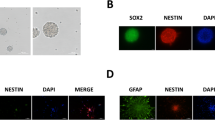
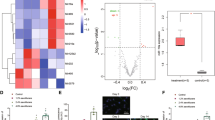
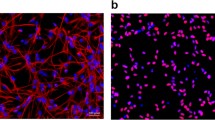
Abstract
The commonly used inhalational anesthetic, sevoflurane, can cause toxicity to the central nervous system of the developing fetus. Lin28 has been reported to regulate let-7a, thereby modulating embryo development, neurodegeneration, and even neuron-related tumorigenesis. We demonstrate that pregnant mice receiving sevoflurane treatment during the early stage of pregnancy give birth to fewer offspring presenting a lower birth weight. We have also treated mouse embryonic stem cells (mESCs) with sevoflurane for 6 h and determined that mESCs self-renewal is repressed, and that differentiation is initiated earlier than in controls. We have induced neural differentiation in the treated mESCs and determined that their neurogenesis is weakened. Furthermore, sevoflurane upregulates the level of let-7a, which might repress mESC self-renewal by directly targeting the Lin28 3′-untranslated region. Lin28 overexpression attenuates the influence of sevoflurane or of let-7a on the self-renewal of mESCs and their subsequent neural differentiation. The let-7a inhibitor also abolishes the influence of sevoflurane. Thus, the let-7a-Lin28 pathway is involved in the sevoflurane-induced inhibition of ESC self-renewal and subsequent neurogenesis. Our study demonstrates the molecular mechanism underlying the side effects of sevoflurane during early development, laying the foundation for studies on the safe and reasonable usage of other inhalational anesthetics.





Similar content being viewed by others
References
Ai Y, Liu Q, Li Y, Duan T (2014) Progesterone signaling/miR-200a/zeb2 axis regulates self-renewal of mouse embryonic stem cells. Biomed Pharm 68:201–208
Beard C, Hochedlinger K, Plath K, Wutz A, Jaenisch R (2006) Efficient method to generate single-copy transgenic mice by site-specific integration in embryonic stem cells. Genesis 44:23–28
Bin G, Jiarong Z, Shihao W, Xiuli S, Cheng X, Liangbiao C, Ming Z (2012) Aire promotes the self-renewal of embryonic stem cells through Lin28. Stem Cells Dev 21:2878–2890
Boyer LA, Lee TI, Cole MF, Johnstone SE, Levine SS, Zucker JP, Guenther MG, Kumar RM, Murray HL, Jenner RG, Gifford DK, Melton DA, Jaenisch R, Young RA (2005) Core transcriptional regulatory circuitry in human embryonic stem cells. Cell 122:947–956
Chen AX, Yu KD, Fan L, Li JY, Yang C, Huang AJ, Shao ZM (2011) Germline genetic variants disturbing the Let-7/LIN28 double-negative feedback loop alter breast cancer susceptibility. PLoS Genet 7:e1002259
Cimadamore F, Amador-Arjona A, Chen C, Huang CT, Terskikh AV (2013) SOX2-LIN28/let-7 pathway regulates proliferation and neurogenesis in neural precursors. Proc Natl Acad Sci U S A 110:E3017–E3026
Crippa S, Cassano M, Sampaolesi M (2012) Role of miRNAs in muscle stem cell biology: proliferation, differentiation and death. Curr Pharm Des 18:1718–1729
Culley DJ, Boyd JD, Palanisamy A, Xie Z, Kojima K, Vacanti CA, Tanzi RE, Crosby G (2011) Isoflurane decreases self-renewal capacity of rat cultured neural stem cells. Anesthesiology 115:754–763
Darr H, Benvenisty N (2009) Genetic analysis of the role of the reprogramming gene LIN-28 in human embryonic stem cells. Stem Cells 27:352–362
Dong Y, Zhang G, Zhang B, Moir RD, Xia W, Marcantonio ER, Culley DJ, Crosby G, Tanzi RE, Xie Z (2009) The common inhalational anesthetic sevoflurane induces apoptosis and increases beta-amyloid protein levels. Arch Neurol 66:620–631
Farh KK, Grimson A, Jan C, Lewis BP, Johnston WK, Lim LP, Burge CB, Bartel DP (2005) The widespread impact of mammalian MicroRNAs on mRNA repression and evolution. Science 310:1817–1821
Glazov EA, Cottee PA, Barris WC, Moore RJ, Dalrymple BP, Tizard ML (2008) A microRNA catalog of the developing chicken embryo identified by a deep sequencing approach. Genome Res 18:957–964
Griffiths-Jones S, Grocock RJ, Dongen S van, Bateman A, Enright AJ (2006) miRBase: microRNA sequences, targets and gene nomenclature. Nucleic Acids Res 34:D140–D144
Guo X, Liu Q, Wang G, Zhu S, Gao L, Hong W, Chen Y, Wu M, Liu H, Jiang C, Kang J (2013) microRNA-29b is a novel mediator of Sox2 function in the regulation of somatic cell reprogramming. Cell Res 23:142–156
Henzler CM, Li Z, Dang J, Arcila ML, Zhou H, Liu J, Chang KY, Bassett DS, Rana TM, Kosik KS (2013) Staged miRNA re-regulation patterns during reprogramming. Genome Biol 14:R149
Jaenisch R, Young R (2008) Stem cells, the molecular circuitry of pluripotency and nuclear reprogramming. Cell 132:567–582
Keller G (2005) Embryonic stem cell differentiation: emergence of a new era in biology and medicine. Genes Dev 19:1129–1155
Kong D, Heath E, Chen W, Cher ML, Powell I, Heilbrun L, Li Y, Ali S, Sethi S, Hassan O, Hwang C, Gupta N, Chitale D, Sakr WA, Menon M, Sarkar FH (2012) Loss of let-7 up-regulates EZH2 in prostate cancer consistent with the acquisition of cancer stem cell signatures that are attenuated by BR-DIM. PLoS One 7:e33729
Lewis BP, Shih IH, Jones-Rhoades MW, Bartel DP, Burge CB (2003) Prediction of mammalian microRNA targets. Cell 115:787–798
Liu Q, Wang G, Chen Y, Li G, Yang D, Kang J (2014) A miR-590/Acvr2a/Rad51b axis regulates DNA damage repair during mESC proliferation. Stem Cell Rep 3:1103–1117
Liu Y, Liu Q, Jia W, Chen J, Wang J, Ye D, Guo X, Chen W, Li G, Wang G, Deng A, Kang J (2013) MicroRNA-200a regulates Grb2 and suppresses differentiation of mouse embryonic stem cells into endoderm and mesoderm. PLoS One 8:e68990
Lozier AM, Rich ME, Grawe AP, Peck AS, Zhao P, Chang AT, Bond JP, Sholler GS (2015) Targeting ornithine decarboxylase reverses the LIN28/Let-7 axis and inhibits glycolytic metabolism in neuroblastoma. Oncotarget 6:196–206
Lu Y, Wu X, Dong Y, Xu Z, Zhang Y, Xie Z (2010) Anesthetic sevoflurane causes neurotoxicity differently in neonatal naïve and Alzheimer disease transgenic mice. Anesthesiology 112:1404–1416
Molenaar JJ, Domingo-Fernandez R, Ebus ME, Lindner S, Koster J, Drabek K, Mestdagh P, Sluis P van, Valentijn LJ, Nes J van, Broekmans M, Haneveld F, Volckmann R, Bray I, Heukamp L, Sprussel A, Thor T, Kieckbusch K, Klein-Hitpass L, Fischer M, Vandesompele J, Schramm A, van Noesel MM, Varesio L, Speleman F, Eggert A, Stallings RL, Caron HN, Versteeg R, Schulte JH (2012) LIN28B induces neuroblastoma and enhances MYCN levels via let-7 suppression. Nat Genet 44:1199–1206
Ní Mhuireachtaigh R, O'Gorman DA (2006) Anesthesia in pregnant patients for nonobstetric surgery. J Clin Anesth 18:60–66
Nie H, Peng Z, Lao N, Dong H, Xiong L (2013) Effects of sevoflurane on self-renewal capacity and differentiation of cultured neural stem cells. Neurochem Res 38:1758–1767
Piskounova E, Viswanathan SR, Janas M, LaPierre RJ, Daley GQ, Sliz P, Gregory RI (2008) Determinants of microRNA processing inhibition by the developmentally regulated RNA-binding protein Lin28. J Biol Chem 283:21310–21314
Rehfeld F, Rohde AM, Nguyen DT, Wulczyn FG (2015) Lin28 and let-7: ancient milestones on the road from pluripotency to neurogenesis. Cell Tissue Res 359:145–160
Reitman E, Flood P (2011) Anaesthetic considerations for non-obstetric surgery during pregnancy. Br J Anaesth 107:i72–i78
Rybak A, Fuchs H, Smirnova L, Brandt C, Pohl EE, Nitsch R, Wulczyn FG (2008) A feedback loop comprising lin-28 and let-7 controls pre-let-7 maturation during neural stem-cell commitment. Nat Cell Biol 10:987–993
Shen X, Dong Y, Xu Z, Wang H, Miao C, Soriano SG, Sun D, Baxter MG, Zhang Y, Xie Z (2013) Selective anesthesia-induced neuroinflammation in developing mouse brain and cognitive impairment. Anesthesiology 118:502–515
Shyh-Chang N, Daley GQ (2013) Lin28: primal regulator of growth and metabolism in stem cells. Cell Stem Cell 12:395–406
Tennakoon JB, Wang H, Coarfa C, Cooney AJ, Gunaratne PH (2013) Chromatin changes in dicer-deficient mouse embryonic stem cells in response to retinoic acid induced differentiation. PLoS One 8:e74556
Toledano H, D'Alterio C, Czech B, Levine E, Jones DL (2012) The let-7-Imp axis regulates ageing of the Drosophila testis stem-cell niche. Nature 485:605–610
Weingart MF, Roth JJ, Hutt-Cabezas M, Busse TM, Kaur H, Price A, Maynard R, Rubens J, Taylor I, Mao XG, Xu J, Kuwahara Y, Allen SJ, Erdreich-Epstein A, Weissman BE, Orr BA, Eberhart CG, Biegel JA, Raabe EH (2015) Disrupting LIN28 in atypical teratoid rhabdoid tumors reveals the importance of the mitogen activated protein kinase pathway as a therapeutic target. Oncotarget 6:3165–3177
West JA, Viswanathan SR, Yabuuchi A, Cunniff K, Takeuchi A, Park IH, Sero JE, Zhu H, Perez-Atayde A, Frazier AL, Surani MA, Daley GQ (2009) A role for Lin28 in primordial germ-cell development and germ-cell malignancy. Nature 460:909–913
Wilder RT, Flick RP, Sprung J, Katusic SK, Barbaresi WJ, Mickelson C, Gleich SJ, Schroeder DR, Weaver AL, Warner DO (2009) Early exposure to anesthesia and learning disabilities in a population-based birth cohort. Anesthesiology 110:796–804
Xu N, Papagiannakopoulos T, Pan G, Thomson JA, Kosik KS (2009) MicroRNA-145 regulates OCT4, SOX2, and KLF4 and represses pluripotency in human embryonic stem cells. Cell 137:647–658
Yu CC, Chen YW, Chiou GY, Tsai LL, Huang PI, Chang CY, Tseng LM, Chiou SH, Yen SH, Chou MY, Chu PY, Lo WL (2011) MicroRNA let-7a represses chemoresistance and tumourigenicity in head and neck cancer via stem-like properties ablation. Oral Oncol 47:202–210
Zhang L, Zhang J, Yang L, Dong Y, Zhang Y, Xie Z (2013) Isoflurane and sevoflurane increase interleukin-6 levels through the nuclear factor-kappa B pathway in neuroglioma cells. Br J Anaesth 110:i82–i91
Zhang Y, Dong Y, Zheng H, Shie V, Wang H, Busscher JJ, Yue Y, Xu Z, Xie Z (2013) Sevoflurane inhibits neurogenesis and the Wnt-catenin signaling pathway in mouse neural progenitor cells. Curr Mol Med 13:1446–1454
Zheng H, Dong Y, Xu Z, Crosby G, Culley DJ, Zhang Y, Xie Z (2013) Sevoflurane anesthesia in pregnant mice induces neurotoxicity in fetal and offspring mice. Anesthesiology 118:516–526
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
None.
Contributions
Xiuwen Yi conducted the study, collected the data, and prepared the manuscript. Yirong Cai collected the data, carried out detailed analysis, and acted as archival author. Nan Zhang conducted the study. Qingxiu Wang designed the study. Wenxian Li designed the study and prepared the manuscript.
Additional information
Xiuwen Yi and Yirong Cai contributed equally to this work.
This research was supported by the Science and Technology Commission of Shanghai Municipality (11ZR1406200) and by the Key Specialty Construction Project of Pudong Health and Family Planning Commission of Shanghai (grant no. PWZz2013-17).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. 1
(related to Fig. 2) Sevoflurane induces apoptosis and decreases cell proliferation and viability of mESCs during neurogenesis. a Detection of mesodermal (Mixi, T, Snail) and endodermal (Gata4, Afp, Apoa1) markers during the induction of neurogenesis on day 14 (ctrl control). For all experiments, n = 4, average ± SD, *P < 0.05, **P < 0.01. b Apoptosis assay during the induction of neurogenesis on day 14. For all experiments, n = 5, average ± SD, **P < 0.01. c Cell viability assay during the induction of neurogenesis on day 14. For all experiments, n = 4, average ± SD, *P < 0.05. d Sevoflurane inhibited the proliferation of mESCs. For all experiments, n = 4, average ± SD. e Apoptosis assay of mESCs cells treated with sevoflurane. For all experiments, n = 5, average ± SD, *P < 0.05. f Cell viability assay of mESCs treated with sevoflurane. For all experiments, n = 5, average ± SD, ***P < 0.01 (GIF 47 kb)
Fig. 2
(related to Fig. 3) let-7a inhibitor abrogated the effect of sevoflurane on apoptosis and cell viability of mESCs. a Apoptosis assay of mESCs transfected with let-7a or control (ctrl). For all experiments, n = 5, average ± SD, **P < 0.01. b Cell viability assay of mESCs transfected with let-7a or control. For all experiments, n = 4, average ± SD, ***P < 0.001. c Overexpression of let-7a inhibited the proliferation of mESCs. For all experiments, n = 4, average ± SD. d Apoptosis assay of mESCs transfected with let-7a inhibitor or control. For all experiments, n = 4, average ± SD. e Cell viability assay of mESCs transfected with let-7a inhibitor or control. For all experiments, n = 3, average ± SD. f let-7a inhibitor had no significant influence on cell proliferation. For all experiments, n = 4, average ± SD. g let-7a inhibitor abrogated the effect of sevoflurane on apoptosis in mESCs. For all experiments, n = 5, average ± SD, *P < 0.05. h let-7a inhibitor abrogated the effect of sevoflurane on cell viability in mESCs. For all experiments, n = 3, average ± SD, *P < 0.05. i let-7a inhibitor abrogated the effect of sevoflurane on cell proliferation in mESCs. For all experiments, n = 5, average ± SD. (GIF 82 kb)
Fig. 3
(related to Fig. 4) Overexpression of Lin28 restored the function of let-7a or sevoflurane in mESCs. a Overexpression of Lin28 restored the effect of let-7a on apoptosis (ctrl mESCs treated with no sevoflurane and transfected with empty overexpression control vector, OV-ctrl empty overexpression control vector). For all experiments, n = 5, average ± SD, *P < 0.05. b Cell viability assay of mESCs. For all experiments, n = 4, average ± SD, **P < 0.01. c Overexpression of Lin28 restored the effect of sevoflurane on apoptosis. For all experiments, n = 6, average ± SD, *P < 0.05. d Cell viability assay of mESCs transfected with let-7a inhibitor or control. For all experiments, n = 6, average ± SD, **P < 0.01. (GIF 34 kb)
Fig. 4
(related to Fig. 5) Sevoflurane regulates apoptosis and cell viability through the let-7a-Lin28 pathway. a Cell apoptosis assay of mESCs in rescue experiments. For all experiments, n = 5, average ± SD, *P < 0.05, **P < 0.01. b Cell viability assay of mESCs in rescue experiments. For all experiments, n = 6, average ± SD, *P < 0.05. c Cell proliferation assay of mESCs in rescue experiments. For all experiments, n = 3, average ± SD. d Cell apoptosis assay of cell derived from the treated mESCs during neurogenesis in rescue experiments. For all experiments, n = 8, average ± SD, *P < 0.05, **P < 0.01. e Cell viability assay of cells derived from the treated mESCs during neurogenesis. For all experiments, n = 5, average ± SD, *P < 0.05, **P < 0.01. (GIF 79 kb)
Rights and permissions
About this article
Cite this article
Yi, X., Cai, Y., Zhang, N. et al. Sevoflurane inhibits embryonic stem cell self-renewal and subsequent neural differentiation by modulating the let-7a-Lin28 signaling pathway. Cell Tissue Res 365, 319–330 (2016). https://doi.org/10.1007/s00441-016-2394-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-016-2394-x