Abstract
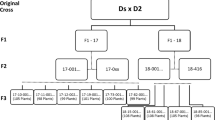
The cultivated potato (Solanum tuberosum L.) is an autotetraploid species. The complexity of tetrasomic inheritance and the lack of pure lines increase the difficulty of genetic analysis of the inherited characteristics. Tuberization is the determinant step for economic yield of potato. To understand the complex genetic basis of tuberization of the cultivated potato, we developed linkage maps for a tetraploid population (F1) of 237 genotypes and mapped QTLs for the percent of in vitro tuberized plantlets (% IVT). The paternal map for E108 (well tuberized) covered 948 cM and included 12 linkage groups, all of which contained all four homologous chromosomes. The maternal map for E20 (nontuberized) covered 1,286 cM and included 14 linkage groups, 12 of which contained all four homologous chromosomes. All 12 chromosomes of potato were tagged using the SSR markers. A major QTL (MT05) with additive effect was detected on chromosome V of E108 which explained 16.23 % of the variation for % IVT, and two minor QTLs (mt05 and mt09) displaying simplex dominant effects were located on chromosome V and chromosome IX of E20 which explained 5.33 and 4.59 % of the variation for % IVT, respectively. Based on the additive model of MT05, the segregation ratio of the gametic genotypes (Q−: qq = 5:1) matched the ratio of the tuberized genotypes to the nontuberized genotypes in the population suggesting that the segregation of in vitro tuberization in this population is controlled by a major-effect gene or genes. The mapping results of three important candidate genes indicated that the QTL causal genes detected in our study are new. In this study, we developed the almost complete linkage maps of a tetraploid population, identified a major QTL on chromosome V affecting in vitro tuberization, suggested a major-effect gene with minor modifiers model controlling this trait and found that the QTLs identified here correspond to new tuberization genes. Our work provides new and useful information about the genetic basis for tuberization of this autotetraploid crop.






Similar content being viewed by others
References
Alsadon AA (1989) Micropropagation techniques as a tool for studying plant growth, tuberization and sprouting of potatoes. Diss Abst Int Biol Sci Eng 50:810B–811B
Alsadon AA, Knutson KW, Wilkinson JC (1988) Relationships between microtuber and minituber production and yield characteristics of six potato cultivars. Am Potato J 65:468
Bachem CW, van der Hoeven RS, de Bruijn SM, Vreugdenhil D, Zabeau M, Visser GF (1996) Visualization of differential gene expression using a novel method of RNA fingerprinting based on AFLP: analysis of gene expression during potato tuber development. Plant J 9:745–753
Bormann CA, Rickert AM, Castillo Ruiz RA, Paal J, Lubeck J, Strahwald J, Buhr K, Gebhardt C (2004) Tagging quantitative trait loci for maturity-corrected late blight resistance in tetraploid potato with PCR-based candidate gene markers. Mol Plant Microbe Interact 17(10):1126–1138
Bradshaw HD Jr, Wilbert SM, Otto KG, Schemske DW (1995) Genetic mapping of floral traits associated with reproductive isolation in monkeyflowers (Mimulus). Nature 376:762–765
Bradshaw JE, Bryan GJ, Hackett CA, McLean K, Pande B, Stewart HE, Waugh R (2004) Dissection and analysis of quantitative disease resistance in tetraploid potato. Euphytica 137(1):13–18
Bradshaw JE, Hackett CA, Pande B, Waugh R, Bryan GJ (2008) QTL mapping of yield, agronomic and quality traits in tetraploid potato (Solanum tuberosum subsp. tuberosum). Theor Appl Genet 116:193–211
Churchill GA, Doerge RW (1994) Empirical threshold values for quantitative trait mapping. Genetics 138:963–971
Collins A, Molbourne D, Ramsay L, Meyer R, Chatot-Balandras C, Oberhagemann P, De Jong W, Gebhardt C, Bonnel E, Waugh R (1999) QTL for field resistance to late blight in potato are strongly correlated with maturity and vigor. Mol Breed 5:387–398
Danan S, Veyrieras JB, Lefebvre V (2011) Construction of a potato consensus map and QTL meta-analysis offer new insights into the genetic architecture of late blight resistance and plant maturity traits. BMC Plant Biol 11:16
Dellaporta SL, Wood J, Hicks JB (1983) A plant DNA minipreparation: version II. Plant Mol Biol Rep 1:19–21
Dodds JH, Huaman Z, Lizarraga R (1991) Potato germplasm conservation. In: Dodds JH (ed) In vitro methods for conservation of plant genetic resources. Chapman and Hall, London, pp 93–109
Ewing EE, Struik PC (1992) Tuber formation in potato: induction, initiation, and growth. Hortic Rev Am Soc Hortic Sci 14:89–197
Ewing EE, Wareing PF (1978) Shoot, stolon, and tuber formation on potato (Solanum tuberosum L.) cuttings in response to photoperiod. Plant Physiol 61:348–353
Feingold S, Lloyd J, Norero N, Bonierbale M, Lorenzen J (2005) Mapping and characterization of new EST-derived microsatellites for potato (Solanum tuberosum L.). Theor Appl Genet 111:456–466
Garner N, Blake J (1989) The induction and development of potato microtubers in vitro on media free of growth regulating substances. Ann Bot 63:663–674
González-Schain ND, Díaz-Mendoza M, Zurczak M, Suárez-López P (2012) Potato CONSTANS is involved in photoperiodic tuberization in a graft-transmissible manner. Plant J 70:678–690
Gopal J (1996) In vitro selection, genetic divergence and cross prediction in potato. Ph D Thesis. Punjab Agricultural University, Ludhiana, Punjab
Gopal J (2001) In vitro and in vivo genetic parameters and character associations in potato. Euphytica 118:145–151
Gopal J, Minocha JL (1998) Effectiveness of in vitro selection for agronomic characters in potato. Euphytica 103:67–74
Groth J, Song Y, Kellermann A, Schwarzfischer A (2013) Molecular characterisation of resistance against potato wart races 1, 2, 6 and 18 in a tetraploid population of potato (Solanum tuberosum subsp. tuberosum). J Appl Genetics 54:169–178
Hackett CA, Bradshaw JE, McNicol JW (2001) Interval mapping of quantitative trait loci in autotetraploid species. Genetics 159:1819–1832
Hackett CA, Pande B, Bryan GJ (2003) Constructing linkage maps in autotetraploid species using simulated annealing. Theor Appl Genet 106:1107–1115
Hackett CA, Milne I, Bradshaw JE, Luo Z (2007) TetraploidMap for Windows: linkage map construction and QTL mapping in autotetraploid species. J Hered 98:727–729
Han YC, Teng CZ, Hu ZL, Song YC (2008) An optimal method of DNA silver staining in polyacrylamide gels. Electrophoresis 29:1355–1358
Hannapel DJ (2010) A model system of development regulated by the long-distance transport of mRNA. J Integr Plant Biol 52:40–52
Henry Y, Vain P, De Buyser J (1994) Genetic analysis of in vitro plant tissue culture response and regeneration capacities. Euphytica 79:45–48
Hussey G, Stacey NJ (1984) Factors affecting the formation of in vitro tubers of potato (Solanum tuberosum L.). Ann Bot 53:565–578
Inui H, Ogura Y, Kiyosue T (2010) Overexpression of Arabidopsis thaliana LOV KELCH REPEAT PROTEIN 2 promotes tuberization in potato (Solanum tuberosum cv. May Queen). FEBS Lett 584:2393–2396
Jansky SH, Davis GL, Peloquin SJ (2004) A genetic model for tuberization in potato haploid-wild species hybrids grown under long-day conditions. Am J Potato Res 81:335–339
Khu D, Lorenzen J, Hackett CA, Love SL (2008) Interval mapping of quantitative trait loci for corky ringspot disease resistance in a tetraploid population of potato (Solanum tuberosum subsp. tuberosum). Am J Potato Res 85:129–139
Kittipadukal P, Bethke PC, Jansky SH (2012) The effect of photoperiod on tuberisation in cultivated × wild potato species hybrids. Potato Res 55:27–40
Kloosterman B, Abelenda JA, Gomez Mdel M, Oortwijn M, de Boer JM, Kowitwanich K, Horvath BM, Van Eck HJ, Smaczniak C, Prat S, Visser RG, Bachem CW (2013) Naturally occurring allele diversity allows potato cultivation in northern latitudes. Nature 495:246–250
Koornneef M, Hanhart CJ, van der Veen JH (1991) A genetic and physiological analysis of late flowering mutants in Arabidopsis thaliana. Mol Gen Genet 229:57–66
Li JC, Lindqvist-Kreuze H, Tian ZD, Liu J, Song BT, Landeo J, Portal L, Gastelo M, Frisancho J, Sanchez L, Meijer D, Xie CH, Bonierbale M (2012) Conditional QTL underlying resistance to late blight in a diploid potato population. Theor Appl Genet 124:1339–1350
Luo ZW, Hackett CA, Bradshaw JE, McNicol JW, Milbourne DM (2000) Predicting parental genotypes and gene segregation for tetrasomic inheritance. Theor Appl Genet 100:1067–1073
Luo ZW, Hackett CA, Bradshaw JE, McNicol JW, Milbourne DM (2001) Construction of a genetic linkage map in tetraploid species using molecular markers. Genetics 157:1369–1385
Malosetti M, Visser RGF, Celis-Gamboa C, van Eeuwijk FA (2006) QTL methodology for response curves on the basis of non-linear mixed models, with an illustration to senescence in potato. Theor Appl Genet 113:288–300
Martínez-García JF, Virgos-Sóler A, Prat S (2002) Control of photoperiod-regulated tuberization in potato by the Arabidopsis flowering-time gene CONSTANS. Proc Natl Acad Sci USA 99:15211–15216
McCord PH, Sosinski BR, Haynes KG, Clough ME, Yencho GC (2011) QTL mapping of internal heat necrosis in tetraploid potato. Theor Appl Genet 122:129–142
Mendoza HA, Haynes FL (1977) Inheritance of tuber initiation in tuber bearing Solanum as influenced by photoperiod. Am Potato J 54:243–253
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassay with tobaco tissue cultures. Physiol Plant 15:473–497
Naik PS, Sarkar D, Gaur PC (1998) Yield components of potato microtubers: in vitro production and field performance. Ann Appl Biol 133:91–99
Navarro C, Abelenda JA, Cruz-Oro E, Cuellar CA, Tamaki S, Silva J, Shimamoto K, Prat S (2011) Control of flowering and storage organ formation in potato by FLOWERING LOCUS T. Nature 478:119–122
Park DH, Somers DE, Kim YS, Choy YH, Lim HK, Soh MS, Kim HJ, Kay SA, Nam HG (1999) Control of circadian rhythms and photoperiodic flowering by the Arabidopsis GIGANTEA gene. Science 285:1579–1582
Roca WM, Bryan JE, Roca MR (1979) Tissue culture for international transfer of potato genetic resources. Am Potato J 55:1–10
Rodríguez-Falcón M, Bou J, Prat S (2006) Seasonal control of tuberization in potato: conserved elements with the flowering response. Annu Rev Plant Biol 57:151–180
Ronning CM, Stegalkina SS, Ascenzi RA, Bougri O, Hart AL, Utterbach TR, Vanaken SE, Riedmuller SB, White JA, Cho J, Pertea GM, Lee Y, Karamycheva S, Sultana R, Tsai J, Quackenbush J, Griffiths HM, Restrepo S, Smart CD, Fry WE, van der Hoeven R, Tanksley S, Zhang P, Jin H, Yamamoto ML, Baker BJ, Buell CR (2003) Comparative analyses of potato expressed sequence tag libraries. Plant Physiol 131:419–429
Simko I, Costanzo S, Ramanjulu V, Christ BJ, Haynes KG (2006) Mapping polygenes for tuber resistance to late blight in a diploid Solanum phureja × S. stenotomum hybrid population. Plant Breeding 125:385–389
Śliwka J, Jakuczun H, Lebecka R, Marczewski W, Gebhardt C, Zimnoch-Guzowska E (2007) Tagging QTLs for late blight resistance and plant maturity from diploid wild relatives in a cultivated potato (Solanum tuberosum) background. Theor Appl Genet 115(1):101–112
Sørensen K, Kirk H, Olsson K, Labouriau R, Christiansen J (2008) A major QTL and an SSR marker associated with glycoalkaloid content in potato tubers from Solanum tuberosum × S. sparsipilum located on chromosome I. Theor Appl Genet 117:1–9
Suárez-López P, Wheatley K, Robson F, Onouchi H, Valverde F, Coupland G (2001) CONSTANS mediates between the circadian clock and the control of flowering in Arabidopsis. Nature 410:1116–1120
Turck F, Fornara F, Coupland G (2008) Regulation and identity of florigen: FLOWERING LOCUS T moves center stage. Annu Rev Plant Biol 59:573–594
Van den Berg JH, Ewing EE, Plaisted RL, McMurry S, Bonierbale MW (1996) QTL analysis of potato tuberization. Theor Appl Genet 93:307–316
Van Os H, Andrzejewski S, Bakker E, Barrena I, Bryan GJ, Caromel B, Ghareeb B, Isidore E, De Jong W, van Koert P, Lefebvre V, Mil-bourne D, Ritter E, van der Voort JNAMR, Rousselle-Bourgeois F, van Vliet J, Waugh R, Visser RGF, Bakker J, van Eck HJ (2006) Construction of a 10,000-marker ultradense genetic recombination map of potato: providing a framework for accelerated gene isolation and a genome wide physical map. Genetics 173:1075–1087
Visker M, Keizer LCP, Van Eck HJ, Jacobsen ELT, Colon LT, Struik PC (2003) Can the QTL for late blight resistance on potato chromosome 5 be attributed to foliage maturity type? Theor Appl Genet 106:317–325
Visker MHPW, Heilersig HJB, Kodde LP, van de Weg WE, Voorrips RE, Struik PC, Colon LT (2005) Genetic linkage of QTLs for late blight resistance and foliage maturity type in six related potato progenies. Euphytica 143:189–199
Voorrips RE (2002) MapChart: software for the graphical presentation of linkage maps and QTLs. J Hered 93:77–78
Vos P, Hogers R, Bleeker M, Reijans M, van de Lee T, Hornes M, Friters A, Pot J, Paleman J, Kuiper M, Zabeau M (1995) AFLP: a new technique for DNA fingerprinting. Nucleic Acid Res 23:4407–4414
Yanovsky MJ, Izaguirre M, Wagmaister JA, Gatz C, Jackson SD, Thomas B, Casal JJ (2000) Phytochrome A resets the circadian clock and delays tuber formation under long days in potato. Plant J 23:223–232
Acknowledgments
This work was supported by the earmarked fund for Modern Agro-industry Technology Research System (CARS-10-P08). We are thankful to Dr. Yongbing Ou and Dr. Jingcai Li for reading this manuscript and giving kind advice. The experiments comply with the current laws of China where they were performed.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Hohmann.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhou, J., Fang, H., Shan, J. et al. A major QTL located on chromosome V associates with in vitro tuberization in a tetraploid potato population. Mol Genet Genomics 289, 575–587 (2014). https://doi.org/10.1007/s00438-014-0832-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-014-0832-6