Abstract
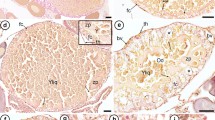
Ex-fissiparous planarians produce infertile cocoons or, in very rare cases, cocoons with very low fertility. Here, we describe the features of programmed cell death (PCD) occurring in the hyperplasic ovary of the ex-fissiparous freshwater planarian Dugesia arabica that may explain this infertility. Based on TEM results, we demonstrate a novel extensive co-clustering of cytoplasmic organelles, such as lysosomes and microtubules, and their fusion with autophagosomes during the early stage of oocyte cell death occurring through an autophagic pattern. During a later stage of cell death, the generation of apoptotic vesicles in the cytoplasm can be observed. The immunohistochemical labeling supports the ultrastructural results because it has been shown that the proapoptotic protein bax was more highly expressed in the hyperplasic ovary than in the normal one, whereas the anti-apoptotic protein bcl2 was slightly more highly expressed in the normal ovary compared to the hyperplasic one. TUNEL analysis of the hyperplasic ovary confirmed that the nuclei of the majority of differentiating oocytes were TUNEL-positive, whereas the nuclei of oogonia and young oocytes were TUNEL-negative; in the normal ovary, oocytes are TUNEL-negative. Considering all of these data, we suggest that the cell death mechanism of differentiating oocytes in the hyperplasic ovary of freshwater planarians is one of the most important factors that cause ex-fissiparous planarian infertility. We propose that autophagy precedes apoptosis during oogenesis, whereas apoptotic features can be observed later.







Similar content being viewed by others
References
Benazzi M (1974) Fissioning in planarians from a genetic standpoint. In: Riser NW, Morse MP (eds) Biology of the Turbellaria. McGraw-Hill Book Company, New York, pp 476–92
Booth LA, Tavallai S, Hamed HA, Cruickshanks N, Dent P (2014) The role of cell signalling in the crosstalk between autophagy and apoptosis. Cell Signal 26(3):549–55. doi:10.1016/j.cellsig.2013.11.028
Bose P, Thakur S, Thalappilly S et al (2013) ING1 induces apoptosis through direct effects at the mitochondria. Cell Death dis 4:e837. doi:10.1038/cddis.2013.398
Bromley HJ (1974) Morpho-karyological types of Dugesia (Turbellaria, Tricladida) in Israel and their distributions patterns. Zool Scr 3:239–242
Brunk UT, Neuzil J, Eaton JW (2001) Lysosomal involvement in apoptosis. Redox Rep 6(2):91–7
Bursch W (2004) Multiple cell death programs: Charon's lifts to Hades. FEMS Yeast Res 5(2):101–10
Bursch W, Hochegger K, Torok L, Marian B, Ellinger A, Hermann RS (2000) Autophagic and apoptotic types of programmed cell death exhibit different fates of cytoskeletal filaments. J Cell Sci 113(Pt 7):1189–98
Chong T, Collins JJ, Brubacher JL, Zarkower D, Newmark PA (2013) A sex-specific transcription factor controls male identity in a simultaneous hermaphrodite. Nat Commun 4:1814 doi:10.1038/Ncomms2811
Cirman T, Oresic K, Mazovec GD et al (2004) Selective disruption of lysosomes in HeLa cells triggers apoptosis mediated by cleavage of Bid by multiple papain-like lysosomal cathepsins. J Biol Chem 279(5):3578–87
Clarke PG (1990) Developmental cell death: morphological diversity and multiple mechanisms. Anat Embryol (Berl) 181(3):195–213
Collins JJ, Hou XW, Romanova EV, et al. (2010) Genome-Wide Analyses Reveal a Role for Peptide Hormones in Planarian Germline Development. Plos Biol 8 (10) doi: ARTN e1000509 doi:10.1371/journal.pbio.1000509
De Duve C, Wattiaux R (1966) Functions of lysosomes. Annu Rev Physiol 28:435–92
Galluzzi L, Vitale I, Abrams JM, et al. Molecular definitions of cell death subroutines: recommendations of the Nomenclature Committee on Cell Death 2012. Cell Death Differ 19(1):107–20
Gonzalez-Estevez C, Felix DA, Aboobaker AA, Salo E (2007a) Gtdap-1 and the role of autophagy during planarian regeneration and starvation. Autophagy 3(6):640–2
Gonzalez-Estevez C, Felix DA, Aboobaker AA, Salo E (2007b) Gtdap-1 promotes autophagy and is required for planarian remodeling during regeneration and starvation. Proc Natl Acad Sci U S A 104(33):13373–8
Grasso M, Benazzi M (1973) Genetic and physiological control of fissioning and sexuality in planarians. J Embryo Exp Morph 30(2):317–328
Gremigni V, Banchetti R (1972a) The origin of hyperplasia in the ovaries of ex-fissiparous individuals in Dugesia gonocephala s.l. . Accad Naz Lincei 53 (477–485)
Gremigni V, Banchetti R (1972b) Submicroscopic morphology of hyperplasic ovaries of ex-fissiparous individuals in Dugesia gonocephala s.l. R C Accad Lincei 52:539–543
Gremigni V, Falleni A (1998) Characters of the female gonad and the phylogeny of Platyhelminthes-Turbellaria. Hydrobiologia 383:235–242
Guicciardi ME, Deussing J, Miyoshi H et al (2000) Cathepsin B contributes to TNF-alpha-mediated hepatocyte apoptosis by promoting mitochondrial release of cytochrome c. J Clin Invest 106(9):1127–37
Guicciardi ME, Leist M, Gores GJ (2004) Lysosomes in cell death. Oncogene 23(16):2881–90
Gump JM, Thorburn A (2011) Autophagy and apoptosis: what is the connection? Trends Cell Biol 21(7):387–92. doi:10.1016/j.tcb.2011.03.007
Handberg-Thorsager M, Salo E (2007) The planarian nanos-like gene Smednos is expressed in germline and eye precursor cells during development and regeneration. Dev Genes Evol 217(5):403–11. doi:10.1007/s00427-007-0146-3
Harrath AH, Alwasal SH, Alhazza I, Zghal F, Tekaya S (2011) An ultrastructural study of oogenesis in the planarian Schmidtea mediterranea (Platyhelminthe, Paludicola). C R Biol 334(7):516–25
Harrath AH, Alwasel S, Zghal F, Tekaya S (2012) Ultrastructure of spermatogenesis and mature spermatozoon of the freshwater planarian Schmidtea mediterranea (Platyhelminthes, Paludicola). C R Biol 335(2):87–95
Harrath AH, Ahmed M, Sayed SR, Saifi MA, Alwasel SH (2013a) An ultrastructural study of oogenesis and cell dynamics during cocoon shell secretion in the subterranean freshwater planarian Dendrocoelum constrictum (Platyhelminthes, Tricladida). Tissue Cell 45(1):39–46
Harrath AH, Sluys R, Aldahmash W, Al-Razaki A, Alwasel S (2013b) Reproductive strategies, karyology, parasites, and taxonomic status of Dugesia populations from Yemen (Platyhelminthes: Tricladida: Dugesiidae). Zoolog Sci 30(6):502–8
Kondo H, Maruo T, Peng X, Mochizuki M (1996) Immunological evidence for the expression of the Fas antigen in the infant and adult human ovary during follicular regression and atresia. J Clin Endocrinol Metab 81(7):2702–10
Lauber K, Bohn E, Krober SM et al (2003) Apoptotic cells induce migration of phagocytes via caspase-3-mediated release of a lipid attraction signal. Cell 113(6):717–30
Maillet G, Feral C, Benhaim A (2005) Apoptosis of the follicular cells: its implication in ovarian induction protocols. Gynecol Obstet Fertil 33(9):653–8
Maiuri MC, Zalckvar E, Kimchi A, Kroemer G (2007) Self-eating and self-killing: crosstalk between autophagy and apoptosis. Nat Rev Mol Cell Biol 8(9):741–52
Matova N, Cooley L (2001) Comparative aspects of animal oogenesis. Dev Biol 231(2):291–320
Matsumura H, Kano K, Marin de Evsikova C et al (2009) Transcriptome analysis reveals an unexpected role of a collagen tyrosine kinase receptor gene, Ddr2, as a regulator of ovarian function. Physiol Genomics 39(2):120–9. doi:10.1152/physiolgenomics.00073.2009
McCormick JH, Stokes GN, Hermanutz RO (1989) Oocyte atresia and reproductive success in fathead minnows (Pimephales promelas) exposed to acidified hardwater environments. Arch Environ Contam Toxicol 18(1–2):207–14
McPhee CK, Baehrecke EH (2009) Autophagy in Drosophila melanogaster. Biochim Biophys Acta 1793(9):1452–60
Miller TM, Moulder KL, Knudson CM et al (1997) Bax deletion further orders the cell death pathway in cerebellar granule cells and suggests a caspase-independent pathway to cell death. J Cell Biol 139(1):205–17
Mpakou VE, Nezis IP, Stravopodis DJ, Margaritis LH, Papassideri IS (2008) Different modes of programmed cell death during oogenesis of the silkmoth Bombyx mori. Autophagy 4(1):97–100
Mujtaba SF, Dwivedi A, Yadav N, Ray RS, Singh G (2013) Singlet oxygen mediated apoptosis by anthrone involving lysosomes and mitochondria at ambient UV exposure. J Hazard Mater 252–253:258–71. doi:10.1016/j.jhazmat.2013.02.055
Muller F, Adori C, Sass M (2004) Autophagic and apoptotic features during programmed cell death in the fat body of the tobacco hornworm (Manduca sexta). Eur J Cell Biol 83(2):67–78. doi:10.1078/0171-9335-00359
Nechushtan A, Smith CL, Lamensdorf I, Yoon SH, Youle RJ (2001) Bax and Bak coalesce into novel mitochondria-associated clusters during apoptosis. J Cell Biol 153(6):1265–76
Nozawa K, Casiano CA, Hamel JC, Molinaro C, Fritzler MJ, Chan EK (2002) Fragmentation of Golgi complex and Golgi autoantigens during apoptosis and necrosis. Arthritis Res 4(4):R3
Salvetti A, Rossi L, Lena A et al (2005) DjPum, a homologue of Drosophila Pumilio, is essential to planarian stem cell maintenance. Development 132(8):1863–74. doi:10.1242/dev.01785
Sato K, Shibata N, Orii H et al (2006) Identification and origin of the germline stem cells as revealed by the expression of nanos-related gene in planarians. Dev Growth Differ 48(9):615–28. doi:10.1111/j.1440-169X.2006.00897.x
Scott RC, Juhasz G, Neufeld TP (2007) Direct induction of autophagy by Atg1 inhibits cell growth and induces apoptotic cell death. Curr Biol 17(1):1–11. doi:10.1016/j.cub.2006.10.053
Servais H, Van Der Smissen P, Thirion G et al (2005) Gentamicin-induced apoptosis in LLC-PK1 cells: involvement of lysosomes and mitochondria. Toxicol Appl Pharmacol 206(3):321–33
Siekierska E (2007) Changes in the ovary cells structure in the earthworm Dendrobaena veneta (Rosa) in neurosecretion deficiency and after exposure to cadmium. Eur J Soil Biol 43:S311-S315 doi:10.1016/j.ejsobi.2007.08.034
Smith JE 3rd, Cummings CA, Cronmiller C (2002) Daughterless coordinates somatic cell proliferation, differentiation and germline cyst survival during follicle formation in Drosophila. Development 129(13):3255–67
Solana J, Lasko P, Romero R (2009) Spoltud-1 is a chromatoid body component required for planarian long-term stem cell self-renewal. Dev Biol 328(2):410–21. doi:10.1016/j.ydbio.2009.01.043
Stocchino GA, Sluys R, Manconi R (2012) A new species of Dugesia (Platyhelminthes, Tricladida, Dugesiidae) from the Afromontane forest in South Africa, with an overview of freshwater planarians from the African continent. Zootaxa (3551):43–58
Taylor RC, Cullen SP, Martin SJ (2008) Apoptosis: controlled demolition at the cellular level. Nat Rev Mol Cell Biol 9(3):231–41
Vaskivuo TE, Tapanainen JS (2003) Apoptosis in the human ovary. Reprod Biomed Online 6(1):24–35
Velentzas AD, Nezis IP, Stravopodis DJ, Papassideri IS, Margaritis LH (2007a) Apoptosis and autophagy function cooperatively for the efficacious execution of programmed nurse cell death during Drosophila virilis oogenesis. Autophagy 3(2):130–2
Velentzas AD, Nezis IP, Stravopodis DJ, Papassideri IS, Margaritis LH (2007b) Mechanisms of programmed cell death during oogenesis in Drosophila virilis. Cell Tissue Res 327(2):399–414. doi:10.1007/s00441-006-0298-x
Wang Y, Zayas RM, Guo T, Newmark PA (2007) nanos function is essential for development and regeneration of planarian germ cells. Proc Natl Acad Sci U S A 104(14):5901–6. doi:10.1073/pnas.0609708104
Wyllie AH, Kerr JFR, Currie AR (1972) Cell death: the significance of apoptosis. Int Rev Cytol 68:251–307
Xiang J, Chao DT, Korsmeyer SJ (1996) BAX-induced cell death may not require interleukin 1 beta-converting enzyme-like proteases. Proc Natl Acad Sci U S A 93(25):14559–63
Yonekawa T, Thorburn A (2013) Autophagy and cell death. Essays Biochem 55(1):105–17. doi:10.1042/bse0550105
Zacarin EC (2007) Autophagy and apoptosis coordinate physiological cell death in larval salivary glands of Apis mellifera (Hymenoptera: Apidae). Autophagy 3(5):516–8
Acknowledgments
The authors extend their appreciation to the Deanship of Scientific Research at King Saud University for funding this work through the research group project NoRGP-VPP-254. We thank Dr. Cristina González-Estévez, Department of Developmental Genetics and Gene Control, Institute of Genetics, Queen’s Medical Centre, The University of Nottingham, for her constructive criticism on an earlier draft of the manuscript.
Conflict of Interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Harrath, A.H., Semlali, A., Mansour, L. et al. Infertility in the hyperplasic ovary of freshwater planarians: the role of programmed cell death. Cell Tissue Res 358, 607–620 (2014). https://doi.org/10.1007/s00441-014-1971-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-014-1971-0