Abstract
Rare disruptions of FOXP2 have been strongly implicated in deficits in language development. Research over the past decade has suggested a role in the formation of underlying neural circuits required for speech. Until recently no evidence existed to suggest that the closely related FOXP1 gene played a role in neurodevelopmental processes. However, in the last few years, novel rare disruptions in FOXP1 have been reported in multiple cases of cognitive dysfunction, including intellectual disability and autism spectrum disorder, together with language impairment. As FOXP1 and FOXP2 form heterodimers for transcriptional regulation, one may assume that they co-operate in common neurodevelopmental pathways through the co-regulation of common targets. Here we compare the phenotypic consequences of FOXP1 and FOXP2 impairment, drawing on well-known studies from the past as well as recent exciting findings and consider what these tell us regarding the functions of these two genes in neural development.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Forkhead box (FOX) proteins are an evolutionarily ancient family of transcription factors characterised by a highly conserved forkhead DNA-binding domain. Despite the similarity in the DNA-binding domain, FOX proteins have a wide range of important biological functions (Hannenhalli and Kaestner 2009). Most FOX proteins bind to their target DNA sequences as monomers, except members of the FOXP subfamily, which includes FOXP1–4. FOXP proteins are somewhat atypical as they also possess a zinc finger and leucine zipper domain, both of which mediate interactions with other proteins, thus allowing FOXP proteins to form homo- and heterodimers to facilitate their binding to DNA for transcriptional regulation (Li et al. 2004). In the past, members of the FOXP subfamily have been implicated in various human diseases (Bennett and Ochs 2001; Jin et al. 2010; Lai et al. 2001), but only FOXP1 and FOXP2 have been linked to cognitive disorders so far.
Perhaps the most prominent of the FOXP genes is FOXP2 (OMIM 605317), whose involvement in speech and language acquisition has now been studied for more than 10 years (Fisher and Scharff 2009). FOXP2 and language development has generated extreme interest as the acquisition of spoken language is central to what makes us human. In the last 2 years, evidence has emerged that implicates FOXP1 (OMIM 605515), the closest relative of FOXP2 (64 % total protein sequence identity, 89 % in the forkhead domain), in the pathology of human cognitive disorders, which involve language impairment. FOXP1 and FOXP2 are already known to co-operate in the regulation of non-neural developmental processes (Shu et al. 2007) and it will be interesting to validate whether this co-operation extends to brain development. A comparison of the clinical phenotypes caused by FOXP1 and FOXP2 disruption would provide important steps towards uncovering those regions of neuropathology in which these genes play a role.
FOXP2 mutations are implicated in developmental verbal dyspraxia
Foxp2 contributes to lung, heart and oesophagus development (Shu et al. 2001, 2007), but the most well-known role of this gene is in the development of speech and language (Fisher and Scharff 2009; Newbury and Monaco 2010). Human FOXP2 was first linked to language development in 2001, when a heterozygous missense mutation in the forkhead domain (R553H) was found to be causative for an inherited language disorder in a large 3-generation pedigree known as the KE family (Lai et al. 2001). The KE family has been extensively studied and the phenotype is now well defined, affecting expressive, receptive and written language (Table 1). A core feature of the disorder is a difficulty in the learning and production of co-ordinated sequences of orofacial movements, which impairs the production of fluent speech (Vargha-Khadem et al. 1995; Watkins et al. 2002a). Therefore the affected members have a primary diagnosis of developmental verbal dyspraxia (DVD), which is also known as childhood apraxia of speech (CAS). Studies on the KE family have also included formal evaluations of non-verbal intelligence and the average IQ of affected KE family members is lower than that of unaffected individuals (Fisher et al. 1998; Vargha-Khadem et al. 1995; Watkins et al. 2002a), raising the possibility that the missense mutation in the FOXP2 gene also impacts other cognitive functions. Nevertheless, the most prominent and consistent features of the disorder are in the domain of speech and language, with more severe and wide-ranging effects on verbal skills than on non-verbal cognition (Watkins et al. 2002a).
The presence of just one de novo mutation within the coding region of FOXP2 did not, by itself, provide compelling evidence that FOXP2 contributes to the pathology of DVD. To assess the contribution of heterozygous FOXP2 mutations in patient samples independent from the KE family, FOXP2 was screened in 49 unrelated children diagnosed with DVD (MacDermot et al. 2005). Three different variants altering the FOXP2 protein sequence in three individual patients were identified. The most interesting of these variants was a heterozygous nonsense mutation (not found in 252 controls) which yielded a stop codon at position 328 of the FOXP2 protein, resulting in either a truncated protein missing the functionally important forkhead, leucine zipper and zinc finger domains or a complete loss of FOXP2 due to nonsense mediated RNA decay. The child had a diagnosis of DVD and displayed articulation problems along with impaired receptive and expressive language development. The same mutation was also detected in an affected sibling as well as the mother, who was reported to have a history of speech difficulties. This finding offered further evidence that FOXP2 is of pathological importance in DVD in the absence of other cognitive disorders, which were exclusion criteria in this study.
Various large-scale 7q31 deletions that include the FOXP2 gene, ranging from 1.57 Mb to 15 Mb in size, have also been reported in patients with DVD (Feuk et al. 2006; Lennon et al. 2007; Palka et al. 2012; Rice et al. 2011; Zeesman et al. 2006; Zilina et al. 2011). DVD was reported in every case and most of the reports concluded that impaired language was due to the loss of FOXP2. However, the large size of these deletions encompassing several to many genes is a complication for understanding the links between phenotypes and FOXP2 dysfunction. Translocation breakpoints directly disrupting the FOXP2 locus in patients diagnosed with DVD provided more clear-cut support for the relevance of this gene to speech and language pathology (Table 1) (Feuk et al. 2006; Lai et al. 2000, 2001; Shriberg et al. 2006; Tomblin et al. 2009).
The reported FOXP2 variants are rare but have not been found in normal individuals from the 1,000 genome project and taken together, three translocation breakpoints disrupting FOXP2 (Feuk et al. 2006; Lai et al. 2000; Shriberg et al. 2006), one missense mutation (Lai et al. 2001) and one nonsense mutation (MacDermot et al. 2005) independently associated with a DVD phenotype provide strong genetic evidence for the role of FOXP2 in the clinical phenotype (summarised in Table 1).
Evidence for a role of FOXP1 in neurodevelopmental disorders
Foxp1 has been associated with a wide range of functions including development of the lung, heart, oesophagus, immune system and spinal motor neurons, as well as cancer (Banham et al. 2001; Dasen et al. 2008; Hu et al. 2006; Jepsen et al. 2008; Palmesino et al. 2010; Rousso et al. 2008; Shi et al. 2004; Shu et al. 2001, 2007; Wang et al. 2004). It has been known for some time that Foxp1 is expressed in particular neuronal subpopulations in the developing brain (Ferland et al. 2003), but its precise roles in brain development have not been defined. Given that the expression of Foxp1 and Foxp2 overlaps in certain areas of the developing brain (Ferland et al. 2003; Teramitsu et al. 2004) and that Foxp1 and Foxp2 are able to form heterodimers for transcriptional repression (Li et al. 2004), it is reasonable to hypothesize that FOXP1 could also be involved in the pathology of DVD. However, in a screen of the entire coding region of FOXP1 in 49 DVD patients, (the identical cohort used in the FOXP2 screening described earlier), no potentially disease-causing variants were found (Vernes et al. 2009). Although the patient cohort was small, it suggested that, unlike FOXP2, mutations in FOXP1 are not sufficient to cause DVD in isolation. Instead, various screening studies carried out within the last few years have revealed that FOXP1 may be of more global importance in a range of neurodevelopmental disorders, which includes but is not restricted to speech and language disorders.
The first hint that FOXP1 may be involved in neurodevelopmental disorders was provided by a heterozygous deletion in 3p14.1, which affected FOXP1, EIF4E3, PROK2 and GPR27, in a patient with speech delay, hypertonia and additional phenotypes (for details see Table 2) (Pariani et al. 2009). The authors optimistically attributed many of the patient’s symptoms including the speech delay to the disruption of FOXP1, but the contribution of PROK2 and GPR27 to the phenotype cannot be ruled out, especially considering that both genes were previously implicated in developmental retardation (Petek et al. 2003). More convincing evidence was provided when a deletion exclusively affecting the FOXP1 gene was found in a male child with impaired language acquisition and motor development delay (Carr et al. 2010). Unfortunately, a confounding factor in this study was the presence of a Chiari I malformation (cerebellar tonsil abnormality) in the patient, which may have contributed to the delay in motor and speech development, therefore convincing evidence for a role of FOXP1 in cognitive disorders was still lacking.
Shortly afterwards, a large-scale screen for copy number variations (CNVs) in 1,523 patients with intellectual disability, uncovered three de novo heterozygous deletions solely affecting FOXP1 (Horn et al. 2010). A large 1.3 Mb deletion affecting FOXP1 and other genes was also found in a control individual (Horn et al. 2010). The three patients had moderate intellectual disability (IQ of <50), gross motor delay and a severe speech and language defect, characterised by a delay in the onset of speech, dysgrammatism and very poor speech articulation. Two of the reported patients also had an oromotor defect, including difficulties with lip protrusion and tongue elevation, but DVD was not diagnosed. These findings provided the first compelling evidence that disruptions in the FOXP1 gene can be causative for multiple neurodevelopmental abnormalities, which include language impairment, implicating FOXP1 in more widespread cognitive processes than were previously described for FOXP2. This hypothesis gained further support from a recent independent study of 110 individuals with intellectual disability and/or autism spectrum disorder (ASD), which identified a de novo intragenic FOXP1 deletion in a patient with both intellectual disability (IQ of 58) and severe language impairment, particularly affecting expressive language (Hamdan et al. 2010). The deletion included sequences corresponding to the leucine zipper and zinc finger domains, which are important for FOXP1 dimerization and transcriptional repression. In addition to the deletion, a de novo nonsense mutation, R525X, was found in the FOXP1 forkhead domain in another patient with non-syndromic intellectual disability (IQ of 48), severe language impairment and ASD. This study added further weight to previous findings that FOXP1 is important for language development and normal intelligence and the discovery of a FOXP1 stop mutation in a patient with ASD was exciting, as it highlighted FOXP1 for the first time as a potential ASD candidate gene.
Whole exome sequencing has the potential to identify all coding variants in an affected individual and was recently used to define rare de novo coding mutations in 20 individuals with ASD and their healthy parents, along with 20 unrelated ethnically matched controls (O’Roak et al. 2011). Among several presumably causative mutations in different genes was a single base insertion in FOXP1, introducing a frameshift and premature stop codon (A339SfsX4). This individual had severe ASD and delayed language development together with intellectual disability (IQ of 34), reminiscent of previously reported FOXP1 phenotypes (Carr et al. 2010; Hamdan et al. 2010; Horn et al. 2010). An additional inherited missense variant, H275R, was also detected in the CNTNAP2 gene. As CNTNAP2 has been implicated in intellectual disability (Gregor et al. 2011; Zweier et al. 2009), ASD (Alarcon et al. 2008) and in language impairment without ASD (Vernes et al. 2008; Whitehouse et al. 2012), it could also have contributed to the patient’s phenotype. As FOXP1 was found to regulate CNTNAP2 expression, the FOXP1 mutant identified in this patient could enhance any potentially damaging effects of the CNTNAP2 H275R variant, highlighting the presence of putative modifier gene effects (O’Roak et al. 2011).
Very recently, direct sequencing of balanced chromosomal breakpoints in 38 patients with ASD uncovered a translocation breakpoint affecting FOXP1 on chromosome 3p and ANK3 on chromosome 10q in an individual with ASD and speech delay (Talkowski et al. 2012). While ANK3 has previously been implicated in bipolar disorder and schizophrenia (Ferreira et al. 2008; Williams et al. 2011), secondary CNV analysis of FOXP1 and ANK3 in 19,566 patients with ASD revealed a significant CNV burden for FOXP1, but not ANK3 (Talkowski et al. 2012). The distinct but complementary approaches of chromosomal breakpoint sequencing followed by an extensive secondary assessment of CNV findings provided further compelling support that FOXP1 represents a gene with a widespread role in neurodevelopmental processes.
Taken together, five FOXP1 single gene deletions (Carr et al. 2010; Hamdan et al. 2010; Horn et al. 2010), two intragenic nonsense and frameshift mutations (Hamdan et al. 2010; O’Roak et al. 2011) and a chromosomal breakpoint disrupting FOXP1 supported by secondary CNV analysis (Talkowski et al. 2012) in patients with intellectual disability, ASD, language disorder and motor development delay, provide strong evidence for FOXP1 underlying various cognitive phenotypes (summarised in Table 2).
Similarities and differences between phenotypes associated with FOXP1 and FOXP2 disruptions
Language disorder
In language disorders, a distinction is often made as to whether the language impairments are primarily receptive (hearing, reading and comprehending) or expressive (speaking and writing), or affect both. People with FOXP2 mutations have both impaired expressive and receptive language (MacDermot et al. 2005; Watkins et al. 2002a), whereas expressive language appears to be more affected in patients with FOXP1 disruptions (Hamdan et al. 2010; Horn et al. 2010). A core feature of the phenotype associated with FOXP2 dysfunction is an abnormal articulation due to verbal dyspraxia, i.e. impairment in the motor programming of orofacial movements required to produce normal speech (MacDermot et al. 2005; Vargha-Khadem et al. 1995). Although articulation problems have been described in some patients with FOXP1 disruption (Horn et al. 2010), orofacial dyspraxia has not been diagnosed so far (Carr et al. 2010; Hamdan et al. 2010; Horn et al. 2010; O’Roak et al. 2011), neither have any FOXP1 variants been identified in the small DVD patient cohort (Vernes et al. 2009). As FOXP1 variants are yet to be associated solely with a language disorder without additional cognitive phenotypes, the possibility exists that the language disorder observed in patients with disruptions in FOXP1 is a consequence of more global cognitive disruption rather than a specific disruption in those neural circuits necessary for speech production. This could be elucidated by comparative brain imaging of patients with FOXP1 and FOXP2 disruptions as well as comparing behavioural, electrophysiological and morphological phenotypes of Foxp1 and Foxp2 knockout mice. Generally, a delay in language acquisition is often observed in ASD individuals along with significantly impaired comprehension, articulation and grammar, which improves with time (Boucher 2011). Similarly, language deficits are often severe in children diagnosed with intellectual disability (Kaufman et al. 2010). Thus, the impaired language observed in patients with FOXP1 disruptions could be a secondary consequence of these disorders. In summary, there is an overlap in the language phenotype of patients with disruptions in FOXP1 and FOXP2, particularly regarding expressive language impairment (Fig. 1), but how similar the mechanistic basis is remains unclear.
Autism spectrum disorder
Autism spectrum disorder is characterised by impaired social interaction and communication, language impairment and the presence of restrictive and repetitive behaviours. In most cases, ASD is inherited and the specific genetic disruptions are known in only a minority of cases (Geschwind 2011). ASD has been diagnosed in patients with various disruptions in the FOXP1 gene (Hamdan et al. 2010; O’Roak et al. 2011; Talkowski et al. 2012), implicating FOXP1 in the pathology of ASD. Chromosome 7q31 is a known autism susceptibility locus, particularly involving the language impairment of the disorder (Alarcon et al. 2002), therefore FOXP2 has been suggested as a potential ASD candidate gene. However, FOXP2 screenings in different cohorts of ASD patients have failed to identify any causative mutations (Gauthier et al. 2003; Newbury et al. 2002; Wassink et al. 2002). Yet, in a recent large-scale haplotype mapping analysis FOXP2 was identified as a novel ASD candidate gene, among 1,218 others (Casey et al. 2011). FOXP2 is also implicated in the etiological pathways of ASD through its target genes MET (Mukamel et al. 2011) and CNTNAP2 (Vernes et al. 2008), although CNTNAP2 has also been linked to language impairment where there is no diagnosis of ASD (Vernes et al. 2008). Additionally, people with isolated mutations of FOXP2 do not show any signs of ASD even though FOXP2 is involved in pathways relevant to autism. Thus, while the possibility is open that screening studies of FOXP2 in larger cohorts of ASD patients could identify causative FOXP2 variants in the future, existing evidence suggests that—in contrast to FOXP1—FOXP2 is not involved in ASD.
Intellectual disability
Intellectual disability is a cognitive disorder characterised by an IQ of <70 (Kaufman et al. 2010). All non-verbal IQs reported in patients with FOXP1 variants are below this threshold (Table 2) indicating that intellectual disability is a consistent feature of the phenotype associated with FOXP1 disruption. Most but not all affected KE family members with FOXP2 missense mutations have a lower IQ than unaffected members (Fisher et al. 1998, 2003; Vargha-Khadem et al. 1995; Watkins et al. 2002a) but normal IQs have been reported in other patients with perturbations in the FOXP2 gene (MacDermot et al. 2005; Tomblin et al. 2009). Intellectual disability therefore cannot be considered a reliable characteristic of the phenotype associated with FOXP2 disruption at this time. The reported IQs of patients with FOXP1 disruptions are much lower than those reported for people with FOXP2 variants (Tables 1, 2); therefore intelligence appears to be influenced more strongly by FOXP1 than FOXP2 dysfunction.
Motor development delay
Although difficulties with fine motor movement and co-ordination are sometimes present in DVD patients with FOXP2 mutations, gross motor and developmental delay appear to be more predominant in patients with FOXP1 disruption (Tables 1, 2). The delayed motor development described in most human patients with disruptions in the FOXP1 gene (Carr et al. 2010; Hamdan et al. 2010; Horn et al. 2010; Pariani et al. 2009) is interesting, as Foxp1 defines the identity of motor neurons in the mouse spinal cord and influences motor neuron migration, axon projection and axonal branching at muscle targets (Dasen et al. 2008; Palmesino et al. 2010; Rousso et al. 2008). Therefore the delayed motor development in individuals with FOXP1 disruptions may be influenced by pathologies in both the central and peripheral nervous system.
Steps towards elucidating neurodevelopmental pathways influenced by FOXP1 and FOXP2
Disruptions of FOXP1 and FOXP2 cause distinct phenotypes with some overlapping features (Fig. 1), pointing to both shared and distinct neurodevelopmental roles for these two genes. Below we discuss potential approaches to elucidate the molecular pathways and circuits involved.
Defining the neural phenotype of patients
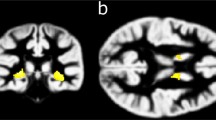
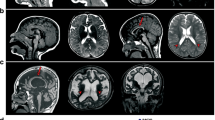
The neural basis of behavioural abnormalities can be identified by structural and functional brain imaging of patients. MRI analyses of some FOXP1 patients have been performed but have revealed no obvious structural brain abnormalities (Carr et al. 2010; Horn et al. 2010). Conventional neuroradiological assessment of MRI scans performed on affected members of the KE family also revealed no obvious abnormalities at first (Vargha-Khadem et al. 2005), but application of voxel-based morphometry, which can detect more subtle differences in grey and white matter and additional volumetric analysis revealed significant differences in specific brain regions of affected KE members (Watkins et al. 2002b). The neuroanatomical effects of the KE mutation have been extensively reviewed elsewhere (Vargha-Khadem et al. 2005), but in brief involve abnormalities in the cortex, cerebellum and basal ganglia, particularly the striatum (Liegeois et al. 2003; Vargha-Khadem et al. 1998; Watkins et al. 2002b). These findings from brain imaging implicate FOXP2 in the development of cortico-striatal circuits, which are involved in sensorimotor integration required for vocal motor learning. This would suggest that the phenotype observed in the KE family stems from disruption in the underlying neural circuits of language development, an idea that has received support from animal studies (see below).
Animal studies
Studies using animal models have taken us a long way towards elucidating the role of Foxp2 in brain development. Several Foxp2 knockout and mutant mouse models have been generated and the different phenotypes have already been extensively reviewed (Fisher and Scharff 2009). In brief, these phenotypes include developmental delay, motor impairment, cerebellar abnormalities and disrupted synaptic plasticity in the striatum. Recently, it was further demonstrated that motor skill learning is significantly impaired in mice carrying the equivalent mutation to that found in affected KE family members, which was explained by aberrant striatal activity during motor skill learning (French et al. 2011).
Two amino acid substitutions in the human FOXP2 protein have emerged during human evolution, possibly linked to the development of speech and language (Enard et al. 2002) and a partially humanised form of the FOXP2 gene has been shown to increase synaptic plasticity in the striatum of mice (Enard et al. 2009). This phenotype is opposite to what has been described in mice with Foxp2 dysfunction (French et al. 2011; Groszer et al. 2008) suggesting that the emergence of human FOXP2 may have enhanced cortico-striatal circuits or circuit properties during the evolution of spoken language. Taken together, these animal studies have revealed that Foxp2 is involved in the development of neural plasticity in circuits contributing to language and a possibly wider cognitive function, which is in agreement with the findings from neuroimaging of KE family members (see above) (Fig. 2).
Foxp1 knockout mouse models have been used to investigate the importance of Foxp1 in a range of non-neural developmental processes (Feng et al. 2010; Hu et al. 2006; Shu et al. 2007; Wang et al. 2004; Wu et al. 2006) and in the development of motor neurons in the spinal cord (Dasen et al. 2008), but have not been used to define the role of Foxp1 in brain development. Homozygous loss of Foxp1 causes embryonic death at E14.5 due to heart failure (Wang et al. 2004). Conditional removal of Foxp1 in the brain will therefore be the key to elucidating the importance of Foxp1 during later stages of brain development. It will be interesting to see whether brain-related aspects of the Foxp1 knockout phenotype are distinct from the Foxp2 knockout phenotype, as is the case in the human situation. Foxp1 and Foxp2 expression overlaps in the striatum, thalamus, superior colliculus and inferior olive in the mature mouse brain, but their expression differs in other regions including the cortex, hippocampus and inferior colliculus (Ferland et al. 2003). It is interesting that histological analyses of brains from Foxp2 knockout mice only revealed gross morphological abnormalities in the cerebellum, where Foxp1 is not co-expressed (French et al. 2007), suggesting that Foxp1 may compensate for the loss of Foxp2 and that a level of redundancy exists between these two genes. It will be interesting to see whether layers 3, 4 and 5 of the cortex or the hippocampus which express Foxp1 only are exclusively abnormal in the Foxp1 knockout mouse.
Identification of target genes
Common FOXP1 and FOXP2 neurodevelopmental pathways seem likely, considering their potential to form heterodimers and their co-expression in certain brain tissues. On the other hand, the distinct phenotypes seen in patients with FOXP1 and FOXP2 disruption would suggest they participate at least to some degree, in independent pathways. The identification and classification of target genes hold the key to determining which pathways and networks involve FOXP1 and FOXP2 during brain development. For FOXP2, genome wide in vivo ChIP-chip screens coupled to expression profiling have already been used to define transcriptional targets in the developing brain; many of these targets were found to be involved in pathways regulating neurite outgrowth, axon guidance and synaptic plasticity (Konopka et al. 2009; Spiteri et al. 2007; Vernes et al. 2007, 2011). Elsewhere, it has been shown that FOXP2 directly regulates CNTNAP2, mutations in which, like FOXP2, were found to be linked to language impairment (Vernes et al. 2008), the autism candidate gene MET, involved in neuronal differentiation (Mukamel et al. 2011) as well as the schizophrenia susceptibility gene Disrupted in schizophrenia 1 (DISC1) in HEK293 cells (76). Whether the identification of DISC1 as a FOXP2 target places FOXP2 in schizophrenia-related pathways remains unclear. There is conflicting evidence for a role of FOXP2 in schizophrenia; significant associations have been reported between certain FOXP2 SNPs and schizophrenia (Sanjuan et al. 2006; Tolosa et al. 2010), but the FOXP2 missense and nonsense mutations associated with DVD (Table 1) were not found in patients with schizophrenia (Sanjuan et al. 2005). In conclusion, the published targets of FOXP2 implicate this transcription factor in pathways regulating neurite outgrowth, axon guidance and synaptic plasticity, which is in agreement with a role of FOXP2 in the development of neural circuits for language development.
The genes regulated by FOXP1 in developing brain tissue have not yet been identified by genome-wide ChIP screens. Overexpression of Foxp1 in a murine striatal cell line has recently revealed a repression of immune-related genes (Tang et al. 2012), which is in agreement with previous studies demonstrating a role of Foxp1 in immune development. Given the level of cognitive disruption caused by FOXP1 variants, it is reasonable to assume that FOXP1 targets will be uncovered in many neurodevelopmental pathways and not limited to immune function. There is evidence that CNTNAP2 expression is regulated by FOXP1 (O’Roak et al. 2011), which has also been shown for FOXP2 (Vernes et al. 2008), providing the first evidence for a common FOXP1–FOXP2 pathway involving CNTNAP2 in neurodevelopmental processes. On the other hand, the fact that CNTNAP2 is implicated in both ASD (Alarcon et al. 2008) and in language impairment without ASD (Vernes et al. 2008) suggests distinct CNTNAP2 pathways exist. Elsewhere, Foxp1 has also been shown to regulate Pitx3 transcription during the differentiation of ES cells into midbrain dopaminergic neurons (Konstantoulas et al. 2010).
Typically, transcription factors do not act alone but rather in complexes together with other transcription factors, chromatin remodellers and cofactor proteins to bind to promoter sequences and regulate target gene expression. FOXP1 and FOXP2 interact with various proteins to regulate gene expression in different tissues (supplementary Table 1) (Chokas et al. 2010; Datta et al. 2008; Jepsen et al. 2008; Li et al. 2004; Otaegi et al. 2011; Ravasi et al. 2010; Takayama et al. 2008; Wu et al. 2006; Zhou et al. 2008) but identification of the combinatorial interactions of FOXP1 and FOXP2 with other proteins specifically in the brain will be needed to understand the roles they play in brain development. The interactions of FOXP1 and FOXP2 with different proteins represent another dimension to the regulation of their target genes.
Concluding remarks
Disease-causing variants in both FOXP1 and FOXP2 are relatively rare, but play a significant role in the pathology of cognitive diseases. While the importance of FOXP2 in the pathology of DVD has been a popular topic of investigation for more than 10 years, it remains to be seen whether perturbations in FOXP2 are confined to language impairment or whether there is also some risk for other diagnostic entities. Genetic evidence emerging in the last 2 years provides compelling evidence that FOXP1 variants are responsible for a more global cognitive phenotype, encompassing language impairment, intellectual disability, ASD and motor development delay. The phenotypic spectra of FOXP1 and FOXP2 disruptions suggest that these two closely related transcription factors are involved in both shared and distinct neurodevelopmental pathways underlying cognitive diseases through the regulation of common and exclusive targets. The findings discussed in this review show that FOXP1 and FOXP2 may provide crucial insight into the molecular pathways involved in human cognitive diseases.
References
Alarcon M, Cantor RM, Liu J, Gilliam TC, Geschwind DH (2002) Evidence for a language quantitative trait locus on chromosome 7q in multiplex autism families. Am J Hum Genet 70:60–71
Alarcon M, Abrahams BS, Stone JL, Duvall JA, Perederiy JV, Bomar JM, Sebat J, Wigler M, Martin CL, Ledbetter DH, Nelson SF, Cantor RM, Geschwind DH (2008) Linkage, association, and gene-expression analyses identify CNTNAP2 as an autism-susceptibility gene. Am J Hum Genet 82:150–159
Banham AH, Beasley N, Campo E, Fernandez PL, Fidler C, Gatter K, Jones M, Mason DY, Prime JE, Trougouboff P, Wood K, Cordell JL (2001) The FOXP1 winged helix transcription factor is a novel candidate tumor suppressor gene on chromosome 3p. Cancer Res 61:8820–8829
Bennett CL, Ochs HD (2001) IPEX is a unique X-linked syndrome characterized by immune dysfunction, polyendocrinopathy, enteropathy, and a variety of autoimmune phenomena. Curr Opin Pediatr 13:533–538
Boucher J (2011) Research review: structural language in autistic spectrum disorder—characteristics and causes. J Child Psychol Psychiatry
Carr CW, Moreno-De-Luca D, Parker C, Zimmerman HH, Ledbetter N, Martin CL, Dobyns WB, Abdul-Rahman OA (2010) Chiari I malformation, delayed gross motor skills, severe speech delay, and epileptiform discharges in a child with FOXP1 haploinsufficiency. Eur J Hum Genet 18:1216–1220
Casey JP, Magalhaes T, Conroy JM, Regan R, Shah N, Anney R, Shields DC, Abrahams BS, Almeida J, Bacchelli E, Bailey AJ, Baird G, Battaglia A, Berney T, Bolshakova N, Bolton PF, Bourgeron T, Brennan S, Cali P, Correia C, Corsello C, Coutanche M, Dawson G, de Jonge M, Delorme R, Duketis E, Duque F, Estes A, Farrar P, Fernandez BA, Folstein SE, Foley S, Fombonne E, Freitag CM, Gilbert J, Gillberg C, Glessner JT, Green J, Guter SJ, Hakonarson H, Holt R, Hughes G, Hus V, Igliozzi R, Kim C, Klauck SM, Kolevzon A, Lamb JA, Leboyer M, Le Couteur A, Leventhal BL, Lord C, Lund SC, Maestrini E, Mantoulan C, Marshall CR, McConachie H, McDougle CJ, McGrath J, McMahon WM, Merikangas A, Miller J, Minopoli F, Mirza GK, Munson J, Nelson SF, Nygren G, Oliveira G, Pagnamenta AT, Papanikolaou K, Parr JR, Parrini B, Pickles A, Pinto D, Piven J, Posey DJ, Poustka A, Poustka F, Ragoussis J, Roge B, Rutter ML, Sequeira AF, Soorya L, Sousa I, Sykes N, Stoppioni V, Tancredi R, Tauber M, Thompson AP, Thomson S, Tsiantis J, Van Engeland H, Vincent JB, Volkmar F, Vorstman JA, Wallace S, Wang K, Wassink TH, White K, Wing K, et al (2011) A novel approach of homozygous haplotype sharing identifies candidate genes in autism spectrum disorder. Hum Genet
Chokas AL, Trivedi CM, Lu MM, Tucker PW, Li S, Epstein JA, Morrisey EE (2010) Foxp1/2/4-NuRD interactions regulate gene expression and epithelial injury response in the lung via regulation of interleukin-6. J Biol Chem 285:13304–13313
Dasen JS, De Camilli A, Wang B, Tucker PW, Jessell TM (2008) Hox repertoires for motor neuron diversity and connectivity gated by a single accessory factor, FoxP1. Cell 134:304–316
Datta J, Kutay H, Nasser MW, Nuovo GJ, Wang B, Majumder S, Liu CG, Volinia S, Croce CM, Schmittgen TD, Ghoshal K, Jacob ST (2008) Methylation mediated silencing of MicroRNA-1 gene and its role in hepatocellular carcinogenesis. Cancer Res 68:5049–5058
Enard W, Przeworski M, Fisher SE, Lai CS, Wiebe V, Kitano T, Monaco AP, Paabo S (2002) Molecular evolution of FOXP2, a gene involved in speech and language. Nature 418:869–872
Enard W, Gehre S, Hammerschmidt K, Holter SM, Blass T, Somel M, Bruckner MK, Schreiweis C, Winter C, Sohr R, Becker L, Wiebe V, Nickel B, Giger T, Muller U, Groszer M, Adler T, Aguilar A, Bolle I, Calzada-Wack J, Dalke C, Ehrhardt N, Favor J, Fuchs H, Gailus-Durner V, Hans W, Holzlwimmer G, Javaheri A, Kalaydjiev S, Kallnik M, Kling E, Kunder S, Mossbrugger I, Naton B, Racz I, Rathkolb B, Rozman J, Schrewe A, Busch DH, Graw J, Ivandic B, Klingenspor M, Klopstock T, Ollert M, Quintanilla-Martinez L, Schulz H, Wolf E, Wurst W, Zimmer A, Fisher SE, Morgenstern R, Arendt T, de Angelis MH, Fischer J, Schwarz J, Paabo S (2009) A humanized version of Foxp2 affects cortico-basal ganglia circuits in mice. Cell 137:961–971
Feng X, Ippolito GC, Tian L, Wiehagen K, Oh S, Sambandam A, Willen J, Bunte RM, Maika SD, Harriss JV, Caton AJ, Bhandoola A, Tucker PW, Hu H (2010) Foxp1 is an essential transcriptional regulator for the generation of quiescent naive T cells during thymocyte development. Blood 115:510–518
Ferland RJ, Cherry TJ, Preware PO, Morrisey EE, Walsh CA (2003) Characterization of Foxp2 and Foxp1 mRNA and protein in the developing and mature brain. J Comp Neurol 460:266–279
Ferreira MA, O’Donovan MC, Meng YA, Jones IR, Ruderfer DM, Jones L, Fan J, Kirov G, Perlis RH, Green EK, Smoller JW, Grozeva D, Stone J, Nikolov I, Chambert K, Hamshere ML, Nimgaonkar VL, Moskvina V, Thase ME, Caesar S, Sachs GS, Franklin J, Gordon-Smith K, Ardlie KG, Gabriel SB, Fraser C, Blumenstiel B, Defelice M, Breen G, Gill M, Morris DW, Elkin A, Muir WJ, McGhee KA, Williamson R, MacIntyre DJ, MacLean AW, St CD, Robinson M, Van Beck M, Pereira AC, Kandaswamy R, McQuillin A, Collier DA, Bass NJ, Young AH, Lawrence J, Ferrier IN, Anjorin A, Farmer A, Curtis D, Scolnick EM, McGuffin P, Daly MJ, Corvin AP, Holmans PA, Blackwood DH, Gurling HM, Owen MJ, Purcell SM, Sklar P, Craddock N (2008) Collaborative genome-wide association analysis supports a role for ANK3 and CACNA1C in bipolar disorder. Nat Genet 40:1056–1058
Feuk L, Kalervo A, Lipsanen-Nyman M, Skaug J, Nakabayashi K, Finucane B, Hartung D, Innes M, Kerem B, Nowaczyk MJ, Rivlin J, Roberts W, Senman L, Summers A, Szatmari P, Wong V, Vincent JB, Zeesman S, Osborne LR, Cardy JO, Kere J, Scherer SW, Hannula-Jouppi K (2006) Absence of a paternally inherited FOXP2 gene in developmental verbal dyspraxia. Am J Hum Genet 79:965–972
Fisher SE, Scharff C (2009) FOXP2 as a molecular window into speech and language. Trends Genet 25:166–177
Fisher SE, Vargha-Khadem F, Watkins KE, Monaco AP, Pembrey ME (1998) Localisation of a gene implicated in a severe speech and language disorder. Nat Genet 18:168–170
Fisher SE, Lai CS, Monaco AP (2003) Deciphering the genetic basis of speech and language disorders. Annu Rev Neurosci 26:57–80
French CA, Groszer M, Preece C, Coupe AM, Rajewsky K, Fisher SE (2007) Generation of mice with a conditional Foxp2 null allele. Genesis 45:440–446
French CA, Jin X, Campbell TG, Gerfen E, Groszer M, Fisher SE, Costa RM (2011) An aetiological Foxp2 mutation causes aberrant striatal activity and alters plasticity during skill learning. Mol Psychiatry
Gauthier J, Joober R, Mottron L, Laurent S, Fuchs M, De Kimpe V, Rouleau GA (2003) Mutation screening of FOXP2 in individuals diagnosed with autistic disorder. Am J Med Genet A 118A:172–175
Geschwind DH (2011) Genetics of autism spectrum disorders. Trends Cogn Sci 15:409–416
Gregor A, Albrecht B, Bader I, Bijlsma EK, Ekici AB, Engels H, Hackmann K, Horn D, Hoyer J, Klapecki J, Kohlhase J, Maystadt I, Nagl S, Prott E, Tinschert S, Ullmann R, Wohlleber E, Woods G, Reis A, Rauch A, Zweier C (2011) Expanding the clinical spectrum associated with defects in CNTNAP2 and NRXN1. BMC Med Genet 12:106
Groszer M, Keays DA, Deacon RM, de Bono JP, Prasad-Mulcare S, Gaub S, Baum MG, French CA, Nicod J, Coventry JA, Enard W, Fray M, Brown SD, Nolan PM, Paabo S, Channon KM, Costa RM, Eilers J, Ehret G, Rawlins JN, Fisher SE (2008) Impaired synaptic plasticity and motor learning in mice with a point mutation implicated in human speech deficits. Curr Biol 18:354–362
Hamdan FF, Daoud H, Rochefort D, Piton A, Gauthier J, Langlois M, Foomani G, Dobrzeniecka S, Krebs MO, Joober R, Lafreniere RG, Lacaille JC, Mottron L, Drapeau P, Beauchamp MH, Phillips MS, Fombonne E, Rouleau GA, Michaud JL (2010) De novo mutations in FOXP1 in cases with intellectual disability, autism, and language impairment. Am J Hum Genet 87:671–678
Hannenhalli S, Kaestner KH (2009) The evolution of Fox genes and their role in development and disease. Nat Rev Genet 10:233–240
Horn D, Kapeller J, Rivera-Brugues N, Moog U, Lorenz-Depiereux B, Eck S, Hempel M, Wagenstaller J, Gawthrope A, Monaco AP, Bonin M, Riess O, Wohlleber E, Illig T, Bezzina CR, Franke A, Spranger S, Villavicencio-Lorini P, Seifert W, Rosenfeld J, Klopocki E, Rappold GA, Strom TM (2010) Identification of FOXP1 deletions in three unrelated patients with mental retardation and significant speech and language deficits. Hum Mutat 31:E1851–E1860
Hu H, Wang B, Borde M, Nardone J, Maika S, Allred L, Tucker PW, Rao A (2006) Foxp1 is an essential transcriptional regulator of B cell development. Nat Immunol 7:819–826
Jepsen K, Gleiberman AS, Shi C, Simon DI, Rosenfeld MG (2008) Cooperative regulation in development by SMRT and FOXP1. Genes Dev 22:740–745
Jin Y, Birlea SA, Fain PR, Mailloux CM, Riccardi SL, Gowan K, Holland PJ, Bennett DC, Wallace MR, McCormack WT, Kemp EH, Gawkrodger DJ, Weetman AP, Picardo M, Leone G, Taieb A, Jouary T, Ezzedine K, van Geel N, Lambert J, Overbeck A, Spritz RA (2010) Common variants in FOXP1 are associated with generalized vitiligo. Nat Genet 42:576–578
Kaufman L, Ayub M, Vincent JB (2010) The genetic basis of non-syndromic intellectual disability: a review. J Neurodev Disord 2:182–209
Konopka G, Bomar JM, Winden K, Coppola G, Jonsson ZO, Gao F, Peng S, Preuss TM, Wohlschlegel JA, Geschwind DH (2009) Human-specific transcriptional regulation of CNS development genes by FOXP2. Nature 462:213–217
Konstantoulas CJ, Parmar M, Li M (2010) FoxP1 promotes midbrain identity in embryonic stem cell-derived dopamine neurons by regulating Pitx3. J Neurochem 113:836–847
Lai CS, Fisher SE, Hurst JA, Levy ER, Hodgson S, Fox M, Jeremiah S, Povey S, Jamison DC, Green ED, Vargha-Khadem F, Monaco AP (2000) The SPCH1 region on human 7q31: genomic characterization of the critical interval and localization of translocations associated with speech and language disorder. Am J Hum Genet 67:357–368
Lai CS, Fisher SE, Hurst JA, Vargha-Khadem F, Monaco AP (2001) A forkhead-domain gene is mutated in a severe speech and language disorder. Nature 413:519–523
Lennon PA, Cooper ML, Peiffer DA, Gunderson KL, Patel A, Peters S, Cheung SW, Bacino CA (2007) Deletion of 7q31.1 supports involvement of FOXP2 in language impairment: clinical report and review. Am J Med Genet A 143A:791–798
Li S, Weidenfeld J, Morrisey EE (2004) Transcriptional and DNA binding activity of the Foxp1/2/4 family is modulated by heterotypic and homotypic protein interactions. Mol Cell Biol 24:809–822
Liegeois F, Baldeweg T, Connelly A, Gadian DG, Mishkin M, Vargha-Khadem F (2003) Language fMRI abnormalities associated with FOXP2 gene mutation. Nat Neurosci 6:1230–1237
MacDermot KD, Bonora E, Sykes N, Coupe AM, Lai CS, Vernes SC, Vargha-Khadem F, McKenzie F, Smith RL, Monaco AP, Fisher SE (2005) Identification of FOXP2 truncation as a novel cause of developmental speech and language deficits. Am J Hum Genet 76:1074–1080
Mukamel Z, Konopka G, Wexler E, Osborn GE, Dong H, Bergman MY, Levitt P, Geschwind DH (2011) Regulation of MET by FOXP2, genes implicated in higher cognitive dysfunction and autism risk. J Neurosci 31:11437–11442
Newbury DF, Monaco AP (2010) Genetic advances in the study of speech and language disorders. Neuron 68:309–320
Newbury DF, Bonora E, Lamb JA, Fisher SE, Lai CS, Baird G, Jannoun L, Slonims V, Stott CM, Merricks MJ, Bolton PF, Bailey AJ, Monaco AP (2002) FOXP2 is not a major susceptibility gene for autism or specific language impairment. Am J Hum Genet 70:1318–1327
O’Roak BJ, Deriziotis P, Lee C, Vives L, Schwartz JJ, Girirajan S, Karakoc E, Mackenzie AP, Ng SB, Baker C, Rieder MJ, Nickerson DA, Bernier R, Fisher SE, Shendure J, Eichler EE (2011) Exome sequencing in sporadic autism spectrum disorders identifies severe de novo mutations. Nat Genet 43:585–589
Otaegi G, Pollock A, Hong J, Sun T (2011) MicroRNA miR-9 modifies motor neuron columns by a tuning regulation of FoxP1 levels in developing spinal cords. J Neurosci 31:809–818
Palka C, Alfonsi M, Mohn A, Cerbo R, Franchi PG, Fantasia D, Morizio E, Stuppia L, Calabrese G, Zori R, Chiarelli F, Palka G (2012) Mosaic 7q31 deletion involving FOXP2 gene associated with language impairment. Pediatrics 129:e183–e188
Palmesino E, Rousso DL, Kao TJ, Klar A, Laufer E, Uemura O, Okamoto H, Novitch BG, Kania A (2010) Foxp1 and lhx1 coordinate motor neuron migration with axon trajectory choice by gating Reelin signalling. PLoS Biol 8:e1000446
Pariani MJ, Spencer A, Graham JM Jr, Rimoin DL (2009) A 785 kb deletion of 3p14.1p13, including the FOXP1 gene, associated with speech delay, contractures, hypertonia and blepharophimosis. Eur J Med Genet 52:123–127
Petek E, Windpassinger C, Simma B, Mueller T, Wagner K, Kroisel PM (2003) Molecular characterisation of a 15 Mb constitutional de novo interstitial deletion of chromosome 3p in a boy with developmental delay and congenital anomalies. J Hum Genet 48:283–287
Ravasi T, Suzuki H, Cannistraci CV, Katayama S, Bajic VB, Tan K, Akalin A, Schmeier S, Kanamori-Katayama M, Bertin N, Carninci P, Daub CO, Forrest AR, Gough J, Grimmond S, Han JH, Hashimoto T, Hide W, Hofmann O, Kamburov A, Kaur M, Kawaji H, Kubosaki A, Lassmann T, van Nimwegen E, MacPherson CR, Ogawa C, Radovanovic A, Schwartz A, Teasdale RD, Tegner J, Lenhard B, Teichmann SA, Arakawa T, Ninomiya N, Murakami K, Tagami M, Fukuda S, Imamura K, Kai C, Ishihara R, Kitazume Y, Kawai J, Hume DA, Ideker T, Hayashizaki Y (2010) An atlas of combinatorial transcriptional regulation in mouse and man. Cell 140:744–752
Rice GM, Raca G, Jakielski KJ, Laffin JJ, Iyama-Kurtycz CM, Hartley SL, Sprague RE, Heintzelman AT, Shriberg LD (2011) Phenotype of FOXP2 haploinsufficiency in a mother and son. Am J Med Genet A
Rousso DL, Gaber ZB, Wellik D, Morrisey EE, Novitch BG (2008) Coordinated actions of the forkhead protein Foxp1 and Hox proteins in the columnar organization of spinal motor neurons. Neuron 59:226–240
Sanjuan J, Tolosa A, Gonzalez JC, Aguilar EJ, Molto MD, Najera C, de Frutos R (2005) FOXP2 polymorphisms in patients with schizophrenia. Schizophr Res 73:253–256
Sanjuan J, Tolosa A, Gonzalez JC, Aguilar EJ, Perez-Tur J, Najera C, Molto MD, de Frutos R (2006) Association between FOXP2 polymorphisms and schizophrenia with auditory hallucinations. Psychiatr Genet 16:67–72
Shi C, Zhang X, Chen Z, Sulaiman K, Feinberg MW, Ballantyne CM, Jain MK, Simon DI (2004) Integrin engagement regulates monocyte differentiation through the forkhead transcription factor Foxp1. J Clin Invest 114:408–418
Shriberg LD, Ballard KJ, Tomblin JB, Duffy JR, Odell KH, Williams CA (2006) Speech, prosody, and voice characteristics of a mother and daughter with a 7;13 translocation affecting FOXP2. J Speech Lang Hear Res 49:500–525
Shu W, Yang H, Zhang L, Lu MM, Morrisey EE (2001) Characterization of a new subfamily of winged-helix/forkhead (Fox) genes that are expressed in the lung and act as transcriptional repressors. J Biol Chem 276:27488–27497
Shu W, Lu MM, Zhang Y, Tucker PW, Zhou D, Morrisey EE (2007) Foxp2 and Foxp1 cooperatively regulate lung and esophagus development. Development 134:1991–2000
Spiteri E, Konopka G, Coppola G, Bomar J, Oldham M, Ou J, Vernes SC, Fisher SE, Ren B, Geschwind DH (2007) Identification of the transcriptional targets of FOXP2, a gene linked to speech and language, in developing human brain. Am J Hum Genet 81:1144–1157
Takayama K, Horie-Inoue K, Ikeda K, Urano T, Murakami K, Hayashizaki Y, Ouchi Y, Inoue S (2008) FOXP1 is an androgen-responsive transcription factor that negatively regulates androgen receptor signaling in prostate cancer cells. Biochem Biophys Res Commun 374:388–393
Talkowski ME, Rosenfeld JA, Blumenthal I, Pillalamarri V, Chiang C, Heilbut A, Ernst C, Hanscom C, Rossin E, Lindgren AM, Pereira S, Ruderfer D, Kirby A, Ripke S, Harris DJ, Lee JH, Ha K, Kim HG, Solomon BD, Gropman AL, Lucente D, Sims K, Ohsumi TK, Borowsky ML, Loranger S, Quade B, Lage K, Miles J, Wu BL, Shen Y, Neale B, Shaffer LG, Daly MJ, Morton CC, Gusella JF (2012) Sequencing chromosomal abnormalities reveals neurodevelopmental loci that confer risk across diagnostic boundaries. Cell 149:525–537
Tang B, Becanovic K, Desplats PA, Spencer B, Hill AM, Connolly C, Masliah E, Leavitt BR, Thomas EA (2012) Forkhead box protein p1 is a transcriptional repressor of immune signaling in the CNS: implications for transcriptional dysregulation in Huntington disease. Hum Mol Genet
Teramitsu I, Kudo LC, London SE, Geschwind DH, White SA (2004) Parallel FoxP1 and FoxP2 expression in songbird and human brain predicts functional interaction. J Neurosci 24:3152–3163
Tolosa A, Sanjuan J, Dagnall AM, Molto MD, Herrero N, de Frutos R (2010) FOXP2 gene and language impairment in schizophrenia: association and epigenetic studies. BMC Med Genet 11:114
Tomblin JB, O’Brien M, Shriberg LD, Williams C, Murray J, Patil S, Bjork J, Anderson S, Ballard K (2009) Language features in a mother and daughter of a chromosome 7;13 translocation involving FOXP2. J Speech Lang Hear Res 52:1157–1174
Vargha-Khadem F, Watkins K, Alcock K, Fletcher P, Passingham R (1995) Praxic and nonverbal cognitive deficits in a large family with a genetically transmitted speech and language disorder. Proc Natl Acad Sci USA 92:930–933
Vargha-Khadem F, Watkins KE, Price CJ, Ashburner J, Alcock KJ, Connelly A, Frackowiak RS, Friston KJ, Pembrey ME, Mishkin M, Gadian DG, Passingham RE (1998) Neural basis of an inherited speech and language disorder. Proc Natl Acad Sci USA 95:12695–12700
Vargha-Khadem F, Gadian DG, Copp A, Mishkin M (2005) FOXP2 and the neuroanatomy of speech and language. Nat Rev Neurosci 6:131–138
Vernes SC, Spiteri E, Nicod J, Groszer M, Taylor JM, Davies KE, Geschwind DH, Fisher SE (2007) High-throughput analysis of promoter occupancy reveals direct neural targets of FOXP2, a gene mutated in speech and language disorders. Am J Hum Genet 81:1232–1250
Vernes SC, Newbury DF, Abrahams BS, Winchester L, Nicod J, Groszer M, Alarcon M, Oliver PL, Davies KE, Geschwind DH, Monaco AP, Fisher SE (2008) A functional genetic link between distinct developmental language disorders. N Engl J Med 359:2337–2345
Vernes SC, MacDermot KD, Monaco AP, Fisher SE (2009) Assessing the impact of FOXP1 mutations on developmental verbal dyspraxia. Eur J Hum Genet 17:1354–1358
Vernes SC, Oliver PL, Spiteri E, Lockstone HE, Puliyadi R, Taylor JM, Ho J, Mombereau C, Brewer A, Lowy E, Nicod J, Groszer M, Baban D, Sahgal N, Cazier JB, Ragoussis J, Davies KE, Geschwind DH, Fisher SE (2011) Foxp2 regulates gene networks implicated in neurite outgrowth in the developing brain. PLoS Genet 7:e1002145
Wang B, Weidenfeld J, Lu MM, Maika S, Kuziel WA, Morrisey EE, Tucker PW (2004) Foxp1 regulates cardiac outflow tract, endocardial cushion morphogenesis and myocyte proliferation and maturation. Development 131:4477–4487
Wassink TH, Piven J, Vieland VJ, Pietila J, Goedken RJ, Folstein SE, Sheffield VC (2002) Evaluation of FOXP2 as an autism susceptibility gene. Am J Med Genet 114:566–569
Watkins KE, Dronkers NF, Vargha-Khadem F (2002a) Behavioural analysis of an inherited speech and language disorder: comparison with acquired aphasia. Brain 125:452–464
Watkins KE, Vargha-Khadem F, Ashburner J, Passingham RE, Connelly A, Friston KJ, Frackowiak RS, Mishkin M, Gadian DG (2002b) MRI analysis of an inherited speech and language disorder: structural brain abnormalities. Brain 125:465–478
Whitehouse AJ, Bishop DV, Ang QW, Pennell CE, Fisher SE (2012) CNTNAP2 variants affect early language development in the general population. Genes Brain Behav 11:501
Williams HJ, Craddock N, Russo G, Hamshere ML, Moskvina V, Dwyer S, Smith RL, Green E, Grozeva D, Holmans P, Owen MJ, O’Donovan MC (2011) Most genome-wide significant susceptibility loci for schizophrenia and bipolar disorder reported to date cross-traditional diagnostic boundaries. Hum Mol Genet 20:387–391
Wu Y, Borde M, Heissmeyer V, Feuerer M, Lapan AD, Stroud JC, Bates DL, Guo L, Han A, Ziegler SF, Mathis D, Benoist C, Chen L, Rao A (2006) FOXP3 controls regulatory T cell function through cooperation with NFAT. Cell 126:375–387
Zeesman S, Nowaczyk MJ, Teshima I, Roberts W, Cardy JO, Brian J, Senman L, Feuk L, Osborne LR, Scherer SW (2006) Speech and language impairment and oromotor dyspraxia due to deletion of 7q31 that involves FOXP2. Am J Med Genet A 140:509–514
Zhou B, Zhong Q, Minoo P, Li C, Ann DK, Frenkel B, Morrisey EE, Crandall ED, Borok Z (2008) Foxp2 inhibits Nkx2.1-mediated transcription of SP-C via interactions with the Nkx2.1 homeodomain. Am J Respir Cell Mol Biol 38:750–758
Zilina O, Reimand T, Zjablovskaja P, Mannik K, Mannamaa M, Traat A, Puusepp-Benazzouz H, Kurg A, Ounap K (2011) Maternally and paternally inherited deletion of 7q31 involving the FOXP2 gene in two families. Am J Med Genet A
Zweier C, de Jong EK, Zweier M, Orrico A, Ousager LB, Collins AL, Bijlsma EK, Oortveld MA, Ekici AB, Reis A, Schenck A, Rauch A (2009) CNTNAP2 and NRXN1 are mutated in autosomal-recessive Pitt-Hopkins-like mental retardation and determine the level of a common synaptic protein in Drosophila. Am J Hum Genet 85:655–666
Acknowledgments
We thank Simon E. Fisher and Ute Moog for valuable comments.
Conflict of interest
The authors declare that they have no conflict of interest.
Open Access
This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Bacon, C., Rappold, G.A. The distinct and overlapping phenotypic spectra of FOXP1 and FOXP2 in cognitive disorders. Hum Genet 131, 1687–1698 (2012). https://doi.org/10.1007/s00439-012-1193-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-012-1193-z