Abstract
Thrombin-related peptide 508 (TP508) accelerates bone regeneration during distraction osteogenesis (DO). We have examined the effect of TP508 on bone regeneration during DO by immunolocalization of Runx2 protein, a marker of osteoblast differentiation, and of osteopontin (OPN) and bone sialoprotein (BSP), two late markers of the osteoblast lineage. Distraction was performed in tibiae of rabbits over a period of 6 days. TP508 (30 or 300 μg) or vehicle was injected into the distraction gap at the beginning and end of the distraction period. Two weeks after active distraction, tissue samples were harvested and processed for immunohistochemical analysis. We also tested the in vitro effect of TP508 on Runx2 mRNA expression in osteoblast-like (MC3T3-E1) cells by polymerase chain reaction analysis. Runx2 and OPN protein were observed in preosteoblasts, osteoblasts, osteocytes of newly formed bone, blood vessel cells and many fibroblast-like cells of the soft connective tissue. Immunostaining for BSP was more restricted to osteoblasts and osteocytes. Significantly more Runx2- and OPN-expressing cells were seen in the group treated with 300 μg TP508 than in the control group injected with saline or with 30 μg TP508. However, TP508 failed to increase Runx2 mRNA levels significantly in MC3T3-E1 cells after 2–3 days of exposure. Our data suggest that TP508 enhances bone regeneration during DO by increasing the proportion of cells of the osteoblastic lineage. Clinically, TP508 may shorten the healing time during DO; this might be of benefit when bone regeneration is slow.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Distraction osteogenesis (DO) is a well-established technique, originally developed in orthopedic surgery for bone lengthening (Kojimoto et al. 1988) and later also used to treat hereditary malformations in the craniomaxillofacial region (Amir et al. 2006). DO is characterized by the formation of new bone between two osteotomized bone segments, which are separated by gradual traction. One of the clinical phases of the distraction technique is the bone consolidation period, defined as the time between the end of active distraction and the removal of the distraction device. It represents the time needed for a complete bridging of the distraction gap by bone and a further maturation of this bone. Typically, the bone consolidation phase takes approximately 6–12 weeks in the craniomaxillofacial region and 3–6 months in long bones (Fischgrund et al. 1994; Smith et al. 1999; Felemovicius et al. 2000; Amir et al. 2006). Although DO is believed to be superior to other bone augmentation techniques, many attempts have been made to improve the technique to enhance the bone regeneration process in the distraction gap (Hagiwara and Bell 2000; Li et al. 2002; Mofid et al. 2002; Takamine et al. 2002; Schortinghuis et al. 2005). Such improvements would provide the opportunity to shorten the bone consolidation period and hence minimize complications such as the development of non-union, infection or fracture. Various methods have been tested to promote bone formation in the distraction gap, e.g. electrical and mechanical stimulation (Hagiwara and Bell 2000; Mofid et al. 2002), transplantation of osteoblast-like cells (Takamine et al. 2002) or administration of growth factors such as bone morphogenetic proteins (Li et al. 2002) or fibroblast growth factor 2 (FGF-2; Okazaki et al. 1999). Another procedure to accelerate bone regeneration involves the application of thrombin-related peptide 508 (TP508; Ryaby et al. 2000; Sheller et al. 2004; Li et al. 2005a,b; Wang et al. 2005).
TP508 is a synthetic peptide consisting of 23 amino acids, which represent the natural amino acid sequence of the receptor-binding domain of human thrombin (pro-thrombin amino acids 508–530; Sower et al. 1999; Stiernberg et al. 2000). Thrombin is an important factor in blood homeostasis, inflammation and wound healing (Coughlin et al. 1992). It plays a role in the formation of fibrin clots and platelet activation. Thrombin also stimulates the cell proliferation, migration and/or differentiation of various cell types, such as fibroblasts, endothelial cells and lymphocytes. All these cell types contain one or more thrombin receptors (Chen and Buchanan 1975; Belloni et al. 1992). TP508 appears to mimic and accelerate many of the effects of native thrombin in initiating wound healing through a non-proteolytic pathway (Sower et al. 1999). Histological studies have indicated that TP508 is chemotactic for neutrophils, lymphocytes and monocytic cells; it enhances the formation of granulation tissue, neovascularization and other wound-healing events (Sower et al. 1999; Ryaby et al. 2000; Naldini et al. 2004).
TP508 has also been tested for its potential to enhance bone formation (Ryaby et al. 2000; Sheller et al. 2004; Li et al. 2005a,b; Wang et al. 2005). A single injection of 1 μg TP508 accelerates fracture healing in rat long bone fracture. The breaking strength of bones of young and aged rats injected with TP508 has been enhanced over that of controls (Ryaby et al. 2000). One recent study of TP508 treatment during DO has revealed a significantly greater bone mineral density in TP508-treated groups than in saline-treated controls (Li et al. 2005a,b). Histological analysis has also shown advanced bone consolidation and remodeling after TP508 treatment.
In the present study, we report the effect of TP508 in the regulation of bone regeneration during DO in more detail. We have examined Runx2/Cbfa1, osteopontin (OPN) and bone sialoprotein (BSP) protein expression in tissue sections of rabbit long bones undergoing DO. Runx2 is an essential transcription factor for osteoblast differentiation and for extracellular matrix gene expression (Komori et al. 1997; Karsenty et al. 1999). In Runx2 knockout mice, both intramembranous and endochondral ossification are blocked because of the maturational arrest of osteoblasts (Komori et al. 1997). Runx2 can directly stimulate the transcription of the collagen type I and osteoblast-related genes, such as those for OPN and BSP, both major non-collagenous proteins found in the extracellular matrix of bone (Karsenty et al. 1999). We have hypothesized that TP508 treatment during DO enhances the expression of Runx2, OPN and BSP.
Materials and methods
Animal model of DO
The paraffin blocks containing distracted rabbit long bones were those prepared previously (Li et al. 2005a,b) from 30 adult male New Zealand White rabbits (age 24 weeks, body weight 2.6–3.5 kg). External fixators were applied to stabilize the osteotomized mid-tibia for 7 days of the latency period and were activated for 6 days, once a day at a rate of 1.4 mm. This distraction rate was intentionally chosen in order to copy the poor bone healing condition as previously described (Li et al. 1997).
During the experiment, seven animals were excluded in the study because of soft tissue complications (n = 2), pinhole fracture (n = 3) and anaesthetic death (n = 2). The remaining 23 rabbits were randomly divided into three experimental groups, each group consisting of minimally seven rabbits. TP508 (OrthoLogic, Tempe, USA) at two different concentrations (30 and 300 μg TP508) or saline were injected percutanously at the beginning and at the end of the lengthening phase. The first group (n = 8) received injections of 300 μl saline containing 300 μg TP508, the second group (n = 8) received injections of 300 μl saline containing 30 μg TP508 and the third group (n = 7) received injections of 300 μl saline alone and served as a control group. The injections were given in three different areas (100 μl in each area): the proximal, central and distal area of the distraction gap. Two weeks after the lengthening phase, all animals were killed. Tissue samples consisting of the distraction gap and 5 mm proximal and distal to the pre-existing bone were then harvested and immediately fixed in 95% ethanol for further examination. Tissues were decalcified with EDTA and embedded in paraffin. Serial sections (5 μm thick) were cut and mounted on poly-lysine-coated glass slides. The peripheral quantitative computed tomography and histological examination of these bones has been published previously (Li et al. 2005a,b).
All animal experimental procedures were approved and performed following the guidelines for animal scientific procedures (Animal Scientific Procedures Act 1986, British Home Office).
Antibodies
The mouse recombinant monoclonal antibody Pebp2alphaA was kindly donated by Dr. K. Sasaguri, Kanawaga Dental School, Japan. This antibody has been used and characterized previously and reacts with all isotypes of Runx2 (Bronckers et al. 2001, 2005). The mouse anti-rat OPN monoclonal antibody (MPIIIB101) was obtained from the Developmental Studies Hybridoma Bank (developed under the auspices of the NICHD and maintained by the University of Iowa, Department of Biological Sciences Iowa City, IA 52242, USA). The mouse anti-human BSP (monoclonal antibody A4232.1) was purchased from Immunodiagnostik, Bensheim, Germany. Both anti-OPN and anti-BSP antibodies have been used and specified previously (Ibrahim et al. 2000; Bronckers et al. 2005; Zerbo et al. 2005).
Immunohistochemical procedures
Tissue sections were de-paraffinized in xylene and rehydrated through a graded series of aqueous ethanol solutions. Endogenous peroxidase was quenched by incubation in 3% H2O2 in 100% ethanol (5 min). A pre-incubation in hot citrate buffer (0.01 M, pH 6.0, 95°C, 20 min) was performed for antigen retrieval prior to incubation with 30% normal horse serum (30 min). Sections were then incubated overnight with primary monoclonal anti-Runx2 antibody (1:200), anti-BSP antibody (1:400) or anti-OPN antibody (1:600) at 4°C in a humidified chamber. After washes with phosphate-buffered saline (PBS), biotinylated horse anti-mouse IgG (ABC-peroxidase Elite kit; Vector Laboratories, Burlingame, Calif., USA) was added at a dilution of 1:200 and the sections were incubated at room temperature for a minimum of 60 min. The sections were washed again with PBS, incubated with ABC peroxidase complex for another 60 min followed by a maximum of 20 min in 3,3′ diaminobenzidine substrate solution (Vectastain kit) to visualize the immunoreaction, counterstained with methyl green for 2–3 min, mounted in 50% glycerol and covered with a glass coverslip. As negative controls, sections were incubated with non-immune mouse IgG in place of the primary antibody.
Scoring
Sections were examined by using a Leica DM RA microscope equipped with a Leica DC 200 digital camera. The central areas of the gap were selected as the region of interest (ROI). First, the boundaries of the distraction gap (indicated by the osteotomy lines) were determined and subsequently a grid was positioned over the ROI halfway between the two osteotomy lines. The grid was 609 μm in width and 479 μm in length. The ROI consisted of some newly formed bone (mostly in the group treated with 300 μg TP508) and fibrous tissue (Fig. 1a). Counting was performed by using the grid at a magnification of 200×, with a minimum of three sections, at least 30 μm apart, per animal. Some tissue blocks were of poor quality or no longer contained a complete intact gap area with an ROI; this reduced the number of animals per group to 5–6 animals. The total number of cells was counted (immunopositive and immunonegative cells). The sections were coded and randomized. Quantification was performed by one investigator (L.A.) who was unaware of the origin of the sections. Numbers of immunopositive cells were noted in absolute terms and were expressed as the percentage of all (immunopositive and negative) cells present in the ROI.
Immunolocalization of Runx2 and osteopontin proteins in TP508-treated groups (a-c, e 300 μg TP508 group, d 30 μg TP508 group). a Overview of immunoreactions in a biopsy taken from rabbit long bone after distraction osteogenesis Runx2 staining (FT fibrous tissue, NB new bone, P periosteum, PC periosteal callus). A white grid was used to count the number of immunopositive and negative cells in the region of interest. ×50. b Runx2 staining in the nuclei and cytoplasm of the cells. Runx2 was strongly expressed in osteoblasts (Ob). Note that young osteocytes (Ocy), which had recently embedded in the bone matrix, expressed Runx2 protein. Cells adjacent to the osteoblast layer, presumably pre-osteoblasts (Pre-Ob), also expressed Runx2, but with lower intensity (B bone). ×400. c Immunostaining for osteopontin. Positive staining was found in the cytoplasm of the cells (white arrows). Note that expression was stronger towards the newly formed bone (NB). ×200. d Runx2 immunostaining in tissue of a rabbit injected with 30 μg TP508. Expression was much lower than that after injection with 300 μg TP508 and was similar to saline-injected control tissue (not shown). Fewer fibrocartilage-like cells (FCC) were stained than after injection with 300 μg TP508 (HC hypertrophic cartilage-like cells). ×200. e Negative control stained with non-immune IgG instead of primary antibodies to Runx2. Note the lack of positive staining. Counter-staining with methyl green. ×200. Bars 20 μm (b, d), 50 μm (c, e), 300 μm (a)
Cell cultures
The mouse osteoblast-like MC3T3-E1 cell line, derived from newborn mouse calvaria, was routinely maintained in α-MEM (Gibco, Paisley, UK) supplemented with 10% fetal bovine serum (HyClone), 120 μg/ml penicillin (Sigma, St Louis, Mo., USA), 100 μg/ml streptomycin sulphate (Sigma), 1.25 μg/ml fungizone (Gibco), 50 μg/ml sodium ascorbate (Merck, Darmstadt, Germany), 10 mM β-glycerophosphate (Sigma) and 300 μg/ml glutamine (Sigma) at 37°C in a humidified atmosphere of 5% CO2 in air. This cell line is capable of expressing Runx2 mRNA (Tsuji et al. 1998). To stimulate cells to express osteoblastic markers, they were first exposed to 10 nM dexamethasone in 25 cm2 flasks (Greiner BioOne, Solingen, Germany) for 1 week. Upon confluency (typically after 5 days), cells were harvested by using 0.25% trypsin and 0.1% EDTA in PBS, centrifuged at 600g for 10 min, washed and plated in 24-well culture dishes (Greiner BioOne) at three different cell densities (3×104, 1.5×104 and 7.5×103 cells/well) in order to obtain a comparable number of cells at the end of each experiment. After 1 day of culture, the medium was replaced by media containing various concentrations of TP508 (0, 10 or 100 μg/ml). Throughout the experiment, all media were supplemented with 10 nM dexamethasone. Cells were exposed to TP508 for 24, 48 and 72 h, after which times, the cells were collected. The experiment was carried out in quadruplicate and repeated once.
RNA analysis and quantitative real-time polymerase chain reaction
Total RNA from cultured cells was isolated by using TRIZOL reagent (Gibco) according to the manufacturer’s instructions. The RNA content was determined by measuring the absorbance in water at 260 nm by means of an Ultrospec III spectrophotometer (Amersham, Buckinghamshire, England). cDNA synthesis was performed by using 750 ng total RNA in a final reaction volume of 20 μl containing 5 U transcriptor (Roche Applied Science), 5× polymerase chain reaction (PCR) buffer, 4 U random primers (Roche), 20 U protector RNase inhibitor (Roche), 1 mmol each dNTP and 10 μl template. The reverse transcription step was performed on a Gene Amp 9700 Thermocycler (Applied Biosystems, Foster City, Calif., USA) at 55°C for 30 min followed by 85°C for 5 min.
Real-time PCR was performed on the ABI PRISM 7700 sequence detection system (Applied Biosystems). The phosphobilinogen deaminase gene (PBGD) served as the endogenous reference (de Vries et al. 1999) to normalize Runx2 expression. The primers for the amplification of Runx2 mRNA were 5′-ATGCTTCATTCGCCTCAC-3′ and 5′-ACTGCTTGCAGCCTTAAAT-3′ (GenBank database, accession no. NM 001024630). The PCR primers for PBGD, amplified as the internal reference, were 5′-AGTGATGAAAGATGGGCAACT-3′ and 5′-TCTGGACCATCTTCTTGCTGA-3′ (accession no. BC 003861). For the amplification of the Runx2 and PBGD products, 37.5 ng cDNA was added to the PCR mixture containing SYBR Green PCR Master Mix consisting of SYBR Green I Dye, AmpliTaq Gold DNA polymerase, dNTPs with dUTP, a passive reference and buffer (Applied Biosystems) and 300 nM of each primer, in a final volume of 25 μl. The enzyme was activated by preheating the samples for 10 min at 95°C, followed by a two-step PCR procedure consisting of a denaturation step at 95°C for 15 s and an annealing and extension step at 60°C for 1 min for 45 cycles. Relative expression was calculated by using the comparative Ct method. Samples were normalized for the expression of PBGD by calculating ΔCt (CtRunx2−CtPBGD); subsequently, the ΔΔCt values were calculated as ΔCtsample−ΔCtcalibrator , where the calibrator was the control sample (without TP508 incubation). Relative expression of the Runx2 gene was expressed as 2-(ΔΔ Ct) (Livak and Schmittgen 2001).
Statistical analysis
Values obtained from the scoring of tissue sections and normalized mRNA levels determined by PCR analysis (presented as means and standard deviations) were analysed by using GraphPad Prism 4 for Windows. For the analysis of the immunostaining, an average value was calculated per animal for each protein and treatment; these average values were used to determine the mean and standard deviation for each treatment group (5–6 animals/group). Analysis of variance (ANOVA) was used to test for statistical significance, which was accepted when P<0.05 (two-tailed). Tukey’s post test was performed to compare individual pairs of groups if P<0.05. Linear regression analysis was used to examine the correlation between Runx2 mRNA expression and culture time.
Results
Immunohistochemistry demonstrated different localization patterns for Runx2, OPN and BSP proteins. Cells expressing these proteins were detected throughout the distraction gap and in the periosteum, the periosteal callus and the old (predistraction) bone (Fig. 1a). Runx2 staining was located in nuclei and cytoplasm, whereas strong staining for OPN and BSP was localized in the cytoplasm (Figs. 1b,c,2i,3c); weak staining for BSP and OPN was also seen in the extracellular bone matrix. Figures 1, 2, 3 present the different staining patterns and Table 1 summarizes the average intensity of immunostaining in the ROI.
Runx2 (a, d, g), OPN (b, e, h) and BSP (c, f, i) expression in the distraction gap (B bone, FT fibrous tissue, FC fibrous cartilage-like tissue, asterisks central area of distraction gap, hatch layers of cells adjacent to the newly formed bone, arrows osteoblasts, arrowheads osteocytes) of the various groups (a-c 0 μg TP508 group, d-f 30 μg TP508 group, g-i 300 μg TP508 group). Inset (i): Newly formed bone in the gap. Original magnifications: a 50×; d, g 100×; b, e, h 100×; c, f, i, inset 200×. Bars 20 μm (c, f, i), 50 μm (b, d, e, g, h), 100 μm (a)
Immunolocalization of Runx2, OPN and BSP in the 300 μg TP508 group. a Runx2 staining of a vessel wall (EC endothelial cells, SMC smooth muscle cells). Note that some cells near the blood vessel weakly express Runx2 (white arrows). ×400. b Osteopontin staining of a vessel wall. Note that the cells surrounding the blood vessel are immunonegative and only stain weakly for methyl green (white arrows). ×400. c BSP staining is restricted to the layer of osteoblasts (Ob) and hardly occurs in fibrous tissue × 400. d OPN staining is expressed in osteoblasts (Ob) and osteocytes of new bone (NB) and some fibrous tissue cells close to osteoblasts, presumably pre-osteoblasts (Pre-Ob). ×200. Bars 20 μm (b), 50 μm (c, d), 100 μm (a)
Expression of Runx2, OPN and BSP in the group injected with saline (control group) was similar to that in the group treated with 30 μg TP508 (Figs. 1d, 2a–f). The intensity of the staining in both groups was however much lower than that in the 300 μg TP508 group (Figs. 1c,2g,h,3c,d). Runx2 and OPN were detected in active osteoblasts, in fibroblasts next to the osteoblastic layer (presumably pre-osteoblasts), in osteocytes in the old (predistraction) and newly formed bone, in fibroblasts of the periosteum, in the central area of the gap, in endothelial and smooth muscle cells of the vessels and in chondrocyte-like cells next to new bone. We found two types of cartilage-like tissue in the group treated with 30 μg TP508 and in control group, i.e. fibrocartilage-like (collagen fibres between lacunae) and hypertrophic-like (evenly dispersed chondrocytes, without clear fibres; Fig. 1d). Higher numbers of cells associated with these two tissues were found in the group treated with 30 μg TP508 and in the control group than in the group treated with 300 μg TP508 (Fig. 2a,d,g). More fibrocartilage-like cells than hypertrophic cartilage-like cells were positive for Runx2 (Fig. 1d). Sections in which primary antibodies had been replaced with non-immune antibodies were negative (Fig. 1e).
The Runx2- and OPN-positive blood vessel cells (both the endothelial and smooth muscle cells) were more evident in the group with 300 μg TP508 (Fig. 3a,b). They appeared to occur at locations with high bone formation activity. Connective tissue cells in close vicinity to Runx2-positive blood vessels also stained faintly for Runx2 but not for OPN. Immunostaining for BSP was more restricted than that for Runx2 and OPN and was seen in osteoblasts and young osteocytes (Fig. 3c,d). Weak expression was found in some of the cells considered to be pre-osteoblasts. Fibroblast-like cells in the central fibrous area of the distraction gap expressed Runx2 (Fig. 2a,d,g) and OPN (Fig. 2b,e,h) but were negative for BSP. The number and the intensity of Runx2 and OPN in the gap increased from fibrous tissue towards the newly formed bone (Fig. 1c). Weak BSP staining was seen in the area within the gap in which bone trabecules started to be formed (Fig. 2c,f,i).
In the group injected with 300 μg TP508, immunostaining for Runx2, OPN and BSP was distributed throughout the regenerating areas. The immunostaining for Runx2, OPN and BSP in the connective tissue in the gap (endosteal area) was relatively high and as intense as in the periosteal callus tissue. In the 30 μg TP508 group and in the control group, staining was less intense in the central gap area than in the periosteal callus area.
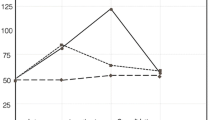
Quantitative immunohistochemistry was performed to investigate the effect of TP508 injection on the total number of cells and on the number of differentiated bone cells in the central area of the gap. The total number of cells (immunostained and unstained) in the ROI was not significantly different between the three groups. Mean values and the standard deviation of the total number of cells in the tissues treated with 0, 30 μg TP508 and 300 μg TP508 were 958±217, 1140±137 and 1161±282, respectively (ANOVA, P=0.5). This indicated that TP508 injection had no overall effect on total number of cells in the distraction gap. Next, we counted the number of cells, in the ROI, expressing each of the three antibodies tested. Absolute values of immunopositive cells in the 0, 30 μg TP508 and 300 μg TP508 groups were as follows: for Runx2, 263±61, 288±171, 560±115, respectively; for OPN, 299±201, 244±40, 677±216, respectively; for BSP, 255±111, 252±73, 480±31, respectively. We also expressed the number of immunopositive cells as a percentage of the total number of cells (Fig. 4). The number of Runx2- and OPN-expressing cells was significantly higher in the group treated with 300 μg TP508 than in the control group that received only saline (P<0.05; Fig. 4). The number of BSP-expressing cells was also higher in the 300 μg TP508 group than in the 30 μg TP508 group (P<0.05). No significant differences were found between the 30 μg TP508 group and the control group receiving saline (P>0.05).
Effect of TP508 injection on the number of immunopositive cells in the central portion of the distraction gap (black bars Runx2-positive cells, grey bars OPN-positive cells, white bars BSP-positive cells). Data are presented as percentage cells immunopositive for each of the three proteins examined (means±SD). *The percentages of Runx2-, OPN- and BSP-positive cells in the 300 μg TP508 group were significantly higher than for cells with the corresponding proteins in the saline-treated control group; the percentage of BSP-positive cells in the 300 μg group was also significantly higher than that in the 30 μg group (ANOVA, P<0.05)
The previous data suggested that TP508 increased the differentiation of osteogenic cells into osteoblasts in situ. To determine whether TP508 had any direct effect on osteogenic cells that were in the process of turning into osteoblasts, we performed in vitro experiments with the (mouse) osteoblast-cell line MC3T3-E1 after pre-culture in the presence of dexamethasone. After a 24-h stimulation with TP508, we found a significant (but slight) downregulation of Runx2 mRNA at 10 μg/ml TP508 (P=0.008) but no change at 100 μg/ml TP508 (Fig. 5).
Quantitative reverse transcription/polymerase chain reaction analysis of Runx2 mRNA expression in mouse osteoblast-like (MC3T3-E1) cells cultured for 24, 48 and 72 h in the presence of TP508 (black bars control group with 0 μg/ml TP508, hatched bars 10 μg/ml TP508, white bars 100 μg/ml TP508). Data are presented as means±SD (n=8) and are representative of two experiments. *The 10 μg/ml TP508 group at 24 h of incubation was significantly lower than the control group (one-way ANOVA; Tukey’s multiple comparison test, P<0.01). Correlation analysis between mRNA and culture time: r2=0.94, P=0.06 for 10 μg/ml TP508; r2=0.99, P=0.06 for 100 μg/ml TP508
Discussion
This study demonstrates a positive effect of TP508 on the synthetic activity of bone-forming cells in the distraction gap in rabbit long bones. After injection of a high dose of TP508, significantly more cells expressing Runx2, OPN and BSP are located in the central part of the distraction gap, predominantly consisting of soft connective tissue. We have also found that these cells stain more intensely for Runx2, OPN and BSP suggesting that TP508 not only proportionally increases the number of osteogenic cells in the distraction gap but also their expression level. The finding that TP508 enhances bone-forming activity in the soft tissue gap area is in agreement with previous morphometric data indicating that TP508 stimulates bone regeneration in the distraction gap (Li et al. 2005a,b).
The underlying molecular mechanism of the stimulation of Runx2 expression in the distraction gap after TP508 treatment is not clear. Our in vitro experiment with osteoblast-like MC3T3-cells suggests that TP508 does not have a significant positive effect on Runx2 mRNA. However, we cannot rule out that the MCT3C3 cells are too far advanced along the osteoblast differentiation pathway to respond to TP508 for they had been pre-cultured in dexamethasone-containing medium before they were exposed to TP508. No definitive conclusions can therefore be drawn from these in vitro data. In vivo, TP508 may have stimulated the differentiation of osteogenic cells though the activity of other non-osteogenic cell types. Injection of TP508 into a bone fracture model upregulates FGF-2 mRNA in (not further specified) tissues at the fracture site (Wang et al. 2005). Since FGF-2 is known to be an upstream regulator of Runx2 (Franceschi and Xiao 2003), one explanation for the increased Runx2 expression in the gap is via an enhanced cytokine release by inflammatory cells, which in turn stimulates osteoblast differentiation (Okazaki et al. 1999; Stiernberg et al. 2000; Franceschi and Xiao 2003; Naldini et al. 2004). Alternatively, TP508 stimulates blood vessel growth in vivo (Norfleet et al. 2000; Li et al. 2005a,b; Vartanian et al. 2005; Wang et al. 2005) and this subsequently enhances bone formation. TP508 increases recruitment of human aortic and microvascular endothelial cells through chemotaxis (Norfleet et al. 2000; Li et al. 2002), upregulates angiogenesis-related genes (Wang et al. 2005) and acts directly on microvascular cells to accelerate sprouting (Vartanian et al. 2005). Vascularization provides the oxygen that is required for the metabolic activity of the osteoblasts (Tuncay et al. 1994; Salim et al. 2004); it provides homes for circulating stem cells that, under proper stimulation, differentiate into osteoblastic cells (Kuznetsov et al. 2001). In addition, vessel cells secrete paracrine factors that can regulate bone cell metabolism (Villars et al. 2000; Street et al. 2002). Thus, the angiogenic properties of TP508 might help to enhance bone growth.
In the present study, we have also found Runx2 protein expression in endothelial cells, vascular smooth muscle cells and loose connective cells surrounding developing blood vessels at sites of active bone and tissue formation. Recent studies have presented evidence for transient Runx2 mRNA and protein expression during angiogenesis in embryonic human orofacial tissues (Bronckers et al. 2005), in human primary bone marrow endothelial cells and in endothelial cell lines (Namba et al. 2000; Sun et al. 2001). Thus, our data suggest that, with respect to Runx2 expression, the formation of blood vessels in regenerating adult tissues during DO resembles vessel formation in embryonic tissues. Of note, under certain conditions such as inflammation, cells associated with vessels (e.g. pericytes, endothelial cells and smooth muscle cells) or their precursors are capable of differentiating into osteoblast-like cells (Schor et al. 1995; Reilly et al. 1998; Kinner and Spector 2002) suggesting that the forming microvasculature is a potential source for osteogenic cells.
The distribution and staining intensity of Runx2, OPN and BSP varies between the different groups. In the control group and in the group treated with a low dose of TP508, staining is more pronounced in the periosteal callus than in the tissue in the distraction gap. However, in the group treated with a high dose of TP508, we have found the same high staining in both periosteal callus and gap tissue. Periosteal callus formation is a common finding during fracture healing (Gerstenfeld et al. 2003). New woven bone is formed rapidly around the fractured segments, because of stimulation of the periosteum that contains many osteoprogenitor cells. In the same section, a high staining intensity in the periosteal callus can therefore be considered as an internal reference. Hence, as the staining for Runx2, OPN and BSP in the distraction gap is as intense as that in the periosteal callus at the high dose of TP508, this is a clear indication that the high dose of TP508 stimulates bone formation in the distraction gap. This observation is in accordance to a previous histological study that has demonstrated advanced bone consolidation in a group treated with a high dose of TP508 (Li et al. 2005b). TP508 might stimulate the migration of osteoblast precursors to the distraction gap (Li et al. 2005a). Li and colleagues have shown that human periosteal-derived osteoblasts and human microvascular endothelial cells are chemotactic to TP508 (Li et al. 2005a). Therefore, the presence of TP508 in the gap might attract osteoprogenitor cells in the periosteum to migrate into the distraction gap.
Runx2 is a critical gene for osteoblast differentiation and for the function of the fully differentiated osteoblast (Komori et al. 1997; Karsenty et al. 1999). The Runx2 protein binds to the osteoblast-specific element 2 that is present in the promoter regions of all major extracellular matrix genes expressed in osteoblasts, including the genes for osteocalcin (OC), OPN and BSP and it regulates the expression of these proteins (Karsenty et al. 1999). OPN and BSP belong to the Small Integrin-Binding LIgand, N-linked Glycoprotein (or SIBLING) family (Fisher et al. 2001). Among other functions, members of this family are known to have a role in mineralization, as they bind strongly to hydroxyapatite (Fisher et al. 2001).
In conclusion, we have demonstrated that the increase of bone regeneration by thrombin-related peptide TP508 is associated with an increase in the immunostaining for Runx2, an essential transcription factor of the osteoblastic lineage, and for the bone matrix proteins BSP and OPN. TP508 may thus be a candidate for enhancing bone regeneration when bone regeneration is slow, as occurs in elderly patients.
References
Amir LR, Becking AG, Jovanovic A, Perdijk FBT, Everts V, Bronckers AL (2006) Vertical distraction osteogenesis in the human mandible: a prospective morphometric study. Clin Oral Implants Res 17:417–425
Belloni PN, Carney DH, Nicolson GL (1992) Organ-derived microvessel endothelial cells exhibit differential responsiveness to thrombin and other growth factors. Microvasc Res 43:20–45
Bronckers AL, Engelse MA, Cavender A, Gaikwad J, D’Souza RN (2001) Cell-specific patterns of Cbfa1 mRNA and protein expression in postnatal murine dental tissues. Mech Dev 101:255–258
Bronckers AL, Sasaguri K, Cavender AC, D’Souza RN, Engelse MA (2005) Expression of Runx2/Cbfa1/Pebp2alphaA during angiogenesis in postnatal rodent and fetal human orofacial tissues. J Bone Miner Res 20:428–437
Chen LB, Buchanan JM (1975) Mitogenic activity of blood components. I. Thrombin and prothrombin. Proc Natl Acad Sci USA 72:131–135
Coughlin SR, Vu TK, Hung DT, Wheaton VI (1992) Characterization of a functional thrombin receptor. Issues and opportunities. J Clin Invest 89:351–355
Felemovicius J, Ortiz MF, Gomez Radillo LS, Serna A (2000) Determining the optimal time for consolidation after distraction osteogenesis. J Craniofac Surg 11:430–436
Fischgrund J, Paley D, Suter C (1994) Variables affecting time to bone healing during limb lengthening. Clin Orthop Relat Res 301:31–37
Fisher LW, Torchia DA, Fohr B, Young MF, Fedarko NS (2001) Flexible structures of SIBLING proteins, bone sialoprotein, and osteopontin. Biochem Biophys Res Commun 280:460–465
Franceschi RT, Xiao G (2003) Regulation of the osteoblast-specific transcription factor, Runx2: responsiveness to multiple signal transduction pathways. J Cell Biochem 88:446–454
Gerstenfeld LC, Cullinane DM, Barnes GL, Graves DT, Einhorn TA (2003) Fracture healing as a post-natal developmental process: molecular, spatial, and temporal aspects of its regulation. J Cell Biochem 88:873–884
Hagiwara T, Bell WH (2000) Effect of electrical stimulation on mandibular distraction osteogenesis. J Craniomaxillofac Surg 28:12–19
Ibrahim T, Leong I, Sanchez-Sweatman O, Khokha R, Sodek J, Tenenbaum HC, Ganss B, Cheifetz S (2000) Expression of bone sialoprotein and osteopontin in breast cancer bone metastases. Clin Exp Metastasis 18:253–260
Karsenty G, Ducy P, Starbuck M, Priemel M, Shen J, Geoffroy V, Amling M (1999) Cbfa1 as a regulator of osteoblast differentiation and function. Bone 25:107–108
Kinner B, Spector M (2002) Expression of smooth muscle actin in osteoblasts in human bone. J Orthop Res 20:622–632
Kojimoto H, Yasui N, Goto T, Matsuda S, Shimomura Y (1988) Bone lengthening in rabbits by callus distraction. The role of periosteum and endosteum. J Bone Joint Surg Br 70:543–549
Komori T, Yagi H, Nomura S, Yamaguchi A, Sasaki K, Deguchi K, Shimizu Y, Bronson RT, Gao YH, Inada M, Sato M, Okamoto R, Kitamura Y, Yoshiki S, Kishimoto T (1997) Targeted disruption of Cbfa1 results in a complete lack of bone formation owing to maturational arrest of osteoblasts. Cell 89:755–764
Kuznetsov SA, Mankani MH, Gronthos S, Satomura K, Bianco P, Robey PG (2001) Circulating skeletal stem cells. J Cell Biol 153:1133–1140
Li G, Simpson AH, Kenwright J, Triffitt JT (1997) Assessment of cell proliferation in regenerating bone during distraction osteogenesis at different distraction rates. J Orthop Res 15:765–772
Li G, Bouxsein ML, Luppen C, Li XJ, Wood M, Seeherman HJ, Wozney JM, Simpson H (2002) Bone consolidation is enhanced by rhBMP-2 in a rabbit model of distraction osteogenesis. J Orthop Res 20:779–788
Li G, Ryaby JT, Carney DH, Wang H (2005a) Bone formation is enhanced by thrombin-related peptide TP508 during distraction osteogenesis. J Orthop Res 23:196–202
Li G, Cui Y, McIlmurray L, Allen WE, Wang H (2005b) rhBMP-2, rhVEGF, rhPTN and thrombin-related peptide, TP508, induce chemotaxis of human osteoblasts and microvascular endothelial cells. J Orthop Res 23:680–685
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408
Mofid MM, Inoue N, Atabey A, Marti G, Chao EY, Manson PN, Vander Kolk CA (2002) Callus stimulation in distraction osteogenesis. Plast Reconstr Surg 109:1621–1629
Naldini A, Carraro F, Baldari CT, Paccani SR, Bernini C, Keherly MJ, Carney DH (2004) The thrombin peptide, TP508, enhances cytokine release and activates signaling events. Peptides 25:1917–1926
Namba K, Abe M, Saito S, Satake M, Ohmoto T, Watanabe T, Sato Y (2000) Indispensable role of the transcription factor PEBP2/CBF in angiogenic activity of a murine endothelial cell MSS31. Oncogene 19:106–114
Norfleet AM, Bergmann JS, Carney DH (2000) Thrombin peptide, TP508, stimulates angiogenic responses in animal models of dermal wound healing, in chick chorioallantoic membranes, and in cultured human aortic and microvascular endothelial cells. Gen Pharmacol 35:249–254
Okazaki H, Kurokawa T, Nakamura K, Matsushita T, Mamada J, Kawaguchi H (1999) Stimulation of bone formation by recombinant fibroblast growth factor-2 in callostasis bone lengthening of rabbits. Calcif Tissue Int 64:542–546
Reilly TM, Seldes R, Luchetti W, Brighton CT (1998) Similarities in the phenotypic expression of pericytes and bone cells. Clin Orthop Relat Res 346:95–103
Ryaby JT, Carney DH, Campbell M, Crowther RS, Yang J, Bain S (2000) Acceleration of fresh fracture healing with an injectable thrombin peptide in a rat model (abstract). Trans ORS 25:877
Salim A, Nacamuli RP, Morgan EF, Giaccia AJ, Longaker MT (2004) Transient changes in oxygen tension inhibit osteogenic differentiation and Runx2 expression in osteoblasts. J Biol Chem 279:40007–40016
Schor AM, Canfield AE, Sutton AB, Arciniegas E, Allen TD (1995) Pericyte differentiation. Clin Orthop Rel Res 313:81–91
Schortinghuis J, Bronckers AL, Stegenga B, Raghoebar GM, Bont LG de (2005) Ultrasound to stimulate early bone formation in a distraction gap: a double blind randomised clinical pilot trial in the edentulous mandible. Arch Oral Biol 50:411–420
Sheller MR, Crowther RS, Kinney JH, Yang J, Di Jorio S, Breunig T, Carney DH, Ryaby JT (2004) Repair of rabbit segmental defects with the thrombin peptide, TP508. J Orthop Res 22:1094–1099
Smith SW, Sachdeva RC, Cope JB (1999) Evaluation of the consolidation period during osteodistraction using computed tomography. Am J Orthod Dentofacial Orthop 116:254–263
Sower LE, Payne DA, Meyers R, Carney DH (1999) Thrombin peptide, TP508, induces differential gene expression in fibroblasts through a nonproteolytic activation pathway. Exp Cell Res 247:422–431
Stiernberg J, Norfleet AM, Redin WR, Warner WS, Fritz RR, Carney DH (2000) Acceleration of full-thickness wound healing in normal rats by the synthetic thrombin peptide, TP508. Wound Repair Regen 8:204–215
Street J, Bao M, deGuzman L, Bunting S, Peale FV Jr, Ferrara N, Steinmetz H, Hoeffel J, Cleland JL, Daugherty A, Bruggen N van, Redmond HP, Carano RA, Filvaroff EH (2002) Vascular endothelial growth factor stimulates bone repair by promoting angiogenesis and bone turnover. Proc Natl Acad Sci USA 99:9656–9661
Sun L, Vitolo M, Passaniti A (2001) Runt-related gene 2 in endothelial cells: inducible expression and specific regulation of cell migration and invasion. Cancer Res 61:4994–5001
Takamine Y, Tsuchiya H, Kitakoji T, Kurita K, Ono Y, Ohshima Y, Kitoh H, Ishiguro N, Iwata H (2002) Distraction osteogenesis enhanced by osteoblast-like cells and collagen gel. Clin Orthop Rel Res 399:240–246
Tsuji K, Ito Y, Noda M (1998) Expression of the PEBP2alphaA/AML3/CBFA1 gene is regulated by BMP4/7 heterodimer and its overexpression suppresses type I collagen and osteocalcin gene expression in osteoblastic and nonosteoblastic mesenchymal cells. Bone 22:87–92
Tuncay OC, Ho D, Barker MK (1994) Oxygen tension regulates osteoblast function. Am J Orthod Dentofacial Orthop 105:457–463
Vartanian KB, Chen HY, Kennedy J, Beck SK, Ryaby JT, Wang H, Hoying JB (2005) The non-proteolytically active thrombin peptide TP508 stimulates angiogenic sprouting. J Cell Physiol 206:175–180
Villars F, Bordenave L, Bareille R, Amedee J (2000) Effect of human endothelial cells on human bone marrow stromal cell phenotype: role of VEGF? J Cell Biochem 79:672–685
Vries TJ de, Fourkour A, Punt CJ, Locht LT van de, Wobbes T, Bosch S van den, Rooij MJ de, Mensink EJ, Ruiter DJ, Muijen GN van (1999) Reproducibility of detection of tyrosinase and MART-1 transcripts in the peripheral blood of melanoma patients: a quality control study using real-time quantitative RT-PCR. Br J Cancer 80:883–891
Wang H, Li X, Tomin E, Doty SB, Lane JM, Carney DH, Ryaby JT (2005) Thrombin peptide (TP508) promotes fracture repair by up-regulating inflammatory mediators, early growth factors, and increasing angiogenesis. J Orthop Res 23:671–679
Zerbo IR, Bronckers AL, Lange G de, Burger EH (2005) Localisation of osteogenic and osteoclastic cells in porous beta-tricalcium phosphate particles used for human maxillary sinus floor elevation. Biomaterials 26:1445–1451
Acknowledgements
The authors thank Dr. James T. Ryaby (OrthoLogic, Tempe, Ariz., USA) for providing TP508 for this study and Dr. K. Sasaguri (Kanagawa Dental College, Yokosuka City, Japan) for providing monoclonal antibodies to Runx2.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License ( https://creativecommons.org/licenses/by-nc/2.0 ), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Amir, L.R., Li, G., Schoenmaker, T. et al. Effect of thrombin peptide 508 (TP508) on bone healing during distraction osteogenesis in rabbit tibia. Cell Tissue Res 330, 35–44 (2007). https://doi.org/10.1007/s00441-007-0448-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-007-0448-9