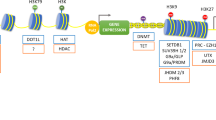
Abstract
Epigenetic mechanisms are fundamental key features of developing cells connecting developmental regulatory factors to chromatin modification. Changes in the environment during renal development can have long-lasting effects on the permanent tissue structure and the level of expression of important functional genes. These changes are believed to contribute to kidney disease occurrence and progression. Although the mechanisms of early patterning and cell fate have been well described for renal development, little is known about associated epigenetic modifications and their impact on how genes interact to specify the renal epithelial cells of nephrons and how this specification is relevant to maintaining normal renal function. A better understanding of the renal cell-specific epigenetic modifications and the interaction of different cell types to form this highly complex organ will not only help to better understand developmental defects and early loss of kidney function in children, but also help to understand and improve chronic disease progression, cell regeneration and renal aging.

Similar content being viewed by others
References
Anglin JL, Song Y (2013) A medicinal chemistry perspective for targeting histone H3 lysine-79 methyltransferase DOT1L. J Med Chem 56:8972–8983
Aoki M, Terada T, Kajiwara M, Ogasawara K, Ikai I, Ogawa O, Katsura T, Inui K (2008) Kidney-specific expression of human organic cation transporter 2 (OCT2/SLC22A2) is regulated by DNA methylation. Am J Physiol Ren Physiol 295:F165–F170
Bechtel W, McGoohan S, Zeisberg EM, Muller GA, Kalbacher H, Salant DJ, Muller CA, Kalluri R, Zeisberg M (2010) Methylation determines fibroblast activation and fibrogenesis in the kidney. Nat Med 16:544–550
Bennett-Baker PE, Wilkowski J, Burke DT (2003) Age-associated activation of epigenetically repressed genes in the mouse. Genetics 165:2055–2062
Bird AP (1986) CpG-rich islands and the function of DNA methylation. Nature 321:209–213
Bird A (2002) DNA methylation patterns and epigenetic memory. Genes Dev 16:6–21
Blackledge NP, Klose R (2011) CpG island chromatin: a platform for gene regulation. Epigenetics 6:147–152
Brenner BM, Garcia DL, Anderson S (1988) Glomeruli and blood pressure. Less of one, more the other? Am J Hypertens 1:335–347
Brilli LL, Swanhart LM, de Caestecker MP, Hukriede NA (2013) HDAC inhibitors in kidney development and disease. Pediatr Nephrol 28:1909–1921
Brunskill EW, Aronow BJ, Georgas K, Rumballe B, Valerius MT, Aronow J, Kaimal V, Jegga AG, Yu J, Grimmond S, McMahon AP, Patterson LT, Little MH, Potter SS (2008) Atlas of gene expression in the developing kidney at microanatomic resolution. Dev Cell 15:781–791
Bush EW, McKinsey TA (2010) Protein acetylation in the cardiorenal axis: the promise of histone deacetylase inhibitors. Circ Res 106:272–284
Cai Y, Lechner MS, Nihalani D, Prindle MJ, Holzman LB, Dressler GR (2002) Phosphorylation of Pax2 by the c-Jun N-terminal kinase and enhanced Pax2-dependent transcription activation. J Biol Chem 277:1217–1222
Cai Y, Brophy PD, Levitan I, Stifani S, Dressler GR (2003) Groucho suppresses Pax2 transactivation by inhibition of JNK-mediated phosphorylation. EMBO J 22:5522–5529
Carroll TJ, Park JS, Hayashi S, Majumdar A, McMahon AP (2005) Wnt9b plays a central role in the regulation of mesenchymal to epithelial transitions underlying organogenesis of the mammalian urogenital system. Dev Cell 9:283–292
Chen S, Bellew C, Yao X, Stefkova J, Dipp S, Saifudeen Z, Bachvarov D, El-Dahr SS (2011) Histone deacetylase (HDAC) activity is critical for embryonic kidney gene expression, growth, and differentiation. J Biol Chem 286:32775–32789
Cheng X, Blumenthal RM (2010) Coordinated chromatin control: structural and functional linkage of DNA and histone methylation. Biochemistry 49:2999–3008
Cho YW, Hong T, Hong S, Guo H, Yu H, Kim D, Guszczynski T, Dressler GR, Copeland TD, Kalkum M, Ge K (2007) PTIP associates with MLL3- and MLL4-containing histone H3 lysine 4 methyltransferase complex. J Biol Chem 282:20395–20406
Colaneri A, Staffa N, Fargo DC, Gao Y, Wang T, Peddada SD, Birnbaumer L (2011) Expanded methyl-sensitive cut counting reveals hypomethylation as an epigenetic state that highlights functional sequences of the genome. Proc Natl Acad Sci USA 108:9715–9720
de Groh ED, Swanhart LM, Cosentino CC, Jackson RL, Dai W, Kitchens CA, Day BW, Smithgall TE, Hukriede NA (2010) Inhibition of histone deacetylase expands the renal progenitor cell population. J Am Soc Nephrol 21:794–802
Deaton AM, Webb S, Kerr AR, Illingworth RS, Guy J, Andrews R, Bird A (2011) Cell type-specific DNA methylation at intragenic CpG islands in the immune system. Genome Res 21:1074–1086
DeLange RJ, Fambrough DM, Smith EL, Bonner J (1968) Calf and pea histone IV. I. Amino acid compositions and the identical COOH-terminal 19-residue sequence. J Biol Chem 243:5906–5913
Dressler GR (2008) Epigenetics, development, and the kidney. J Am Soc Nephrol 19:2060–2067
Dressler GR (2009) Advances in early kidney specification, development and patterning. Development 136:3863–3874
Dressler GR, Wilkinson JE, Rothenpieler UW, Patterson LT, Williams-Simons L, Westphal H (1993) Deregulation of Pax-2 expression in transgenic mice generates severe kidney abnormalities. Nature 362:65–67
Ferguson-Smith AC, Surani MA (2001) Imprinting and the epigenetic asymmetry between parental genomes. Science 293:1086–1089
Fraga MF, Esteller M (2007) Epigenetics and aging: the targets and the marks. Trends Genet 23:413–418
Gershey EL, Vidali G, Allfrey VG (1968) Chemical studies of histone acetylation. The occurrence of epsilon-N-acetyllysine in the f2a1 histone. J Biol Chem 243:5018–5022
Goldknopf IL, Taylor CW, Baum RM, Yeoman LC, Olson MO, Prestayko AW, Busch H (1975) Isolation and characterization of protein A24, a "histone-like" non-histone chromosomal protein. J Biol Chem 250:7182–7187
Huang N, Tan L, Xue Z, Cang J, Wang H (2012) Reduction of DNA hydroxymethylation in the mouse kidney insulted by ischemia reperfusion. Biochem Biophys Res Commun 422:697–702
Illingworth R, Kerr A, Desousa D, Jorgensen H, Ellis P, Stalker J, Jackson D, Clee C, Plumb R, Rogers J, Humphray S, Cox T, Langford C, Bird A (2008) A novel CpG island set identifies tissue-specific methylation at developmental gene loci. PLoS Biol 6:e22
Issaeva I, Zonis Y, Rozovskaia T, Orlovsky K, Croce CM, Nakamura T, Mazo A, Eisenbach L, Canaani E (2007) Knockdown of ALR (MLL2) reveals ALR target genes and leads to alterations in cell adhesion and growth. Mol Cell Biol 27:1889–1903
Jaenisch R (1997) DNA methylation and imprinting: why bother? Trends Genet 13:323–329
Jenuwein T, Allis CD (2001) Translating the histone code. Science 293:1074–1080
Jones PL, Veenstra GJ, Wade PA, Vermaak D, Kass SU, Landsberger N, Strouboulis J, Wolffe AP (1998) Methylated DNA and MeCP2 recruit histone deacetylase to repress transcription. Nat Genet 19:187–191
Kikuchi R, Kusuhara H, Hattori N, Kim I, Shiota K, Gonzalez FJ, Sugiyama Y (2007) Regulation of tissue-specific expression of the human and mouse urate transporter 1 gene by hepatocyte nuclear factor 1 alpha/beta and DNA methylation. Mol Pharmacol 72:1619–1625
Kikuchi R, Yagi S, Kusuhara H, Imai S, Sugiyama Y, Shiota K (2010) Genome-wide analysis of epigenetic signatures for kidney-specific transporters. Kidney Int 78:569–577
Kim GD, Ni J, Kelesoglu N, Roberts RJ, Pradhan S (2002) Co-operation and communication between the human maintenance and de novo DNA (cytosine-5) methyltransferases. EMBO J 21:4183–4195
Kleinsmith LJ, Allfrey VG, Mirsky AE (1966) Phosphoprotein metabolism in isolated lymphocyte nuclei. Proc Natl Acad Sci USA 55:1182–1189
Kohli RM, Zhang Y (2013) TET enzymes, TDG and the dynamics of DNA demethylation. Nature 502:472–479
Komohara Y, Suekane S, Noguchi M, Matsuoka K, Yamada A, Itoh K (2007) Expression of APOBEC3G in kidney cells. Tissue Antigens 69:95–98
Kurdistani SK, Grunstein M (2003) Histone acetylation and deacetylation in yeast. Nat Rev Mol Cell Biol 4:276–284
Laird PW, Jaenisch R (1996) The role of DNA methylation in cancer genetic and epigenetics. Annu Rev Genet 30:441–464
Lefevre GM, Patel SR, Kim D, Tessarollo L, Dressler GR (2010) Altering a histone H3K4 methylation pathway in glomerular podocytes promotes a chronic disease phenotype. PLoS Genet 6:e1001142
Lehnertz B, Ueda Y, Derijck AA, Braunschweig U, Perez-Burgos L, Kubicek S, Chen T, Li E, Jenuwein T, Peters AH (2003) Suv39h-mediated histone H3 lysine 9 methylation directs DNA methylation to major satellite repeats at pericentric heterochromatin. Curr Biol 13:1192–1200
Liang P, Song F, Ghosh S, Morien E, Qin M, Mahmood S, Fujiwara K, Igarashi J, Nagase H, Held WA (2011) Genome-wide survey reveals dynamic widespread tissue-specific changes in DNA methylation during development. BMC Genomics 12:231
Ling C, Groop L (2009) Epigenetics: a molecular link between environmental factors and type 2 diabetes. Diabetes 58:2718–2725
Litt MD, Simpson M, Gaszner M, Allis CD, Felsenfeld G (2001) Correlation between histone lysine methylation and developmental changes at the chicken beta-globin locus. Science 293:2453–2455
Little MH, McMahon AP (2012) Mammalian kidney development: principles, progress, and projections. Cold Spring Harb Perspect Biol 4
Manke IA, Lowery DM, Nguyen A, Yaffe MB (2003) BRCT repeats as phosphopeptide-binding modules involved in protein targeting. Science 302:636–639
Maunakea AK, Nagarajan RP, Bilenky M, Ballinger TJ, D'Souza C, Fouse SD, Johnson BE, Hong C, Nielsen C, Zhao Y, Turecki G, Delaney A, Varhol R, Thiessen N, Shchors K, Heine VM, Rowitch DH, Xing X, Fiore C, Schillebeeckx M, Jones SJ, Haussler D, Marra MA, Hirst M, Wang T, Costello JF (2010) Conserved role of intragenic DNA methylation in regulating alternative promoters. Nature 466:253–257
McLaughlin N, Wang F, Saifudeen Z, El-Dahr SS (2013a) In situ histone landscape of nephrogenesis. Epigenetics 9:222–235
McLaughlin N, Yao X, Li Y, Saifudeen Z, El-Dahr SS (2013b) Histone signature of metanephric mesenchyme cell lines. Epigenetics 8:970–978
Monk M (1990) Changes in DNA methylation during mouse embryonic development in relation to X-chromosome activity and imprinting. Philos Trans R Soc Lond B 326:299–312
Murray K (1964) The Occurrence of Epsilon-N-Methyl Lysine in Histones. Biochemistry 3:10–15
Nan X, Ng HH, Johnson CA, Laherty CD, Turner BM, Eisenman RN, Bird A (1998) Transcriptional repression by the methyl-CpG-binding protein MeCP2 involves a histone deacetylase complex. Nature 393:386–389
Ord MG, Stocken LA (1967) Phosphate and thiol groups in histone f3 from rat liver and thymus nuclei. Biochem J 102:631–636
O'Sullivan L, Combes AN, Moritz KM (2012) Epigenetics and developmental programming of adult onset diseases. Pediatr Nephrol 27:2175–2182
Patel SR, Kim D, Levitan I, Dressler GR (2007) The BRCT-domain containing protein PTIP links PAX2 to a histone H3, lysine 4 methyltransferase complex. Dev Cell 13:580–592
Quaggin SE, Kreidberg JA (2008) Development of the renal glomerulus: good neighbors and good fences. Development 135:609–620
Rea S, Eisenhaber F, O'Carroll D, Strahl BD, Sun ZW, Schmid M, Opravil S, Mechtler K, Ponting CP, Allis CD, Jenuwein T (2000) Regulation of chromatin structure by site-specific histone H3 methyltransferases. Nature 406:593–599
Reddy MA, Natarajan R (2011) Epigenetics in diabetic kidney disease. J Am Soc Nephrol 22:2182–2185
Reidy KJ, Rosenblum ND (2009) Cell and molecular biology of kidney development. Semin Nephrol 29:321–337
Reik W, Walter J (2001) Evolution of imprinting mechanisms: the battle of the sexes begins in the zygote. Nat Genet 27:255–256
Rhyu DY, Yang Y, Ha H, Lee GT, Song JS, Uh ST, Lee HB (2005) Role of reactive oxygen species in TGF-beta1-induced mitogen-activated protein kinase activation and epithelial-mesenchymal transition in renal tubular epithelial cells. J Am Soc Nephrol 16:667–675
Riggs AD,Porter TN(1996)Overviewofepigeneticmechanisms.In:Russo VEA,Martienssen RA,Riggs AD(eds)Epigeneticmechanismsofgeneregulation.ColdSpringHarborLaboratory,ColdSpringHarbor,pp29–45
Ritz E, Amann K, Koleganova N, Benz K (2011) Prenatal programming-effects on blood pressure and renal function. Nat Rev Nephrol 7:137–144
Rosenberg SL, Chen S, McLaughlin N, El-Dahr SS (2011) Regulation of kidney development by histone deacetylases. Pediatr Nephrol 26:1445–1452
Rusche LN, Kirchmaier AL, Rine J (2003) The establishment, inheritance, and function of silenced chromatin in Saccharomyces cerevisiae. Annu Rev Biochem 72:481–516
Santos-Rosa H, Schneider R, Bannister AJ, Sherriff J, Bernstein BE, Emre NC, Schreiber SL, Mellor J, Kouzarides T (2002) Active genes are tri-methylated at K4 of histone H3. Nature 419:407–411
Saxen L, Sariola H (1987) Early organogenesis of the kidney. Pediatr Nephrol 1:385–392
Sharma M, Brantley JG, Vassmer D, Chaturvedi G, Baas J, Vanden Heuvel GB (2009) The homeodomain protein Cux1 interacts with Grg4 to repress p27 kip1 expression during kidney development. Gene 439:87–94
Shiio Y, Eisenman RN (2003) Histone sumoylation is associated with transcriptional repression. Proc Natl Acad Sci USA 100:13225–13230
Song F, Mahmood S, Ghosh S, Liang P, Smiraglia DJ, Nagase H, Held WA (2009) Tissue specific differentially methylated regions (TDMR): changes in DNA methylation during development. Genomics 93:130–139
Song R, Van Buren T, Yosypiv IV (2010) Histone deacetylases are critical regulators of the renin-angiotensin system during ureteric bud branching morphogenesis. Pediatr Res 67:573–578
Stadler MB, Murr R, Burger L, Ivanek R, Lienert F, Scholer A, van Nimwegen E, Wirbelauer C, Oakeley EJ, Gaidatzis D, Tiwari VK, Schubeler D (2011) DNA-binding factors shape the mouse methylome at distal regulatory regions. Nature 480:490–495
Staruschenko A (2013) Aldosterone-dependent trans-activation and epigenetic derepression of ENaC: where is the balance? Am J Physiol Ren Physiol 305:F968–F969
Suetake I, Shinozaki F, Miyagawa J, Takeshima H, Tajima S (2004) DNMT3L stimulates the DNA methylation activity of Dnmt3a and Dnmt3b through a direct interaction. J Biol Chem 279:27816–27823
Sun W, Guan M, Li X (2014) 5-hydroxymethylcytosine-mediated DNA demethylation in stem cells and development. Stem Cells Dev (in press)
Surendran K, Kopan R (2011) Chromatin-based mechanisms of renal epithelial differentiation. J Am Soc Nephrol 22:1208–1212
Szemes M, Dallosso AR, Melegh Z, Curry T, Li Y, Rivers C, Uney J, Magdefrau AS, Schwiderski K, Park JH, Brown KW, Shandilya J, Roberts SG, Malik K (2013) Control of epigenetic states by WT1 via regulation of de novo DNA methyltransferase 3A. Hum Mol Genet 22:74–83
Takano K, Kawasaki Y, Imaizumi T, Matsuura H, Nozawa R, Tannji M, Suyama K, Isome M, Suzuki H, Hosoya M (2007) Development of glomerular endothelial cells, podocytes and mesangial cells in the human fetus and infant. Tohoku J Exp Med 212:81–90
Tamaru H, Zhang X, McMillen D, Singh PB, Nakayama J, Grewal SI, Allis CD, Cheng X, Selker EU (2003) Trimethylated lysine 9 of histone H3 is a mark for DNA methylation in Neurospora crassa. Nat Genet 34:75–79
Tonna S, El-Osta A, Cooper ME, Tikellis C (2010) Metabolic memory and diabetic nephropathy: potential role for epigenetic mechanisms. Nat Rev Nephrol 6:332–341
Ueda K, Omachi A, Kawaichi M, Hayaishi O (1975) Natural occurrence of poly(ADP-ribosyl) histones in rat liver. Proc Natl Acad Sci USA 72:205–209
Vidali G, Gershey EL, Allfrey VG (1968) Chemical studies of histone acetylation. The distribution of epsilon-N-acetyllysine in calf thymus histones. J Biol Chem 243:6361–6366
Villeneuve LM, Natarajan R (2010) The role of epigenetics in the pathology of diabetic complications. Am J Physiol Ren Physiol 299:F14–F25
Vincent A, Van Seuningen I (2009) Epigenetics, stem cells and epithelial cell fate. Differentiation 78:99–107
Wade PA, Gegonne A, Jones PL, Ballestar E, Aubry F, Wolffe AP (1999) Mi-2 complex couples DNA methylation to chromatin remodelling and histone deacetylation. Nat Genet 23:62–66
Wiggins JE, Goyal M, Sanden SK, Wharram BL, Shedden KA, Misek DE, Kuick RD, Wiggins RC (2005) Podocyte hypertrophy, "adaptation," and "decompensation" associated with glomerular enlargement and glomerulosclerosis in the aging rat: prevention by calorie restriction. J Am Soc Nephrol 16:2953–2966
Wiggins JE, Goyal M, Wharram BL, Wiggins RC (2006) Antioxidant ceruloplasmin is expressed by glomerular parietal epithelial cells and secreted into urine in association with glomerular aging and high-calorie diet. J Am Soc Nephrol 17:1382–1387
Wing MR, Devaney JM, Joffe MM, Xie D, Feldman HI, Dominic EA, Guzman NJ, Ramezani A, Susztak K, Herman JG, Cope L, Harmon B, Kwabi-Addo B, Gordish-Dressman H, Go AS, He J, Lash JP, Kusek JW, Raj DS (2014) DNA methylation profile associated with rapid decline in kidney function: findings from the CRIC Study. Nephrol Dial Transplant 29(4):864–872
Wu H, Zhang Y (2014) Reversing DNA methylation: mechanisms, genomics, and biological functions. Cell 156:45–68
Yokote S, Yamanaka S, Yokoo T (2012) De novo kidney regeneration with stem cells. J Biomed Biotechnol 2012:453519
Yosypiv IV, Boh MK, Spera MA, El-Dahr SS (2008) Downregulation of Spry-1, an inhibitor of GDNF/Ret, causes angiotensin II-induced ureteric bud branching. Kidney Int 74:1287–1293
Acknowledgments
We apologize to the colleagues whose work has not been cited because of length restrictions. We thank all members of our laboratory for their support and helpful discussions. This work is supported by grants to W.B. (Marie Curie EU grant (CIG 293568) and Margarete von Wrangell Habilitationsprogramm, Ministerium für Wissenschaft Baden-Württemberg) and T.B.H. (DFG, BMBF Gerontosys II - NephAge (031 5896A), BIOSS and Excellence Initiative of the German Federal and State Governments (EXC 294).
Disclosures
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bechtel-Walz, W., Huber, T.B. Chromatin dynamics in kidney development and function. Cell Tissue Res 356, 601–608 (2014). https://doi.org/10.1007/s00441-014-1884-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-014-1884-y