity compared to uninfected populations. We identify and discuss three reasons for the discrepancy between our experiment and the predictions.



Similar content being viewed by others
References
Altermatt F, Ebert D (2007) Thegenotype specific competitive ability does not correlate with infection in natural Daphnia magna populations. PLoS ONE 2:e1280
Altermatt F, Hottinger J, Ebert D (2007) Parasites promote host gene flow in a metapopulation. Evol Ecol 21:561–575
Anderson RM (1982a) Population dynamics of infectious diseases: theory and applications. Chapman and Hall, London
Anderson RM (1982b) Theoretical basis for the use of pathogens as biological control agents of pest species. Parasitology 84:3–33
Anderson RM, Gordon DM (1982) Processes influencing the distribution of parasite numbers within host populations with special emphasis on parasite-induced host mortalities. Parasitology 85:373–398
Anderson RM, May RM (1981) The population dynamics of microparasites and their invertebrate hosts. Philos Trans R Soc Lond B Biol Sci 291:451–524
Bedhomme S, Agnew P, Vital Y, Sidobre C, Michalakis Y (2005) Prevalence-dependent costs of parasite virulence. PLoS Biol 3:1403–1408
Bell T, Freckelton RP, Lewis OT (2006) Plant pathogens drive density-dependent seedling mortality in a tropical tree. Ecol Lett 9:569–574
Bittner K, Rothaupt KO, Ebert D (2002) Ecological interactions of the microparasite Caullerya mesnili and its host Daphnia galeata. Limnol Oceanogr 47:300–305
Brown M, Loosli R, Schmid-Hempel P (2000) Condition dependent expression of virulence in a Trypanosome infecting bumbelbees. Oikos 91:421–427
Bull JJ (1994) Perspective: virulence. Evolution 48:1423–1437
Carius HJ, Little TJ, Ebert D (2001) Genetic variation in a host–parasite association: potential for coevolution and frequency-dependent selection. Evolution 55:911–921
Cole LC (1954) The population consequences of life-history phenomena. Q Rev Biol 29:103–137
Day T, Burns JG (2003) A consideration of patterns of virulence arising from host–parasite co-evolution. Evolution 57:671–676
Ebert D (1994) Virulence and local adaptation of a horizontally transmitted parasite. Science 265:1084–1086
Ebert D (2005) Ecology, epidemiology, and evolution of parasitism in Daphnia. National Library of Medicine (US), National Center for Biotechnology Information, Bethesda. Available from: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=Books
Ebert D, Bull J (2008) The evolution and expression of virulence. In: Stearns SC, Koella JC (eds) Evolution in health and disease. Oxford University Press, Oxford, pp 153–154
Ebert D, Hamilton WD (1996) Sex against virulence: the coevolution of parasitic diseases. Trends Ecol Evol 11:79–81
Ebert D, Zschokke-Rohringer CD, Carius HJ (1998) Within- and between-population variation for resistance of Daphnia magna to the bacterial endoparasite Pasteuria ramosa. Proc R Soc Lond B Biol Sci 265:2127–2134
Ebert D, Lipsitch M, Mangin KL (2000) The effect of parasites on host population density and extinction: experimental epidemiology with Daphnia and six microparasites. Am Nat 156:459–477
Ebert D, Hottinger JW, Pajunen VI (2001) Temporal and spatial dynamics of parasites in a Daphnia metapopulation: which factors explain parasite richness? Ecology 82:3417–3434
Ebert D, Carius HJ, Little T, Decaestecker E (2004) The evolution of virulence when parasites cause host castration and gigantism. Am Nat 164:19–32
Ewald PW (1983) Host–parasite relations, vectors, and the evolution of disease severity. Annu Rev Ecol Syst 14:456–485
Haag CR, Riek M, Hottinger W, Pajunen VI, Ebert D (2006) Founder events as determinants of within-island and among-island genetic structure of Daphnia metapopulations. Heredity 96:150–158
Hartl D, Clark A (1997) Principles of population genetics, 3rd edn. Sinauer, Sunderland
Hanski I, Ranta E (1983) Coexistence in a patchy environment: three species of Daphnia in rock pools. J Anim Ecol 52:263-279
Hudson PJ, Dobson AP (1997) Host–parasite processes and demographic consequences. Oxford University Press, New York
Hudson PJ, Newborn D, Dobson AP (1992) Regulation and stability of a free-living host–parasite system: Trichostrongylus tenuis in red grouse. I. Monitoring and parasite reduction experiments. J Anim Ecol 61:477–486
Jokela J, Taskinen J, Mutikainen P, Kopp K (2005) Virulence of parasites in hosts under environmental stress: experiments with anoxia and starvation. Oikos 108:156–164
Jollens AE, Cooper DV, Levin SA (2005) Hidden effects of chronic tuberculosis in African buffalo. Ecology 86:2258–2264
Klüttgen B, Dülmer U, Engels M, Ratte HT (1994) ADaM, an artificial freshwater for the culture of zooplankton. Water Res 28:743–746
Lass S, Ebert D (2006) Apparent seasonality of parasite dynamics: analysis of cyclic prevalence patterns. Proc R Soc Lond B Biol Sci 273:199–206
Lively CM (2006) The ecology of virulence. Ecol Lett 9:1089–1095
Mackinnon MJ, Gaffney D, Read A (2002) Virulence in rodent malaria: host genotype by parasite genotype interactions. Infect Genet Evol 1:287–296
May RM, Anderson RM (1983) Epidemiology and genetics in the coevolution of parasites and hosts. Proc R Soc Lond B Biol Sci 219:281–313
Pajunen VI (1986) Distributional dynamics of Daphnia species in a rock-pool environment. Ann Zool Fenn 23:131-140
Pajunen VI, Pajunen I (2003) Long-term dynamics in rock pool Daphnia metapopulations. Ecography 26:731–738
Perlmann SJ, Jaenike J (2003) Evolution of multiple components of virulence in Drosophila-nematode associations. Evolution 57:1543–1551
Pulkkinen K, Ebert D (2004) Host starvation decreases parasite load and mean host size in experimental populations. Ecology 85:823–833
R Development Core Team (2006) R: a language and environment for statistical computing. R Foundation for Statistical Computing. Vienna
Ranta E (1979) Niche of Daphnia species in rock pools. Arch Hydrobiol 87:205–223
Read AF (1994) The evolution of virulence. Trends Microbiol 2:73–76
Roth O, Ebert D, Vizoso DB, Bieger A, Lass S (2008) Male-biased sex-ratio distortion caused by Octosporea bayeri, a vertically and horizontally-transmitted parasite of Daphnia magna. Int J Parasitol 38:969–979
Seppälä O, Liljeroos K, Karvonen A, Jokela J (2008) Host condition as a constraint for parasite reproduction. Oikos 117:749–753
Stearns SJ (1976) Life-history tactics: a review of the ideas. Q Rev Biol 51:3–47
Vizoso D, Ebert D (2004) Within-host dynamics of a microsporidium with horizontal and vertical transmission: Octosporea bayeri in Daphnia magna. Parasitology 128:31–38
Vizoso D, Ebert D (2005) Phenotypic plasticity of host–parasite interactions in response to the route of infection. J Evol Biol 18:911–921
Vizoso D, Lass S, Ebert D (2005) Different mechanisms of transmission of the microsporidium Octosporea bayeri: a cocktail of solutions for the problem of parasite permanence. Parasitology 130:501–509
Acknowledgements
We thank Curt Lively and an anonymous reviewer for their comments improving this manuscript. Thanks to Florian Altermatt and Thomas Zumbrunn for ideas and help during the whole project. We also thank David Duneau and Dita Vizoso for helpful comments on the manuscript and Suzanne Zweizig for improving the English. A. B. was supported by the Werenfels fonds (FAG) and the Anna Caroline Stiftung. The study was supported by the Swiss National Science Foundation. This work is part of project no. 97524006 at Tvärminne Zoological Station. This research was carried out in compliance with the current laws of Finland governing ethical conduct and the use of animals in research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by R. Brandl.
Rights and permissions
About this article
Cite this article
Bieger, A., Ebert, D. Expression of parasite virulence at different host population densities under natural conditions. Oecologia 160, 247–255 (2009). https://doi.org/10.1007/s00442-009-1297-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-009-1297-x