Abstract
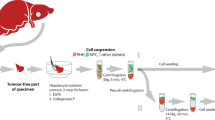
The use of porcine hepatocytes in xenotransplantation, bioartificial liver support or pharmacological approaches demands serum-free cryopreservation protocols yielding high quality, viable, functional hepatocytes. Here, primary porcine hepatocytes were frozen without serum in liquid nitrogen by the use of a computer-assisted freezing device. After thawing, more than 90% of the initial hepatocytes were lost, in part because of damage to genomic DNA. When cryoprotectants were used, the loss was lowered to 70% of the initial cell number; 90% of the remaining cells excluded trypan blue indicating a high degree of viability. Cells were seeded serum-free onto collagen-coated plastic dishes to determine proliferation and retainment of specific functions representing prominent features of hepatocytes in vivo. Whereas no cells adhered to the substratum effectively in conventional culture medium, the addition of conditioned medium derived from hepatic non-parenchymal cells improved attachment. Cells proliferated, retained hepatocyte-specific functions, such as urea production and cytochrome P450 activity, and expressed liver-specific genes to levels observed in non-cryopreserved hepatocytes. Thus, serum-free cryopreserved primary porcine hepatocytes may serve as a valid source of cells for downstream applications. The cells seem to function adequately when an appropriate environment is chosen for recovery after cryopreservation, an ultimate demand for the clinical application of human hepatocytes.








Similar content being viewed by others
References
Alexandre E, Viollon-Abadie C, David P, Gandillet A, Coassolo P, Heyd B, Mantion G, Wolf P, Bachellier P, Jaeck D, Richert L (2002) Cryopreservation of adult human hepatocytes obtained from resected liver biopsies. Cryobiology 44:103–113
Arikura J, Kobayashi N, Okitsu T, Noguchi H, Totsugawa T, Watanabe T, Matsumura T, Maruyama M, Kosaka Y, Tanaka N, Onodera K, Kasai S (2002) UW solution: a promising tool for cryopreservation of primarily isolated rat hepatocytes. J Hepatobiliary Pancreat Surg 9:742–749
Berry MN, Friend DS (1969) High-yield preparation of isolated rat liver parenchymal cells: a biochemical and fine structural study. J Cell Biol 43:506–520
Berthou L, Langouet S, Grude P, Denefle P, Branellec D, Guillouzo A (1998) Negative regulation of Apo A-I gene expression by retinoic acid in rat hepatocytes maintained in a coculture system. Biochim Biophys Acta 1391:329–336
Bilir BM, Guinette D, Karrer F, Kumpe DA, Krysl J, Stephens J, McGavran L, Ostrowska A, Durham J (2000) Hepatocyte transplantation in acute liver failure. Liver Transpl 6:32–40
Canaple L, Nurdin N, Angelova N, Hunkeler D, Desvergne B (2001) Development of a coculture model of encapsulated cells. Ann N Y Acad Sci 944:350–361
Chen Z, Ding Y, Zhang H (2001) Cryopreservation of suckling pig hepatocytes. Ann Clin Lab Sci 31:391–398
Chesne C, Guillouzo A (1988) Cryopreservation of isolated rat hepatocytes: a critical evaluation of freezing and thawing conditions. Cryobiology 25:323–330
Chesne C, Guyomard C, Fautrel A, Poullain MG, Fremond B, De Jong H, Guillouzo A (1993) Viability and function in primary culture of adult hepatocytes from various animal species and human beings after cryopreservation. Hepatology 18:406–414
Choucroun P, Gillet D, Dorange G, Sawicki B, Dewitte JD (2001) Comet assay and early apoptosis. Mutat Res 478:89–96
Chowdhury JR, Chowdhury NR, Strom SC, Kaufman SS, Horslen S, Fox IJ (1998) Human hepatocyte transplantation: gene therapy and more? Pediatrics 102:647–648
Crocker CL (1967) Rapid determination of urea nitrogen in serum or plasma without deproteinization. Am J Med Technol 33:361–365
Dandri M, Burda MR, Gocht A, Torok E, Pollok JM, Rogler CE, Will H, Petersen J (2001) Woodchuck hepatocytes remain permissive for hepadnavirus infection and mouse liver repopulation after cryopreservation. Hepatology 34:824–833
Dunn TB, Kumins NH, Raofi V, Holman DM, Mihalov M, Blanchard J, Law WR, Rastellini C, Benedetti E (2000) Multiple intrasplenic hepatocyte transplantations in the Dalmatian dog. Surgery 127:193–199
Fox IJ, Chowdhury JR, Kaufman SS, Goertzen TC, Chowdhury NR, Warkentin PI, Dorko K, Sauter BV, Strom SC (1998) Treatment of the Crigler–Najjar syndrome type I with hepatocyte transplantation. N Engl J Med 338:1422–1426
Fu T, Guo D, Huang X, O’Gorman MR, Huang L, Crawford SE, Soriano HE (2001) Apoptosis occurs in isolated and banked primary mouse hepatocytes. Cell Transplant 10:59–66
Gebhardt R, Hengstler JG, Muller D, Glockner R, Buenning P, Laube B, Schmelzer E, Ullrich M, Utesch D, Hewitt N, Ringel M, Hilz BR, Bader A, Langsch A, Koose T, Burger HJ, Maas J, Oesch F (2003) New hepatocyte in vitro systems for drug metabolism: metabolic capacity and recommendations for application in basic research and drug development, standard operation procedures. Drug Metab Rev 35:145–213
Guguen-Guillouzo C, Clement B, Baffet G, Beaumont C, Morel-Chany E, Glaise D, Guillouzo A (1983) Maintenance and reversibility of active albumin secretion by adult rat hepatocytes co-cultured with another liver epithelial cell type. Exp Cell Res 143:47–54
Guyomard C, Rialland L, Fremond B, Chesne C, Guillouzo A (1996) Influence of alginate gel entrapment and cryopreservation on survival and xenobiotic metabolism capacity of rat hepatocytes. Toxicol Appl Pharmacol 141:349–356
Hengstler JG, Ringel M, Biefang K, Hammel S, Milbert U, Gerl M, Klebach M, Diener B, Platt KL, Bottger T, Steinberg P, Oesch F (2000a) Cultures with cryopreserved hepatocytes: applicability for studies of enzyme induction. Chem Biol Interact 125:51–73
Hengstler JG, Utesch D, Steinberg P, Platt KL, Diener B, Ringel M, Swales N, Fischer T, Biefang K, Gerl M, Bottger T, Oesch F (2000b) Cryopreserved primary hepatocytes as a constantly available in vitro model for the evaluation of human and animal drug metabolism and enzyme induction. Drug Metab Rev 32:81–118
Hewitt NJ, Buhring KU, Dasenbrock J, Haunschild J, Ladstetter B, Utesch D (2001) Studies comparing in vivo: in vitro metabolism of three pharmaceutical compounds in rat, dog, monkey, and human using cryopreserved hepatocytes, microsomes, and collagen gel immobilized hepatocyte cultures. Drug Metab Dispos 29:1042–1050
Houle R, Raoul J, Levesque JF, Pang KS, Nicoll-Griffith DA, Silva JM (2003) Retention of transporter activities in cryopreserved, isolated rat hepatocytes. Drug Metab Dispos 31:447–451
Ikeda S, Mitaka T, Harada K, Sugimoto S, Hirata K, Mochizuki Y (2002) Proliferation of rat small hepatocytes after long-term cryopreservation. J Hepatol 37:7–14
Jamal HZ, Weglarz TC, Sandgren EP (2000) Cryopreserved mouse hepatocytes retain regenerative capacity in vivo. Gastroenterology 118:390–394
Koebe HG, Dahnhardt C, Muller-Hocker J, Wagner H, Schildberg FW (1996) Cryopreservation of porcine hepatocyte cultures. Cryobiology 33:127–141
Kunieda T, Maruyama M, Okitsu T, Shibata N, Takesue M, Totsugawa T, Kosaka Y, Arata T, Kobayashi K, Ikeda H, Oshita M, Nakaji S, Ohmoto K, Yamamoto S, Kurabayashi Y, Kodama M, Tanaka N, Kobayashi N (2003) Cryopreservation of primarily isolated porcine hepatocytes with UW solution. Cell Transplant 12:607–616
Kuri-Harcuch W, Mendoza-Figueroa T (1989) Cultivation of adult rat hepatocytes on 3T3 cells: expression of various liver differentiated functions. Differentiation 41:148–157
Lubet RA, Nims RW, Mayer RT, Cameron JW, Schechtman LM (1985) Measurement of cytochrome P-450 dependent dealkylation of alkoxyphenoxazones in hepatic S9s and hepatocyte homogenates: effects of dicumarol. Mutat Res 142:127–131
Maganto P, Cienfuegos JA, Santamaria L, Rodriguez V, Eroles G, Andres S, Castillo-Olivares JL, Municio AM (1990) Auxiliary liver by transplanted frozen-thawed hepatocytes. J Surg Res 48:24–32
Malhi H, Gupta S (2001) Hepatocyte transplantation: new horizons and challenges. J Hepatobiliary Pancreat Surg 8:40–50
McKelvey-Martin VJ, Green MH, Schmezer P, Pool-Zobel BL, De Meo MP, Collins A (1993) The single cell gel electrophoresis assay (comet assay): a European review. Mutat Res 288:47–63
Muller P, Stock T, Bauer S, Wolff I (2002) Genotoxicological characterisation of complex mixtures. Genotoxic effects of a complex mixture of perhalogenated hydrocarbons. Mutat Res 515:99–109
Ostling O, Johanson KJ (1984) Microelectrophoretic study of radiation-induced DNA damages in individual mammalian cells. Biochem Biophys Res Commun 123:291–298
Puschel GP, Jungermann K (1994) Integration of function in the hepatic acinus: intercellular communication in neural and humoral control of liver metabolism. Prog Liver Dis 12:19–46
Rialland L, Guyomard C, Scotte M, Chesne C, Guillouzo A (2000) Viability and drug metabolism capacity of alginate-entrapped hepatocytes after cryopreservation. Cell Biol Toxicol 16:105–116
Ries K, Krause P, Solsbacher M, Schwartz P, Unthan-Fechner K, Christ B, Markus PM, Probst I (2000) Elevated expression of hormone-regulated rat hepatocyte functions in a new serum-free hepatocyte-stromal cell coculture model. In Vitro Cell Dev Biol Anim 36:502–512
Runge DM, Runge D, Foth H, Strom SC, Michalopoulos GK (1999) STAT 1alpha/1beta, STAT 3 and STAT 5: expression and association with c-MET and EGF-receptor in long-term cultures of human hepatocytes. Biochem Biophys Res Commun 265:376–381
Sarkis R, Honiger J, Chafai N, Baudrimont M, Sarkis K, Delelo R, Becquemont L, Benoist S, Balladur P, Capeau J, Nordlinger B (2001) Semiautomatic macroencapsulation of fresh or cryopreserved porcine hepatocytes maintain their ability for treatment of acute liver failure. Cell Transplant 10:601–607
Sauer IM, Kardassis D, Zeillinger K, Pascher A, Gruenwald A, Pless G, Irgang M, Kraemer M, Puhl G, Frank J, Muller AR, Steinmuller T, Denner J, Neuhaus P, Gerlach JC (2003) Clinical extracorporeal hybrid liver support—phase I study with primary porcine liver cells. Xenotransplantation 10:460–469
Seglen PO (1976) Preparation of isolated rat liver cells. Methods Cell Biol 13:29–83
Sheil AG (2002) Xenogeneic bioartificial liver support: where are we now? Transplant Proc 34:2493–2495
Silva JM, Day SH, Nicoll-Griffith DA (1999) Induction of cytochrome-P450 in cryopreserved rat and human hepatocytes. Chem Biol Interact 121:49–63
Singh NP, McCoy MT, Tice RR, Schneider EL (1988) A simple technique for quantitation of low levels of DNA damage in individual cells. Exp Cell Res 175:184–191
Sousa G de, Nicolas F, Placidi M, Rahmani R, Benicourt M, Vannier B, Lorenzon G, Mertens K, Coecke S, Callaerts A, Rogiers V, Khan S, Roberts P, Skett P, Fautrel A, Chesne C, Guillouzo A (1999) A multi-laboratory evaluation of cryopreserved monkey hepatocyte functions for use in pharmaco-toxicology. Chem Biol Interact 121:77–97
Southard JH (2002) Hypothermic and cryo-preservation of hepatocytes. Single Topic Conference, Warrenton, Va., USA
Steinberg P, Fischer T, Kiulies S, Biefang K, Platt KL, Oesch F, Bottger T, Bulitta C, Kempf P, Hengstler J (1999) Drug metabolizing capacity of cryopreserved human, rat, and mouse liver parenchymal cells in suspension. Drug Metab Dispos 27:1415–1422
Strom SC, Chowdhury JR, Fox IJ (1999) Hepatocyte transplantation for the treatment of human disease. Semin Liver Dis 19:39–48
Sundback CA, Vacanti JP (2000) Alternatives to liver transplantation: from hepatocyte transplantation to tissue-engineered organs. Gastroenterology 118:438–442
Swales NJ, Luong C, Caldwell J (1996) Cryopreservation of rat and mouse hepatocytes. I. Comparative viability studies. Drug Metab Dispos 24:1218–1223
Utesch D, Oesch F (1992) Dependency of the in vitro stabilization of differentiated functions in liver parenchymal cells on the type of cell line used for co-culture. In Vitro Cell Dev Biol 28A:193–198
Vian L, Yusuf A, Guyomard C, Cano JP (2002) The liverbeads as a tool for the comet assay. Mutat Res 519:163–170
Vreugdenhil PK, Marsh DC, Southard JH (1996) Comparison of isolated hepatocytes and tissue slices for study of liver hypothermic preservation/reperfusion injury. Cryobiology 33:430–435
Watanabe FD, Arnaout WS, Ting P, Navarro A, Khalili T, Kamohara Y, Kahaku E, Rozga J, Demetriou AA (1999) Artificial liver. Transplant Proc 31:371–373
Yagi K, Yamada C, Serada M, Sumiyoshi N, Michibayashi N, Miura Y, Mizoguchi T (1995) Reciprocal regulation of prothrombin secretion and tyrosine aminotransferase induction in hepatocytes. Eur J Biochem 227:753–756
Yagi T, Hardin JA, Valenzuela YM, Miyoshi H, Gores GJ, Nyberg SL (2001) Caspase inhibition reduces apoptotic death of cryopreserved porcine hepatocytes. Hepatology 33:1432–1440
Acknowledgements
The excellent technical assistance of Sabine Ebensing and Madlen Hempel is greatly appreciated.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Müller, P., Aurich, H., Wenkel, R. et al. Serum-free cryopreservation of porcine hepatocytes. Cell Tissue Res 317, 45–56 (2004). https://doi.org/10.1007/s00441-004-0894-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-004-0894-6