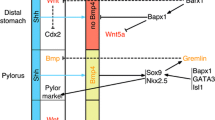
Abstract
During development, the gastrointestinal (GI) tract arises from a primary tube composed of mesoderm and endoderm. The mesoderm gives rise to the digestive mesenchyme, which in turn differentiates into multiple tissues, namely the submucosa, the interstitial cells of Cajal and the smooth muscle cells (SMCs). Concomitant with these early patterning events, the primitive GI tract is colonized by vagal enteric neural crest-derived cells (vENCDCs), a population of cells that gives rise to the enteric nervous system, the intrinsic innervation of the GI tract. Reciprocal neuro-mesenchymal interactions are essential for the coordinated development of GI musculature. The aim of this study is to examine and compare the kinetics of mesenchymal cell differentiation into SMCs along the anterior–posterior axis to the pattern of vENCDCs migration using whole-mount in situ hybridization and paraffin section immunofluorescence analyses on chick embryonic GI tracts from E4-Stage 23 to E7-Stages 30–31. We confirmed that gastric and pre-umbilical intestine mesenchyme differentiation into SMCs occurs after vENCDCs colonization. However, we found that colonic and post-umbilical intestine mesenchyme differentiation occurs before vENCDCs colonization. These findings suggest that regional-specific mechanisms are involved in the mesenchyme differentiation into SMCs along the GI anterior–posterior axis.





Similar content being viewed by others
References
Antonucci A, Fronzoni L, Cogliandro L, Cogliandro RF, Caputo C, De Giorgio R, Pallotti F, Barbara G, Corinaldesi R, Stanghellini V (2008) Chronic intestinal pseudo-obstruction. World J Gastroenterol 14:2953–2961
Boeckxstaens GE, Rumessen JJ, de Wit L, Tytgat GN, Vanderwinden JM (2002) Abnormal distribution of the interstitial cells of cajal in an adult patient with pseudo-obstruction and megaduodenum. Am J Gastroenterol 97:2120–2126
Breau MA, Pietri T, Eder O, Blanche M, Brakebusch C, Fässler R, Thiery JP, Dufour S (2006) Lack of beta1 integrins in enteric neural crest cells leads to a Hirschsprung-like phenotype. Development 133:1725–1734
Burns AJ, Le Douarin NM (1998) The sacral neural crest contributes neurons and glia to the post-umbilical gut: spatiotemporal analysis of the development of the enteric nervous system. Development 125:4335–4347
Burns AJ, Champeval D, Le Douarin NM (2000) Sacral neural crest cells colonise aganglionic hindgut in vivo but fail to compensate for lack of enteric ganglia. Dev Biol 219:30–43
Chetaille P, Preuss C, Burkhard S, Côté JM, Houde C, Castilloux J, Piché J, Gosset N, Leclerc S, Wünnemann F, Thibeault M, Gagnon C, Galli A, Tuck E, Hickson GR, El Amine N, Boufaied I, Lemyre E, de Santa Barbara P, Faure S, Jonzon A, Cameron M, Dietz HC, Gallo-McFarlane E, Benson DW, Moreau C, Labuda D, Canada Consortium FORGE, Zhan SH, Shen Y, Jomphe M, Jones SJ, Bakkers J, Andelfinger G (2014) Mutations in SGOL1 cause a novel cohesinopathy affecting heart and gut rhythm. Nat Genet 46:1245–1249
de Santa Barbara P, Roberts DJ (2002) Tail gut endoderm and gut/genitourinary/tail development: a new tissue-specific role for Hoxa13. Development 129:551–561
de Santa Barbara P, van den Brink GR, Roberts DJ (2002) Molecular etiology of gut malformations and diseases. Am J Med Genet 115:221–230
de Santa Barbara P, Williams J, Goldstein AM, Doyle AM, Nielsen C, Winfield S, Faure S, Roberts DJ (2005) Bone morphogenetic protein signaling pathway plays multiple roles during gastrointestinal tract development. Dev Dyn 234:312–322
Fairman CL, Clagett-Dame M, Lennon VA, Epstein ML (1995) Appearance of neurons in the developing chick gut. Dev Dyn 204:192–201
Faure S, de Santa Barbara P (2011) Molecular embryology of the foregut. J Pediatr Gastroenterol Nutr 52:S2–3
Faure S, Georges M, McKey J, Sagnol S, de Santa Barbara P (2013) Expression pattern of the homeotic gene Bapx1 during early chick gastrointestinal tract development. Gene Expr Patterns 13:287–292
Faure S, McKey J, Sagnol S, de Santa Barbara P (2015) Enteric neural crest cells regulate vertebrate stomach patterning and differentiation. Development 142:331–342
Fu M, Lui VC, Sham MH, Pachnis V, Tam PK (2004) Sonic hedgehog regulates the proliferation, differentiation, and migration of enteric neural crest cells in gut. J Cell Biol 166:673–684
Gabella G (2002) Development of visceral smooth muscle. Results Probl Cell Differ 38:1–37
Grapin-Botton A (2005) Antero-posterior patterning of the vertebrate digestive tract: 40 years after Nicole Le Douarin’s PhD thesis. Int J Dev Biol 49:335–347
Hamburger V, Hamilton HL (1951) A series of normal stages in the development of the chick embryo. J Morph 88:49–92
Hao MM, Foong JP, Bornstein JC, Li ZL, Vanden Berghe P, Boesmans W (2016) Enteric nervous system assembly: functional integration within the developing gut. Dev Biol 417:168–181
Heanue TA, Shepherd IT, Burns AJ (2016) Enteric nervous system development in avian and zebrafish models. Dev Biol 417:129–138
Jayewickreme CD, Shivdasani RA (2015) Control of stomach smooth muscle development and intestinal rotation by transcription factor BARX1. Dev Biol 405:21–32
Kim BM, Buchner G, Miletich I, Sharpe PT, Shivdasani RA (2005) The stomach mesenchymal transcription factor Barx1 specifies gastric epithelial identity through inhibition of transient Wnt signaling. Dev Cell 8:611–622
Le Douarin NM, Teillet MA (1973) The migration of neural crest cells to the wall of the digestive tract in avian embryo. J Embryol Exp Morphol 30:31–48
Le Guen L, Notarnicola C, de Santa Barbara P (2009) Intermuscular tendons are essential for the development of vertebrate stomach. Development 136:791–801
Le Guen L, Marchal S, Faure S, de Santa Barbara P (2015) Mesenchymal-epithelial interactions during digestive tract development and epithelial stem cell regeneration. Cell Mol Life Sci 72:3883–3896
McKey J, Martire D, de Santa Barbara P, Faure S (2016) LIX1 regulates YAP1 activity and controls the proliferation and differentiation of stomach mesenchymal progenitors. BMC Biol 14:34
Moniot B, Biau S, Faure S, Nielsen CM, Berta P, Roberts DJ, de Santa Barbara P (2004) SOX9 specifies the pyloric sphincter epithelium through mesenchymal-epithelial signals. Development 131:3795–3804
Notarnicola C, Rouleau C, Le Guen L, Virsolvy A, Richard S, Faure S, De Santa Barbara P (2012) The RNA-binding protein RBPMS2 regulates development of gastrointestinal smooth muscle. Gastroenterology 143:687–697
Olden T, Akhtar T, Beckman SA, Wallace KN (2008) Differentiation of the zebrafish enteric nervous system and intestinal smooth muscle. Genesis 46:484–498
Pomeranz HD, Gershon MD (1990) Colonization of the avian hindgut by cells derived from the sacral neural crest. Dev Biol 137:378–394
Raghavan S, Bitar KN (2014) The influence of extracellular matrix composition on the differentiation of neuronal subtypes in tissue engineered innervated intestinal smooth muscle sheets. Biomaterials 35:7429–7440
Roberts DJ (2000) Molecular mechanisms of development of the gastrointestinal tract. Dev Dyn 219:109–120
Roberts DJ, Smith DM, Goff DJ, Tabin CJ (1998) Epithelial-mesenchymal signaling during the regionalization of the chick gut. Development 125:2791–2801
Sagnol S, Marchal S, Yang Y, Allemand F, de Santa Barbara P (2016) Epithelial splicing regulatory protein 1 (ESRP1) is a new regulator of stomach smooth muscle development and plasticity. Dev Biol 414:207–218
Sukegawa A, Narita T, Kameda T, Saitoh K, Nohno T, Iba H, Yasugi S, Fukuda K (2000) The concentric structure of the developing gut is regulated by Sonic hedgehog derived from endodermal epithelium. Development 127:1971–1980
Theodosiou NA, Tabin CJ (2003) Wnt signaling during development of the gastrointestinal tract. Dev Biol 259:258–271
Tucker GC, Aoyama H, Lipinski M, Tursz T, Thiery JP (1984) Identical reactivity of monoclonal antibodies HNK-1 and NC-1: conservation in vertebrates on cells derived from the neural primordium and on some leukocytes. Cell Differ 14:223–230
Wallace AS, Burns AJ (2005) Development of the enteric nervous system, smooth muscle and interstitial cells of Cajal in the human gastrointestinal tract. Cell Tissue Res 319:367–382
Yntema CL, Hammond WS (1954) The origin of intrinsic ganglia of trunk viscera from vagal neural crest in the chick embryos. J Comp Neurol 101:515–541
Acknowledgements
The author thanks the members of INSERM U1046.
Author contribution
AB, NC, PdSB and SF performed experiments. PdSB, NC and SF analysed results, contributed expertise and wrote the paper. PdSB and SF obtained funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Research was supported by a Trampolin grant (N°15681) from the Association Française contre les Myopathies (AFM) to SF and grants from AFM (N°18766), Fondation ARC and the French Association for CIPO patients to PdSB.
Conflict of interest
No competing interests declared.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Fig 1
Differentiation of GI smooth muscle at E7.5. Whole-mount in situ hybridization analysis of SM22 expression. A faint level of SM22 transcripts was detected in the pylorus (white arrowhead). st stomach. (GIF 187 kb)
Rights and permissions
About this article
Cite this article
Bourret, A., Chauvet, N., de Santa Barbara, P. et al. Colonic mesenchyme differentiates into smooth muscle before its colonization by vagal enteric neural crest-derived cells in the chick embryo. Cell Tissue Res 368, 503–511 (2017). https://doi.org/10.1007/s00441-017-2577-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-017-2577-0