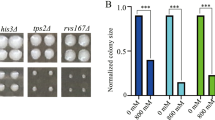
Abstract
Cobalt is an essential micronutrient but is toxic when present in excess. To study cobalt homeostasis we performed a genome-wide screen for deletion strains that show sensitivity or resistance to CoCl2. Among 54 cobalt-sensitive strains, 18 are supersensitive strains, which are involved in histidine biosynthetic process, ubiquitination, mitochondria function, membrane trafficking, transporter and a variety of other known functions or still unknown functions. Furthermore, we identified 56 cobalt-resistant deletion strains, which are mainly involved in mitochondria function, signal transduction, ubiquitination, and gene expression and chromatin remodeling. Notably, deletion of the zhf1 + gene, encoding a zinc ion transporter, confers supersensitivity to cobalt and overexpression of the zhf1 + gene confers marked tolerance to cobalt, indicating that Zhf1 play key roles in cobalt detoxification. Interestingly, all the histidine-auxotrophic mutants displayed cobalt sensitivity and deletion of cationic amino acid transporter Cat1, which was shown to be involved in histidine uptake, suppressed the CoCl2-sensitive growth defect of the his2 mutants, suggesting that CoCl2 may be transported into the cell together with histidine via histidine transporters including Cat1. In addition, we obtained results suggesting that the E2 ubiquitin conjugating enzyme Rhp6 and Sty1 stress MAP kinase pathway are involved in the regulation of cobalt homeostasis. Altogether, our genome-wide study demonstrates for the first time the mechanisms of cobalt homeostasis, particularly its uptake and detoxification in fission yeast.







Similar content being viewed by others
Abbreviations
- CRE:
-
cAMP-response element
- EMM:
-
Edinburgh minimal medium
- MAPK:
-
Mitogen-activated protein kinase
- ORF:
-
Open reading frame
- RLU:
-
Relative light units
- YES:
-
Yeast extract with supplements
- YPD:
-
Yeast extract–peptone–dextrose
References
Aspuria PJ, Tamanoi F (2008) The Tsc/Rheb signaling pathway controls basic amino acid uptake via the Cat1 permease in fission yeast. Mol Genet Genomics 279:441–450
Beyersmann D, Hartwig A (1992) The genetic toxicology of cobalt. Toxicol Appl Pharmacol 115:137–145
Calvo IA, Gabrielli N, Iglesias-Baena I, Garcia-Santamarina S, Hoe KL, Kim DU, Sanso M, Zuin A, Perez P, Ayte J, Hidalgo E (2009) Genome-wide screen of genes required for caffeine tolerance in fission yeast. PLoS One 4:e6619
Catalani S, Rizzetti MC, Padovani A, Apostoli P (2012) Neurotoxicity of cobalt. Hum Exp Toxicol 31:421–437
Cirla AM (1994) Cobalt-related asthma: clinical and immunological aspects. Sci Total Environ 150:85–94
Clemens S, Bloss T, Vess C, Neumann D, Nies DH, Zur Nieden U (2002) A transporter in the endoplasmic reticulum of Schizosaccharomyces pombe cells mediates zinc storage and differentially affects transition metal tolerance. J Biol Chem 277:18215–18221
Conklin DS, McMaster JA, Culbertson MR, Kung C (1992) COT1, a gene involved in cobalt accumulation in Saccharomyces cerevisiae. Mol Cell Biol 12:3678–3688
Dalhamn T (1953) The effect of cobalt on blood pressure, respiration and mortality, and the use of dimercaprol (BAL) as antidote. Acta Pharmacol Toxicol (Copenh) 9:259–266
Fischer T, Rystedt I (1983) Cobalt allergy in hard metal workers. Contact Dermat 9:115–121
Gardner FH (1953) The use of cobaltous chloride in the anemia associated with chronic renal disease. J Lab Clin Med 41:56–64
Heim R, Cubitt AB, Tsien RY (1995) Improved green fluorescence. Nature 373:663–664
Kamizono A, Nishizawa M, Teranishi Y, Murata K, Kimura A (1989) Identification of a gene conferring resistance to zinc and cadmium ions in the yeast Saccharomyces cerevisiae. Mol Gen Genet 219:161–167
Kasten U, Mullenders LH, Hartwig A (1997) Cobalt(II) inhibits the incision and the polymerization step of nucleotide excision repair in human fibroblasts. Mutat Res 383:81–89
Kennedy PJ, Vashisht AA, Hoe KL, Kim DU, Park HO, Hayles J, Russell P (2008) A genome-wide screen of genes involved in cadmium tolerance in Schizosaccharomyces pombe. Toxicol Sci 106:124–139
Kim DU, Hayles J, Kim D, Wood V, Park HO, Won M, Yoo HS, Duhig T, Nam M, Palmer G, Han S, Jeffery L, Baek ST, Lee H, Shim YS, Lee M, Kim L, Heo KS, Noh EJ, Lee AR, Jang YJ, Chung KS, Choi SJ, Park JY, Park Y, Kim HM, Park SK, Park HJ, Kang EJ, Kim HB, Kang HS, Park HM, Kim K, Song K, Song KB, Nurse P, Hoe KL (2010) Analysis of a genome-wide set of gene deletions in the fission yeast Schizosaccharomyces pombe. Nat Biotechnol 28:617–623
Kita A, Sugiura R, Shoji H, He Y, Deng L, Lu Y, Sio SO, Takegawa K, Sakaue M, Shuntoh H, Kuno T (2004) Loss of Apm1, the micro1 subunit of the clathrin-associated adaptor-protein-1 complex, causes distinct phenotypes and synthetic lethality with calcineurin deletion in fission yeast. Mol Biol Cell 15:2920–2931
Leonard S, Gannett PM, Rojanasakul Y, Schwegler-Berry D, Castranova V, Vallyathan V, Shi X (1998) Cobalt-mediated generation of reactive oxygen species and its possible mechanism. J Inorg Biochem 70:239–244
Li L, Kaplan J (1998) Defects in the yeast high affinity iron transport system result in increased metal sensitivity because of the increased expression of transporters with a broad transition metal specificity. J Biol Chem 273:22181–22187
Lin H, Kumanovics A, Nelson JM, Warner DE, Ward DM, Kaplan J (2008) A single amino acid change in the yeast vacuolar metal transporters ZRC1 and COT1 alters their substrate specificity. J Biol Chem 283:33865–33873
Ma Y, Jiang W, Liu Q, Ryuko S, Kuno T (2011) Genome-wide screening for genes associated with FK506 sensitivity in fission yeast. PLoS One 6:e23422
MacDiarmid CW, Gaither LA, Eide D (2000) Zinc transporters that regulate vacuolar zinc storage in Saccharomyces cerevisiae. EMBO J 19:2845–2855
Millar JB, Buck V, Wilkinson MG (1995) Pyp1 and Pyp2 PTPases dephosphorylate an osmosensing MAP kinase controlling cell size at division in fission yeast. Genes Dev 9:2117–2130
Moreno S, Klar A, Nurse P (1991) Molecular genetic analysis of fission yeast Schizosaccharomyces pombe. Methods Enzymol 194:795–823
Pearce DA, Sherman F (1999) Toxicity of copper, cobalt, and nickel salts is dependent on histidine metabolism in the yeast Saccharomyces cerevisiae. J Bacteriol 181:4774–4779
Rodrigue A, Effantin G, Mandrand-Berthelot MA (2005) Identification of rcnA (yohM), a nickel and cobalt resistance gene in Escherichia coli. J Bacteriol 187:2912–2916
Stadler JA, Schweyen RJ (2002) The yeast iron regulon is induced upon cobalt stress and crucial for cobalt tolerance. J Biol Chem 277:39649–39654
Thorgersen MP, Downs DM (2007) Cobalt targets multiple metabolic processes in Salmonella enterica. J Bacteriol 189:7774–7781
Toda T, Dhut S, Superti-Furga G, Gotoh Y, Nishida E, Sugiura R, Kuno T (1996) The fission yeast pmk1 + gene encodes a novel mitogen-activated protein kinase homolog which regulates cell integrity and functions coordinately with the protein kinase C pathway. Mol Cell Biol 16:6752–6764
Torii S, Kurihara A, Li XY, Yasumoto K, Sogawa K (2011) Inhibitory effect of extracellular histidine on cobalt-induced HIF-1alpha expression. J Biochem 149:171–176
Zhou X, Ma Y, Sugiura R, Kobayashi D, Suzuki M, Deng L, Kuno T (2010) MAP kinase kinase kinase (MAPKKK)-dependent and -independent activation of Sty1 stress MAPK in fission yeast. J Biol Chem 285:32818–32823
Acknowledgments
We thank Susie O. Sio for critical reading of the manuscript, and Kayo Osaka for her contribution to the preliminary stages of this study. Financial support for this study was provided by Grant-in-Aid for Scientific Research, on Scientific Research (B) to Takayoshi Kuno (No. 22390048) and on Grant-in-Aid for Young Scientists (B) to Yan Ma (No. 21790241), from Japan Society for the Promotion of Science (http://www.jsps.go.jp/english/e-grants/grants.html).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by T. Ito.
S. Ryuko and Y. Ma contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ryuko, S., Ma, Y., Ma, N. et al. Genome-wide screen reveals novel mechanisms for regulating cobalt uptake and detoxification in fission yeast. Mol Genet Genomics 287, 651–662 (2012). https://doi.org/10.1007/s00438-012-0705-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-012-0705-9