Abstract
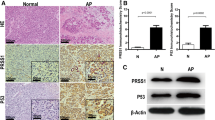
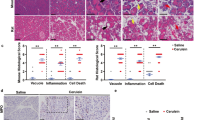
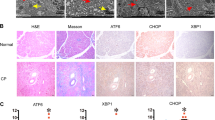
Secretory stress proteins (SSP) are a family of proteins including isoforms of pancreatitis-associated protein (PAP) and pancreatic stone protein (PSP/reg). In vitro exposure to trypsin results in the formation of insoluble fibrillar structures. SSP are constitutively secreted into pancreatic juice at low levels. The WBN/Kob rat is a model for chronic pancreatitis, displaying focal inflammation, destruction of the parenchyma and changes in the architecture of the acinar cell; the synthesis and secretion of SSP are also increased. We have investigated the secretory apparatus by SSP immunohistochemistry at the light- and electron-microscopical (EM) levels. Immunocytochemistry of PSP/reg in Wistar control rats reveals low levels, with individual acinar cells exhibiting high immunoreactivity in zymogen granules. PAP is not detectable. In the WBN/Kob rat, PSP/reg and PAP immunoreactivity is markedly increased. Double immunofluorescence for PSP/reg and PAP I or II demonstrates that these proteins colocalize to the same cell. Acinar cells change their secretory architecture by fusion of zymogen granules and elongation of the fused organelles. The immunogold technique has demonstrated an increase of SSP in zymogen granules in WBN/Kob rats. PSP/reg-positive zymogen granules fuse to form elongated structures with fibrillar contents. An extensive PSP/reg-positive fibrillar network is established in the cytosol. Extracellular fibrils have been observed in several ductules. Thus, SSP-derived fibrils form concomitantly with acinar damage in the WBN/Kob rat. Based on the known tryptic cleavage site of SSP, the in vivo generation of fibrils is presumably the result of premature trypsin activation.




Similar content being viewed by others
References
Ashizawa N, Endoh H, Hidaka K, Watanabe M, Fukumoto S (1995) Scanning electron microscopic observation of pancreatic ducts in WBN/Kob rats. Pancreas 11:389–395
Bimmler D, Frick TW, Scheele GA (1995) High-level secretion of native rat pancreatic lithostathine in a baculovirus expression system. Pancreas 11:63–76
Bimmler D, Angst E, Valeri V, Bain M, Scheele GA, Frick TW, Graf R (1999a) Regulation of PSP/reg in rat pancreas: immediate and steady state adaptation to different diets. Pancreas 19:255–267
Bimmler D, Schiesser M, Angst E, Bain M, Frick TW, Graf R (1999b) In a rat model of chronic pancreatitis (WBN/Kob), levels of PSP/reg in pancreatic juice are highly increased in the early phase of the disease. Pancreas 19:416
Bockman DE (1997) Morphology of the exocrine pancreas related to pancreatitis. Microsc Res Tech 37:509–519
Cerini C, Peyrot V, Garnier C, Duplan L, Veesler S, Le-Caer JP, Bernard JP, Bouteille H, Michel R, Vazi A, Dupuy P, Michel B, Berland Y, Verdier JM (1999) Biophysical characterization of lithostathine. Evidences for a polymeric structure at physiological pH and a proteolysis mechanism leading to the formation of fibrils. J Biol Chem 274:22266–22274
Crawford HC, Scoggins CR, Washington MK, Matrisian LM, Leach SD (2002) Matrix metalloproteinase-7 is expressed by pancreatic cancer precursors and regulates acinar-to-ductal metaplasia in exocrine pancreas. J Clin Invest 109:1437–1444
Graf R, Schiesser M, Scheele GA, Marquardt K, Frick TW, Ammann R, Bimmler D (2001) A family of 16 kDa pancreatic secretory stress proteins form highly organized fibrillar structures upon tryptic activation. J Biol Chem 276:21028–21038
Graf R, Schiesser M, Lüssi A, Went P, Scheele GA, Bimmler D (2002) Coordinate regulation of secretory stress proteins (PSP/reg and PAP I, II and III) in the rat exocrine pancreas during experimental acute pancreatitis. J Surg Res 105:136–144
Hashimoto T, Yamada T, Yokoi T, Sano H, Ando H, Nakazawa T, Ohara H, Nomura T, Joh T, Itoh M (2000) Apoptosis of acinar cells is involved in chronic pancreatitis in WBN/Kob rats: role of glucocorticoids. Pancreas 21:296–304
Lerch MM, Gorelick FS (2000) Early trypsinogen activation in acute pancreatitis. Med Clin North Am 84:549–563
Luthen R, Niederau C, Grendell JH (1995) Intrapancreatic zymogen activation and levels of ATP and glutathione during caerulein pancreatitis in rats. Am J Physiol 66:66–66
Maxwell MH (1978) Two rapid and simple methods used for the removal of resins from 1.0 micron thick epoxy sections. J Microsc 112:253–255
Morisset J, Iovanna J, Grondin G (1997) Localization of rat pancreatitis-associated protein during bile salt-induced pancreatitis. Gastroenterology 112:543-550
Ohashi K, Kim JH, Hara H, Aso R, Akimoto T, Nakama K (1990) WBN/Kob rats. A new spontaneously occurring model of chronic pancreatitis. Int J Pancreatol 6:231–247
Rouquier S, Verdier JM, Iovanna J, Dagorn JC, Giorgi D (1991) Rat pancreatic stone protein messenger RNA. Abundant expression in mature exocrine cells, regulation by food content, and sequence identity with the endocrine reg transcript. J Biol Chem 266:786–791
Schiesser M, Bimmler D, Frick TW, Graf R (2001) Conformational changes of pancreatitis-associated protein (PAP) activated by trypsin lead to insoluble protein aggregates. Pancreas 22:186–192
Unno M, Yonekura H, Nakagawara K, Watanabe T, Miyashita H, Moriizumi S, Okamoto H, Itoh T, Teraoka H (1993) Structure, chromosomal localization, and expression of mouse reg genes, reg I and reg II. A novel type of reg gene, reg II, exists in the mouse genome. J Biol Chem 268:15974–15982
Wagner M, Greten FR, Weber CK, Koschnick S, Mattfeldt T, Deppert W, Kern H, Adler G, Schmid RM (2001) A murine tumor progression model for pancreatic cancer recapitulating the genetic alterations of the human disease. Genes Dev 15:286–293
Whitcomb DC, Gorry MC, Preston RA, Furey W, Sossenheimer MJ, Ulrich CD, Martin SP, Gates LK Jr, Amann ST, Toskes PP, Liddle R, McGrath K, Uomo G, Post JC, Ehrlich GD (1996) Hereditary pancreatitis is caused by a mutation in the cationic trypsinogen gene. Nat Genet 14:141–145
Acknowledgements
We are grateful for the technical support and advice of the EM team headed by Prof. Thomas Bächi at the EM-Zentrum, Universität Zürich. The excellent technical help of R. Gassmann and M. Bain is highly appreciated. This report is a partial fulfillment of the requirements for the doctoral thesis of S.M.
Author information
Authors and Affiliations
Corresponding author
Additional information
Financial support by the Swiss National Science Foundation (3200–066728.01) and the Amelie Waring Stiftung is greatly appreciated
Rights and permissions
About this article
Cite this article
Meili, S., Graf, R., Perren, A. et al. Secretory apparatus assessed by analysis of pancreatic secretory stress protein expression in a rat model of chronic pancreatitis. Cell Tissue Res 312, 291–299 (2003). https://doi.org/10.1007/s00441-003-0733-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-003-0733-1