Abstract
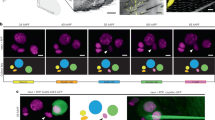
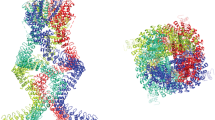
Trichoid sensilla are the most common mechanoreceptors in insects; depending on their distribution, they can act as either exteroceptors or proprioceptors. In this study, the internal structure of the trichoid sensillum from Nilaparvata lugens was studied, using focused ion beam scanning electron microscopy (FIB-SEM). We reconstructed a three-dimensional (3D) model derived from the FIB-SEM data set. The model displayed characteristic mechanosensory sensilla components, including a hair inserted in the socket, a dendrite going through the laminated cuticle, and an electron-dense tubular body at the dendrite terminal. The detailed 3D model showed the relationship between the microtubules within the tubular body and those outside of the tubular body. We also found an autocellular junction in the tormogen cell, indicating that the tormogen cell grows around the dendrite sheath to form a hollow column shape during sensilla morphogenesis.




Similar content being viewed by others
References
Aljunid SF, Anderson M (1983) Ultrastructure of sensilla on the antennal pedicel of the brown planthopperNilaparvata-lugens Stal (Insecta, Homoptera) - 1. Plaque organs and trichoid sensilla. Cell Tissue Res 228:313–322
Ban L-P, Sun Y-P, Wang Y, Tu X-B, Zhang S-G, Zhang Y-T, Wu Y-S, Zhang Z-H (2015) Ultrastructure of antennal sensilla of the peach aphid Myzus persicae sulzer, 1776. J Morphol 276:219–227. https://doi.org/10.1002/jmor.20335
Bechstedt S, Albert JT, Kreil DP, Muller-Reichert T, Gopfert MC, Howard J (2010) A doublecortin containing microtubule-associated protein is implicated in mechanotransduction in Drosophila sensory cilia. Nat Commun 1:1–11. https://doi.org/10.1038/ncomms1007
Brozek J, Bourgoin T (2013) Morphology and distribution of the external labial sensilla in Fulgoromorpha (Insecta: Hemiptera). Zoomorphology 132:33–65. https://doi.org/10.1007/s00435-012-0174-z
Chevalier RL (1969) The fine structure of campaniform sensilla on the halteres of Drosophila melanogaster. J Morphol 128:443–463
de Bakker BS, de Jong KH, Hagoort J, de Bree K, Besselink CT, de Kanter FE, Veldhuis T, Bais B, Schildmeijer R, Ruijter JM, Oostra RJ, Christoffels VM, Moorman AFM (2016) An interactive three-dimensional digital atlas and quantitative database of human development. Science 354:1019 aag0053-+
Dent EW, Baas PW (2014) Microtubules in neurons as information carriers. J Neurochem 129:235–239. https://doi.org/10.1111/jnc.12621
Di Giulio A, Maurizi E, Stacconi MV, Romani R (2012) Functional structure of antennal sensilla in the myrmecophilous beetle Paussus favieri (Coleoptera, Carabidae, Paussini). Micron 43:705–719. https://doi.org/10.1016/j.micron.2011.10.013
French AS (1988) Transduction mechanisms of mechanosensilla. Annu Rev Entomol 33:39–58
Fu BX, Bellis G-A, Hong J, Wang J-R, Wu Q, Tang Q-Y, Cheng J-A, Zhu Z-R (2012) Morphology, distribution, and abundance of antennal sensilla of male and female macropterous and brachypterous small brown planthopper, Laodelphax striatellus (Fallen) (Hemiptera: Delphacidae). Microsc Res Tech 75:1492–1512. https://doi.org/10.1002/jemt.22093
Gaffal KP, Tichy H, Theiß J, Seelinger G (1975) Structural polarities in mechanosensitive sensilla and their influence on stimulus transmission (Arthropoda). Zoomorphologie 82:79–103. https://doi.org/10.1007/bf00993585
Guthrie DM (1966) The function and fine structure of the cephalic airflow receptor in Schistocerca gregaria. J Cell Sci 1:463–470
Hartenstein V, Posakony JW (1989) Development of adult sensilla on the wing and notum of Drosophila melanogaster. Development 107:389–405
Kalogianni E (1995) Physiological properties of wind-sensitive and tactile trichoid sensilla on the ovipositor and their role during oviposition in the locust. J Exp Biol 198:1359–1369
Keil TA (1984) Very tight contact of tormogen cell membrane and sensillum cuticle: Ultrastructural basis for high electrical resistance between receptorlymph and subcuticular spaces in silkmoth olfactory hairs. Tissue Cell 16:131–135
Keil TA (1997) Functional morphology of insect mechanoreceptors. Microsc Res Tech 39:506–531
Klowden MJ (2008) Physiological Systems in Insects. Academic Press, Cambridge
Küppers J (1974) Measurements on the ionic milieu of the receptor terminal in mechanoreceptive sensilla of insects. In: Symposium Mechanoreception: Abhandlungen der Rheinisch-Westfälischen Akademie der Wissenschaften. VS Verlag für Sozialwissenschaften, Wiesbaden, pp 387–394.
Küppers J, Thurm U (1979) Active ion transport by a sensory epithelium. J Comp Physiol 134:131–136. https://doi.org/10.1007/bf00610471
Liang X, Madrid J, Saleh HS, Howard J (2011) NOMPC, a member of the TRP channel family, localizes to the tubular body and distal cilium of Drosophila campaniform and chordotonal receptor cells. Cytoskeleton 68:1–7. https://doi.org/10.1002/cm.20493
Liang X, Madrid J, Gärtner R, Verbavatz J, Schiklenk C, Wilsch-Braeuninger M, Bogdanova AI, Stenger F, Voigt A, Howard J (2013) A NOMPC-dependent membrane-microtubule connector is a candidate for the gating spring in fly mechanoreceptors. Curr Biol 23:755–763
Matheson T (2012) The insects: Structure and function, fifth edn. Cambridge University Press, Cambridge
Matsumoto DE, Farley RG (1978) Comparison of the ultrastructure of stimulated and unstimulated mechanoreceptors in the taste hairs of the blowfly Phaenicia serricata. Tissue Cell 10:63–76
Moran DT, Rowley JC, Zill SN, Varela FG (1976) The mechanism of sensory transduction in a mechanoreceptor. Functional stages in campaniform sensilla during the molting cycle. J Cell Biol 71:832–847. https://doi.org/10.1083/jcb.71.3.832
Newland PL (1991) Physiological-properties of afferents from tactile hairs on the hindlegs of the locust. J Exp Biol 155:487–503
Rice MJ, Galun R, Margalit J (1973) Mouthpart sensilla of the tsetse fly and their function: III: Labrocibarial sensilla. Ann Trop Med Parasitol 67:109–116
Schroeder TBH, Houghtaling J, Wilts BD, Mayer M (2018) It’s not a bug, it’s a feature: functional materials in insects. Adv Mater. https://doi.org/10.1002/adma.201705322
Sun L, Gao Y, He J, Cui L, Meissner J, Verbavatz JM, Li B, Feng X, Liang X (2019) Ultrastructural organization of NompC in the mechanoreceptive organelle of Drosophila campaniform mechanoreceptors. Proc Natl Acad Sci 116:7343–7352
Tao J, Yu B (2012) Hair flow sensors: From bio-inspiration to bio-mimicking - a review. Smart Mater Struct. https://doi.org/10.1088/0964-1726/21/11/113001
Tautz J (1978) Reception of medium vibration by thoracal hairs of caterpillars Ofbarathra brassicae L. (Lepidoptera, Noctuidae). J Comp Physiol 125:67–77. https://doi.org/10.1007/bf00656832
Thurm U (1964) Mechanoreceptors in the cuticle of the honey bee: fine structure and stimulus mechanism. Science 145:1063–1065. https://doi.org/10.1126/science.145.3636.1063
Funding
This work was supported by the National Natural Science Foundation of China, grants 31630057 and 31471765.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Informed consent
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM1: Interactive 3D PDF showing the 3D structure of the reconstructed sensillum. This file is available on MS Windows or Mac OS systems, using Adobe Acrobat or Acrobat Reader.
ESM2: Supplementary Fig. S1, with legend.
ESM3: The volume data of a mechanosensory trichoid sensillum, obtained using FIB/SEM. The sensillum image data used to create Figs.1, 2, 3, 4, together with the surrounding structures, are presented in x-y, x-z, and y-z planes. The data shown in this video were collected as a stack of x-y plane images, and the other two plane views were created in Amira by aligning the images.
ESM 1
(PDF 59312 kb)
ESM 2
(PDF 921 kb)
ESM 3
(AVI 44008 kb)
Rights and permissions
About this article
Cite this article
Guo, JS., Wang, XQ., Li, DT. et al. Three-dimensional architecture of a mechanoreceptor in the brown planthopper, Nilaparvata lugens, revealed by FIB-SEM. Cell Tissue Res 379, 487–495 (2020). https://doi.org/10.1007/s00441-019-03122-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-019-03122-7