Abstract
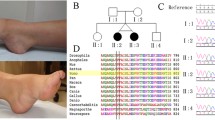
RNA sequencing (RNA-seq) is a complementary diagnostic tool to exome sequencing (ES), only recently clinically available to undiagnosed patients post-ES, that provides functional information on variants of unknown significance (VUS) by evaluating its effect on RNA transcription. ES became clinically available in the early 2010s and promised an agnostic platform for patients with a neurological disease, especially for those who believed to have a genetic etiology. However, the massive data generated by ES pose challenges in variant interpretation, especially for rare missense, synonymous, and deep intronic variants that may have a splicing effect. Without functional study and/or family segregation analysis, these rare variants would be likely interpreted as VUS which is difficult for clinicians to use in clinical care. Clinicians are able to assess the VUS for phenotypic overlap, but this additional information alone is usually not enough to re-classify a variant. Here, we report a case of a 14-month-old male who presented to clinic with a history of seizures, nystagmus, cerebral palsy, oral aversion, global developmental delay, and poor weight gain requiring gastric tube placement. ES revealed a previously unreported homozygous missense VUS, c.7406A > G p.(Asn2469Ser), in VPS13D. This variant has not been previously reported in genome aggregation database (gnomAD), ClinVar, or in any peer-reviewed published literature. By RNA-seq, we demonstrated that this variant mainly impacts splicing and results in a frameshift and early termination. It is expected to generate either a truncated protein, p.(Val2468fs*19), or no protein from this transcript due to nonsense-mediated mRNA decay leading to VPS13D deficiency. To our knowledge, this is the first case utilizing RNA-seq to further functionally characterize a homozygous novel missense VUS in VPS13D and confirm its impact on splicing. This confirmed pathogenicity gave the diagnosis of VPS13D movement disorder to this patient. Therefore, clinicians should consider utilizing RNA-seq to clarify VUS by evaluating its effect on RNA transcription.


Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article. The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Baralle D, Buratti E (2017) RNA splicing in human disease and in the clinic. Clin Sci (lond) 131(5):355–368. https://doi.org/10.1042/CS20160211
Bournazos AM, Riley LG, Bommireddipalli S, Ades L, Akesson LS, Al-Shinnag M et al (2022) Standardized practices for RNA diagnostics using clinically accessible specimens reclassifies 75% of putative splicing variants. Genet Med 24(1):130–145. https://doi.org/10.1016/j.gim.2021.09.001
Cummings BB, Marshall JL, Tukiainen T, Lek M, Donkervoort S, Foley AR, MacArthur DG et al (2017) Improving genetic diagnosis in Mendelian disease with transcriptome sequencing. Sci Transl Med 9(386). https://doi.org/10.1126/scitranslmed.aal5209
Curry PDK, Broda KL, Carroll CJ (2021) The role of RNA-sequencing as a new genetic diagnosis tool. Curr Genet Med Rep 9:13–21. https://doi.org/10.1007/s40142-021-00199-x
Durand CM, Angelini C, Michaud V, Delleci C, Coupry I, Goizet C, Trimouille A (2022) Whole-exome sequencing confirms implication of VPS13D as a potential cause of progressive spastic ataxia. BMC Neurol 22(1):53. https://doi.org/10.1186/s12883-022-02553-0
Fresard L, Smail C, Ferraro NM, Teran NA, Li X, Smith KS, Montgomery SB et al (2019) Identification of rare-disease genes using blood transcriptome sequencing and large control cohorts. Nat Med 25(6):911–919. https://doi.org/10.1038/s41591-019-0457-8
Gauthier J, Meijer IA, Lessel D, Mencacci NE, Krainc D, Hempel M et al (2018) Recessive mutations in VPS13D cause childhood onset movement disorders. Ann Neurol 83(6):1089–1095. https://doi.org/10.1002/ana.25204
Gonorazky HD, Naumenko S, Ramani AK, Nelakuditi V, Mashouri P, Wang P, Dowling JJ et al (2019) Expanding the boundaries of RNA sequencing as a diagnostic tool for rare mendelian disease. Am J Hum Genet 104(3):466–483. https://doi.org/10.1016/j.ajhg.2019.01.012
Huang X, Fan DS (2022) Autosomal recessive spinocerebellar ataxia type 4 with a VPS13D mutation: a case report. World J Clin Cases 10(2):703–708. https://doi.org/10.12998/wjcc.v10.i2.703
Kremer LS, Bader DM, Mertes C, Kopajtich R, Pichler G, Iuso A, Prokisch H et al (2017) Genetic diagnosis of Mendelian disorders via RNA sequencing. Nat Commun 8:15824. https://doi.org/10.1038/ncomms15824
Keller N, Paketci C, Altmueller J, Fuhrmann N, Wunderlich G, Schrank B, Karakaya M et al (2021) Genomic variants causing mitochondrial dysfunction are common in hereditary lower motor neuron disease. Hum Mutat 42(4):460–472. https://doi.org/10.1002/humu.24181
Koh K, Ishiura H, Shimazaki H, Tsutsumiuchi M, Ichinose Y, Nan H, Takiyama Y et al (2020) VPS13D-related disorders presenting as a pure and complicated form of hereditary spastic paraplegia. Mol Genet Genomic Med 8(3):e1108. https://doi.org/10.1002/mgg3.1108
Kwong AK, Tsang MH, Fung JL, Mak CC, Chan KL, Rodenburg RJT, Fung CW (2021) Exome sequencing in paediatric patients with movement disorders. Orphanet J Rare Dis 16(1):32. https://doi.org/10.1186/s13023-021-01688-6
Lee H, Huang AY, Wang LK, Yoon AJ, Renteria G, Eskin A, Nelson SF et al (2020a) Diagnostic utility of transcriptome sequencing for rare Mendelian diseases. Genet Med 22(3):490–499. https://doi.org/10.1038/s41436-019-0672-1
Lee JS, Yoo T, Lee M, Lee Y, Jeon E, Kim SY, Chae JH et al (2020b) Genetic heterogeneity in Leigh syndrome: highlighting treatable and novel genetic causes. Clin Genet 97(4):586–594. https://doi.org/10.1111/cge.13713
Marco-Puche G, Lois S, Benitez J, Trivino JC (2019) RNA-Seq perspectives to improve clinical diagnosis. Front Genet 10:1152. https://doi.org/10.3389/fgene.2019.01152
Meijer IA (2019) VPS13D movement disorder. In: Adam MP, Ardinger HH, Pagon RA, Wallace SE, Bean LJH, Gripp KW, Mirzaa GM, Amemiya A (eds) GeneReviews((R)). Seattle
Murdock DR, Dai H, Burrage LC, Rosenfeld JA, Ketkar S, Muller MF, Lee B et al (2021) Transcriptome-directed analysis for Mendelian disease diagnosis overcomes limitations of conventional genomic testing. J Clin Invest 131(1). https://doi.org/10.1172/JCI141500
Pertea M, Pertea GM, Antonescu CM, Chang TC, Mendell JT, Salzberg SL (2015) StringTie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat Biotechnol 33(3):290–295. https://doi.org/10.1038/nbt.3122
Petry-Schmelzer JN, Keller N, Karakaya M, Wirth B, Fink GR, Wunderlich G (2021) VPS13D: one family, same mutations, two phenotypes. Mov Disord Clin Pract 8(5):803–806. https://doi.org/10.1002/mdc3.13232
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J et al (2015) Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med 17(5):405–424. https://doi.org/10.1038/gim.2015.30
Seong E, Insolera R, Dulovic M, Kamsteeg EJ, Trinh J, Bruggemann N et al (2018) Mutations in VPS13D lead to a new recessive ataxia with spasticity and mitochondrial defects. Ann Neurol 83(6):1075–1088. https://doi.org/10.1002/ana.25220
Vural A, Simsir G, Tekgul S, Kocoglu C, Akcimen F, Kartal E, Basak AN et al (2021) The complex genetic landscape of hereditary ataxias in Turkey and implications in clinical practice. Mov Disord 36(7):1676–1688. https://doi.org/10.1002/mds.28518
Yepez VA, Gusic M, Kopajtich R, Mertes C, Smith NH, Alston CL et al (2022) Clinical implementation of RNA sequencing for Mendelian disease diagnostics. Genome Med 14(1):38. https://doi.org/10.1186/s13073-022-01019-9
Zech M, Jech R, Boesch S, Skorvanek M, Weber S, Wagner M, Winkelmann J (2020) Monogenic variants in dystonia: an exome-wide sequencing study. Lancet Neurol 19(11):908–918. https://doi.org/10.1016/S1474-4422(20)30312-4
Author information
Authors and Affiliations
Contributions
EKB, JH, WZ drafted the initial manuscript, critically reviewed, and revised the manuscript. EKB, RJH, and BEH cared the patient. JH and WZ analyzed exome sequencing. WL and MR analyzed the RNA sequencing. All authors commented on previous versions.
Corresponding author
Ethics declarations
Conflict of interest
William Langley and Michael Roett are both employees of MNG Laboratories, a Labcorp company.
Informed consent
Written and verbal informed consent from the legal guardian was obtained for publication purposes including facial pictures of the manuscript. All authors read and approved the final manuscript.
Additional information
Communicated by Shuhua Xu.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Baker, E.K., Han, J., Langley, W.A. et al. RNA sequencing reveals a complete picture of a homozygous missense variant in a patient with VPS13D movement disorder: a case report and review of the literature. Mol Genet Genomics 298, 1185–1199 (2023). https://doi.org/10.1007/s00438-023-02044-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-023-02044-y