Abstract
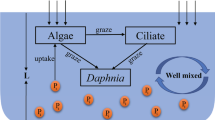
Realistic functional responses are required for accurate model predictions at the community level. However, controversy remains regarding which types of dependencies need to be included in functional response models. Several studies have shown an effect of very high predator densities on per capita predation rates, but it is unclear whether this predator dependence is also important at low predator densities. We fit integrated functional response models to predation data from 4-h experiments where we had varied both predator and prey densities. Using an information theoretic approach we show that the best-fit model includes moderate predator dependence, which was equally strong even at low predator densities. The best fits of Beddington–DeAngelis and Arditi–Akçakaya functional responses were closely followed by the fit of the Arditi–Ginzburg model. A Holling type III functional response did not describe the data well. In addition, independent behavioral observations revealed high encounter rates between predators. We quantified the number of encounters between predators and the time the focal predator spent interacting with other individuals per encounter. This time “wasted” on conspecifics reduced the total time available for foraging and may therefore account for lower predation rates at higher predator densities. Our findings imply that ecological theory needs to take realistic levels of predator dependence into account.




Similar content being viewed by others
References
Abrams PA (1994) The fallacies of ratio-dependent predation. Ecology 75:1842–1850
Abrams PA, Ginzburg LR (2000) The nature of predation: prey dependent, ratio dependent or neither? Trends Ecol Evol 15:337–341
Altwegg R, Eng M, Caspersen S, Anholt BR (2006) Functional response and prey defence level in an experimental predator–prey system. Evol Ecol Res 8:115–128
Anholt BR, Werner EE (1995) Interaction between food availability and predation mortality mediated by activity. Ecology 76:2230–2234
Arditi R, Ginzburg LR (1989) Coupling in predator prey dynamics: ratio-dependence. J Theor Biol 139:311–326
Arditi R, Akçakaya HR (1990) Underestimation of mutual interference of predators. Oecologia 83:358–361
Arditi R, Saïah H (1992) Empirical evidence of the role of heterogeneity in ratio-dependent consumption. Ecology 73:1544–1551
Arditi R, Callois JM, Tyutyunov Y, Jost C (2004) Does mutual interference always stabilize predator–prey dynamics? A comparison of models. C R Biol 327:1037–1105
Beddington JR (1975) Mutual interference between parasites or predators and its effect on search efficiency. J Anim Ecol 44:331–340
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach. Springer, New York
Cosner C, DeAngelis DL, Ault JS, Olson DB (1999) Effects of spatial grouping on the functional response of predators. Theor Popul Biol 56:65–75
Crowley PH, Martin EK (1989) Functional responses and interference within and between year classes of a dragonfly population. J North Am Benthol Soc 8:211–221
Crowley PH, Dillon PM, Johnson DM, Watson CN (1987) Intraspecific interference among larvae in a semivoltine dragonfly population. Oecologia 71:447–456
Crumrine PW (2005) Size structure and substitutability in an odonate intraguild predation. Oecologia 145:132–139
DeAngelis DL, Goldstein RA, O’Neill RV (1975) A model for trophic interaction. Ecology 56:881–892
Fussmann GF, Weithoff G, Yoshida T (2005) A direct, experimental test of resource vs. consumer dependence. Ecology 86:2924–2930
Fussmann GF, Weithoff G, Yoshida T (2007) A direct, experimental test of resource vs. consumer dependence: reply. Ecology 88:1603–1604
Grabowski JH (2004) Habitat complexity disrupts predator–prey interactions but not the trophic cascade on oyster reefs. Ecology 85:995–1004
Griffen BD, Delaney DG (2007) Species invasion shifts the importance of predator dependence. Ecology 88:3012–3021
Gross T, Ebenhöh W, Feudel U (2004) Enrichment and food chain stability: the impact of different forms of predator–prey interactions. J Theor Biol 227:349–358
Hansson S, DeStasio BT, Gorokhova E, Mohammadian MA (2001) Ratio-dependent functional responses—tests with the zooplanktivore Mysis mixta. Mar Ecol Prog Ser 216:181–189
Hayward RS, Gallup DN (1976) Feeding, filtering and assimilation in Daphnia schoedleri Sars as affected by environmental conditions. Arch Hydrobiol 77:139–163
Helgen JC (1987) Feeding rate inhibition in crowded Daphnia pulex. Hydrobiologia 154:113–119
Hillebrand H, Cardinale BJ (2004) Consumer effects decline with prey diversity. Ecol Lett 7:192–201
Holling CS (1959) The components of predation as revealed by a study of small-mammal predation of the European pine sawfly. Can Entomol 91:293–320
Holling CS (1966) The functional response of invertebrate predators to prey density. Mem Entomol Soc Can 48:1–87
Jensen CXJ, Jeschke JM, Ginzburg LR (2007) A direct, experimental test of resource vs consumer dependence: comment. Ecology 88:1600–1602
Jost C, Arditi R (2000) Identifying predator–prey processes from time-series. Theor Popul Biol 57:325–337
Jost C, Arditi R (2001) From patterns to process: identifying predator–prey models from time-series data. Popul Ecol 43:229–243
Jost C, Ellner SP (2000) Testing for predator-dependence in predator–prey dynamics: a non-parametric approach. Proc R Soc B 267:1611–1620
Jost C, Devulder G, Vucetich JA, Peterson RO, Arditi R (2005) The wolves of Isle Royale display scale-invariant satiation and ratio-dependent predation on moose. J Anim Ecol 74:809–816
Juliano SA (2001) Non-linear curve fitting: predation and functional response curves. In: Scheiner SM, Gurevitch J (eds) Design and analysis of ecological experiments. Oxford University Press, Oxford, pp 178–196
Kolasa J (2001) Flatworms: Turbellaria and Nemertea. In: Thorp JH, Covich AP (eds) Ecology and classification of North American freshwater invertebrates. Academic Press, London, pp 155–180
Kratina P, Vos M, Anholt BR (2007) Species diversity modulates predation. Ecology 88:1917–1923
Kusch J (1993) Behavioural and morphological changes in ciliates induced by the predator Amoeba proteus. Oecologia 96:354–359
Lürling M, Roozen F, Van Donk E, Goser B (2003) Response of Daphnia to substances released from crowded congeners and conspecifics. J Plankton Res 25:967–978
May RM (1973) Stability and complexity in model ecosystems. Monographs in population biology, vol 6. Princeton University Press, Princeton
McCann K, Rasmussen J, Umbanhowar J (2005) The dynamics of spatially coupled food webs. Ecol Lett 8:513–523
Mills NJ, Lacan I (2004) Ratio dependence in the functional response of insect parasitoids: evidence from Trichogramma minutum foraging for eggs in small host patches. Ecol Entomol 29:208–216
Oaten A, Murdoch WW (1975) Functional response and stability in predator–prey systems. Am Nat 109:289–298
Osenberg CW, Sarnelle O, Cooper SD, Holt RD (1999) Resolving ecological questions through meta-analysis: goals, metrics, and models. Ecology 80:1105–1117
R Development Core Team (2007) R: a language and environment for statistical computing (version 2.6.0). R Foundation for Statistical Computing, URL http://www.R-project.org
Rall BC, Guill C, Brose U (2008) Food-web connectance and predator interference dampen the paradox of enrichment. Oikos 117:202–213
Reeve JD (1997) Predation and bark beetle dynamics. Oecologia 112:48–54
Rogers D (1972) Random search and insect population models. J Anim Ecol 41:369–383
Royama T (1971) A comparative study of models for predation and parasitism. Res Popul Ecol Suppl 1:1–99
Rudolf VHW (2006) The influence of size-specific indirect interactions in predator–prey systems. Ecology 87:362–371
Rudolf VHW (2007) Consequences of stage-structured predators: cannibalism, behavioral effects, and trophic cascades. Ecology 88:2991–3003
Salt GW (1967) Predation in an experimental protozoan population (Woodruffia-Paramecium). Ecol Mon 37:113–144
Salt GW (1974) Predator and prey densities as controls of capture by predator Didinium nasutum. Ecology 55:434–439
Schenk D, Bersier LF, Bacher S (2005) An experimental test of the nature of predation: neither prey- nor ratio-dependent. J Anim Ecol 74:86–91
Skalski GT, Gilliam JF (2001) Functional responses with predator interference: viable alternative to the Holling type II model. Ecology 82:3083–3092
Solomon ME (1949) The natural control of animal populations. J Anim Ecol 18:1–35
Stow CA, Carpenter SR, Cottingham KL (1995) Resource vs. ratio-dependent consumer–resource models: a Bayesian perspective. Ecology 76:1986–1990
Tschanz B, Bersier LF, Bacher S (2007) Functional responses: a question of alternative prey and predator density. Ecology 88:1300–1308
Vos M, Moreno Berrocal S, Karamaouna F, Hemerik L, Vet LEM (2001) Plant-mediated indirect effects and the persistence of parasitoid—herbivore communities. Ecol Lett 4:38–45
Vucetich JA, Peterson RO, Schaefer CL (2002) The effects of prey and predator densities on wolf predation. Ecology 11:3003–3013
Werner EE, Anholt BR (1993) Ecological consequences of the tradeoff between growth and mortality rates mediated by foraging activity. Am Nat 142:242–272
Acknowledgments
We thank E. Hammill, A. Narwani, and S. Pavey for valuable comments on previous drafts of this manuscript and E. von Aderkas for technical assistance with behavioral observations. This research was funded by the Canada Research Chairs Program and an NSERC of Canada Discovery grant to B. R. A. This research conforms to all applicable regulatory requirements.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Libby Marschall.
Rights and permissions
About this article
Cite this article
Kratina, P., Vos, M., Bateman, A. et al. Functional responses modified by predator density. Oecologia 159, 425–433 (2009). https://doi.org/10.1007/s00442-008-1225-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-008-1225-5