Abstract
Strigea falconis is a common parasite of birds of prey and owls widely distributed in the Holarctic. We aimed to characterise S. falconis from Iceland via integrative taxonomic approach and to contribute to the understanding of its circulation in the Holarctic. We recovered adult S. falconis from two gyrfalcons (Falco rusticolus) collected in 2011 and 2012 in Iceland (Reykjanes Peninsula, Westfjords) and characterised them by morphological and molecular genetic (D2 of rDNA, cox1, ND1 of the mDNA) methods. We provide the first species record of S. falconis in Iceland which to the best of our knowledge is its northernmost distributional range. The presence of S. falconis in Iceland is surprising, as there are no suitable intermediate hosts allowing completion of its life cycle. Gyrfalcons are fully sedentary in Iceland; thus, the only plausible explanation is that they acquired their infection by preying upon migratory birds arriving from Europe. Our data indicate that the most likely candidates are Anseriformes and Charadriiformes. Also, we corroborate the wide geographical distribution of S. falconis, as we found a high degree of similarity between our haplotypes and sequences of mesocercariae from frogs in France and of a metacercaria from Turdus naumanni in Japan, and adults from Buteo buteo and Circus aeruginosus from the Czech Republic. The case of Strigea falconis shows the advantages of a complex life cycle and also depicts its pitfalls when a parasite is introduced to a new area with no suitable intermediate hosts. In Iceland, gyrfalcons are apparently dead-end hosts for S. falconis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Strigea falconis Szidat, 1928 of the family Strigeidae Railliet, 1919 is a trematode parasite widely distributed in the Holarctic region, reported from a wide range of birds of prey and owls (Bykhovskaya-Pavlovskaya 1962; Dubois 1968; Sudarikov 1959, 1984; Krone and Streich 2000; Heneberg et al. 2018). The systematics of the family Strigeidae is still quite unsettled, and DNA sequences of S. falconis and other members of the family have been scarce. So far, Heneberg et al. (2018) provided the most comprehensive analysis of European strigeids applying the approach of integrative taxonomy. Most recently, there have been attempts to tackle the phylogenetic relationships of the species of the genus Strigea Abildgaard, 1790 and related genera (López-Jiménez et al. 2023), which together with Heneberg et al. (2018) confirmed the status of S. falconis as a well-defined species.
Strigea falconis has a complex, four-host life cycle involving several intermediate and paratenic hosts (Bykhovskaya-Pavlovskaya 1962; Dubois 1968; Sudarikov 1959, 1984). The pulmonate snail Planorbis planorbis (L.) (Gastropoda, Planorbidae) is used as the first intermediate host (Odening 1964); the second intermediate hosts are tadpoles and frogs harbouring the unencysted stage of mesocercaria; as the third intermediate hosts, amphibians, snakes, birds and mammals are used which harbour the encysted stage of metacercariae, and some of these hosts are considered acting as paratenic hosts (Sudarikov 1959, 1984; Odening 1967; Dubois 1968, Niewiadomska (2002); Sitko et al. 2006). Concerning definitive hosts, Heneberg et al. (2018) confirmed by DNA sequence analysis, that S. falconis is using primarily birds of prey, particularly Buteo buteo (L.) and Circus aeruginosus (L.) (the type-host), less frequently species of Falco, while owls are considered as rare definitive hosts. Krone and Streich (2000) noted that S. falconis is the most often recorded trematode in birds of prey in Europe, and indeed, there are numerous reports regarding it as a common, generalist parasite of birds of prey (see Okulewicz et al. 1993; Sitko 1998; Sanmartín et al. 2004; Sitko et al. 2006; Santoro et al. 2010; Jantošková and Halán 2014; Komorová et al. 2016, 2017; Zafra et al. 2022). Dubois (1968) in his compilation provided a long list of definitive hosts (Accipitriformes, Falconiformes and Strigiformes) from the Holarctic, including Great Britain, where S. falconis was recorded from Falco rusticolus islandus Brünnich, which is to the best of our knowledge the only record of S. falconis from a gyrfalcon (see Baylis 1939).
The gyrfalcon, Falco rusticolus L., has a circumpolar distribution, and its populations are found breeding in the high and the sub-Arctic (Cramp and Simmons 1980; Koskimies and Sulkava 2011; Nielsen 2011). In Iceland, the estimated size of the gyrfalcon population is 3–400 breeding pairs (Icelandic Institute of Natural History (IINH), 2000), and the birds are fully sedentary (Nielsen and Cade 1990a). The gyrfalcons are prey-specialists relying on willow and rock ptarmigan (Lagopus lagopus (L.), L. muta (Montin)); however, during summer, ptarmigan is supplemented by alternative prey, mainly waterfowl (Anseriformes), waders (Charadriiformes), auks (Alcidae) and mammals (hares, microtine rodents) (Bengtson 1971; Nielsen 2003, 2011; Nielsen and Cade 1990b). Given the range of prey, gyrfalcons might be potential hosts for several trematodes; however, the records have so far been quite sporadic (Clausen and Gudmundsson 1981; Christensen et al. 2015). As mentioned above, Baylis (1939) recorded S. falconis from a gyrfalcon in Great Britain (Scotland), and Dubois and Rausch (1965) reported another strigeid, Strigea macropharynx Dubois & Rausch, 1965, and a diplostomid, Neodiplostomum spathula banghami Penrod, 1947, from a gyrfalcon in North America, Alaska. In Iceland, Clausen and Gudmundsson (1981) recorded Plagiorchis elegans (Rudolphi, 1802) from gyrfalcons; and Christensen (2013) and Christensen et al. (2015) found few trematode species in a large-scale sample of gyrfalcons: Cryptocotyle concava (Creplin, 1825), C. lingua (Creplin, 1825), Cryptocotyle sp., Levinseniella propinqua Jägerskiöld, 1907, Microphallus pygmaeus (Levinsen, 1881), P. elegans and Strigea sp., the latter not being further identified. Except for Strigea sp., the authors assumed that the trematodes might have been accidentally ingested with prey and left the question open if gyrfalcons are used as proper definitive hosts by most of these species.
In this study, our aim was to identify via an integrative taxonomic approach these Strigea spp. isolated from gyrfalcons in Iceland and to contribute to the understanding of the reason for their presence in this host at these latitudes and their circulation within the Holarctic zone. This new species record for Iceland is the more surprising, as the intermediate hosts (P. planorbis, amphibians, reptiles) necessary for completion of the four-host life cycle of S. falconis are absent in Iceland, while gyrfalcons are sedentary (Nielsen and Cade 1990a). Below, we provide data on morphology combined with genetic data and information on the geographical distribution and the life cycle of S. falconis including a possible explanation for the record from Iceland.
Material and methods
Sampling and processing of bird hosts and parasite material
This study, focused on digenetic trematodes of Strigea spp., benefited from a collection of carcasses of gyrfalcons, Falco rusticolus, provided by the Icelandic Institute of Natural History in Garðabær, and which was the basis for a thesis examining their endo- and ectoparasites (Christensen 2013; and Christensen et al. 2015). In total, 25 gyrfalcon carcasses from Iceland were examined for helminth endoparasites (for details see Christensen 2013), and those specimens preliminarily identified as Strigea spp. were set aside for the present study.
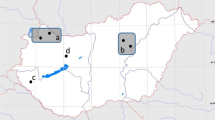
The two gyrfalcons infected with Strigea spp. were collected in the western part of Iceland: (i) a 2nd cal. year male (id. N. Fr-12–39) was found dead on 31 May 2011 at Vogastapi, Vatnsleysuströnd, Reykjanes (63° 58′ 14.791″ N 22° 27′ 44.120″ W); (ii) a 2nd cal. year male (id. N. Fr-12–30) was found emaciated on 3 July 2012 in Önundarfjörður, Westfjords (65° 59′ 09.8″ N 23° 22′ 23.8″ W). The carcasses were frozen as a whole and stored in a freezer until dissection. Combined, a total of six adult and ten juvenile Strigea spp. were found in the birds’ intestines. The worms were recovered from the intestines under a stereomicroscope, washed in saline, rinsed and fixed in 70% ethanol for further analyses. We followed the concept of Pleijel et al. (2008) and cut a small piece of the posterior part of two adult specimens (hologenophores) for DNA sequence analyses; one well-preserved, gravid adult was kept as a paragenophore.
Morphological examination
The material of the recovered worms was identified with aid of Dubois (1968) and the key of Niewiadomska (2002). The three vouchers (two hologenophores and the paragenophore, see above) were stained in iron acetocarmine, dehydrated in ethanol, cleared in clove oil and mounted in Canada balsam. Detailed morphological examination was carried out with aid of a light microscope Olympus BX51; drawings were made with aid of a drawing attachment. Measurements were taken from microphotographs of total mounts in Canada balsam with aid of ImageJ image analysis software (Schneider et al. 2012). All measurements are given in micrometres as the range.
For the description, we used the terminology of Niewiadomska (2002); for anterior and posterior parts of body, we used the terms ‘prosoma’ and ‘opisthosoma’ proposed by Achatz et al. (2021) for Diplostomoidea.
The voucher material of three specimens of S. falconis is deposited in the Icelandic Institute for Natural History (IINH), Urriðaholtsstræti 6, 210 Garðabær, Iceland.
DNA processing and phylogenetic analyses
A small piece of the posterior part of two adult specimens (hologenophores ex birds Fr-12–30 and Fr-12–39) was used for the molecular analysis (Table 1). After removing ethanol from the samples, DNA was extracted using the QIAamp DNA Mini Kit (Qiagen, Germany) following manufacturer’s instructions. During the first step (tissue lysis), parasites were crushed one by one using a piston pellet (Treff, Switzerland). Polymerase chain reactions (PCR) of the D2 domain of the 28S subunit of rDNA were performed under conditions described by Jouet et al. (2009) with primers C2′B (5′-GAA AAG TAC TTT GRA RAG AGA-3′) and D2 (5′-TCC GTG TTT CAA GAC GGG-3′) (Mollaret et al. 1997). Two domains of the mitochondrial DNA were amplified and sequenced using a couple of primers: (i) forward NDJ11 (5′-AGA TTC GTA AGG GGC CTA ATA-3′) and reverse NDJ2a (5′-CTT CAG CCT CAG CAT AAT-3′) for the cytochrome c oxidase subunit I (cox1) according to Kostadinova et al. (2003); (ii) and JB3 (5′-TTT TTT GGG CAT CCT GAG GTT TAT-3′) and JB4.5 (5′-TAA AGA AAG AAC ATA ATG AAA ATG-3′) for the NADH dehydrogenase subunit 1 (ND1) according to Bowles et al. (1995). PCR products were directly sequenced in both directions with the primers used for DNA amplification (GenoScreen, France). The sequences obtained from this study are deposited in GenBank under the accession numbers PP218148–PP218149, PP229533–PP229534 and PP239093–PP239094 (Table 1).
Sequences were aligned using ClustalW that is included in MEGA version 11 software (Tamura et al. 2021) and then checked by eye. Sequences obtained were compared with sequences of Strigeidae previously identified from different developmental stages available in GenBank Database (Apatemon spp.: 158 sequences, Apharyngostrigea spp.: 57 sequences, Australapatemon spp.: 313 sequences, Cardiocephaloides spp.: 65 sequences, Cotylurus spp.: 286 sequences, Ichthyocotylurus spp.: 26 sequences, Nematostrigea spp.: 6 sequences, Parastrigea spp.: 77 sequences, Strigea spp.: 69 sequences and Strigeidae sp.: 61 sequences). The D2 domain of the rDNA (556 bp) and the partial ND1 (395 bp) and cox1 (236 bp) domains of the mDNA were used for molecular comparisons and tree constructions. Phylogenetic trees were constructed based on previous analyses (Heneberg et al. 2018; López-Jiménez et al. 2023) and new taxa available in GenBank (Table 2), using the neighbour joining (NJ), the maximum likelihood (ML) and minimum evolution (ME) methods, using the MEGA version 11 software. Bayesian inference (BI) analysis was conducted using MrBayes v. 3.2.6 (Huelsenbeck and Ronquist 2001; Dereeper et al. 2008, 2010). Markov chain Monte Carlo (MCMC) chains were run for 10,000,000 generations, and a sampling frequency of 100 generations resulted in 100,000 generations being saved. A burn‐in setting of 25% was used. The most appropriate nucleotide substitution model was determined (HKY + G for D2 and ND1; HKY + G + I for cox1). For NJ, ML and ME analyses, gaps were treated as missing data and internal node support was assessed by bootstrapping over 1000 replicates. Tylodelphys aztecae (Acc. N. MF398837), Hysteromorpha triloba (Acc. N. MH536511) and H. triloba (Acc. N. MF628069) were used as outgroup for D2, ND1 and cox1 domains, respectively. Visualisation of the trees and distance matrices for all datasets were calculated in MEGA ver. 11 (Tamura et al. 2021).
Results
Morphological description of the voucher material
Strigeidae Railliet, 1919
Strigea Abildgaard, 1790
Strigea falconis Szidat, 1928
Host: Falco rusticolus L., 1758.
Locality: Önundarfjörður Westfjords Region (65° 59′ 09.8″ N 23° 22′ 23.8″ W); Vogastapi, Vatnsleysuströnd, Reykjanes Peninsula (N 63° 58′ 14.791″ W 22° 27′ 44.120″), Iceland.
Site in host: small intestine.
Infection rates: intensity—2 adult specimens and 10 juveniles per bird from 2011; 4 adult worms per bird from 2012.
Material: two hologenophores STR2 (Coll. No. IINH 301234, ex bird Fr-12–39, coll. 31.v. 2011) and STR3 (Coll. No. IINH 301235, ex bird Fr-12–30, coll. 3.vii. 2012), one paragenophore STR1 (Coll. No. IINH 301233, ex bird Fr-12–39, coll. 31.v. 2011).
Representative DNA sequences: Isolates STR2 and STR3 (see Table 1)—EA7510 ESCAPE-DJ-Reims, France.
Adult (Fig. 1 )
[Description and measurements based on three specimens fixed in ethanol (not all specimens contributed a data point to all variables, e.g., total body length taken only from paragenophore).] Body distinctly bipartite, neck-region absent, slightly curved, 3313 long. Prosoma cup-shaped, with maximum width at its mid-level, well separated from opisthosoma, 766–903 × 666–827. Opisthosoma long, sub-cylindrical, elongate, with almost parallel margins, slightly curved, 2546 × 655–892, with slight constriction just anterior to copulatory bursa. Opisthosoma much longer than prosoma; opisthosoma/prosoma length ratio 1:3.3. Tegument smooth.
Oral sucker small, weakly muscular, elongate-oval, ventro-subterminal, 75–143 × 68–113. Ventral sucker weakly muscular, sub-spherical, at mid-level of prosoma; larger than oral sucker, 197–217 × 166–247; ventral sucker/oral sucker length ratio 1:1.52–2.62. Pseudosuckers present, of similar size as oral sucker. Prepharynx absent. Pharynx well-developed, feebly muscular, 74–123 × 61–90. Oesophagus, intestinal bifurcation and caeca in prosoma not observed. Caeca long, narrow, posterior extension not observed. Holdfast organ in prosoma, consisting of two large lobes, slightly protruding out of prosoma, with a deep slit. Proteolytic gland distinct, at base of prosoma, 166–262 × 192–287.
Testes two, large, tandem, lobed, post-ovarian, contiguous, in second and last third of opisthosoma; post-testicular region 434 long, 17% of opisthosoma length. Anterior testis square-shaped to transversely-oval, with three lobes turned dorsally, 313–635 × 565–824. Posterior testis slightly smaller, square-shaped or asymmetrical, with two lobes turned dorsally, 650 × 577–782. Seminal vesicle coiled, post-testicular, relatively small.
Ovary reniform, entire, or lobed, sub-median, pretesticular, contiguous with anterior testis; in second quarter of opisthosoma, 176–232 × 166–368. Vitellarium follicular, vitelline follicles numerous, small, present in prosoma and opisthosoma. In prosoma, vitellarium extending up to its three quarters or more. In opisthosoma, vitelline follicles confluent anteriorly, filling its full width up to level of ovary, at level of testes forming a ventral field expanding to half-width of body, in post-testicular region confluent, extending ventrally to copulatory bursa. Vitelline reservoir conspicuous, between testes. Mehlis’ gland diffuse, dorsal, between testes. Uterus extending well in front of ovary (first quarter of opisthosoma), continuing on ventral side towards posterior end. Eggs numerous, 83–111 × 55–66. Copulatory bursa large, 454 × 618, becoming wider towards posterior extremity, opening terminally, occupying 18% of opisthosoma length. Genital cone well delimited from parenchyma, with muscular ring (Ringnapf), just post-testicular, 452 × 288. Excretory vesicle and excretory pore not observed.
Remarks
The present material agrees well with the diagnosis of the genus Strigea Abildgaard, 1790 by Niewiadomska (2002) in the presence of a distinctly bipartite body, with vitelline follicles evenly distributed in both prosoma and opisthosoma, in prosoma extending into body wall and holdfast organ, well-developed pharynx, multilobed testes and large copulatory bursa with genital cone well delimited from parenchyma and with a muscular ring.
The morphology of our present material agrees well with the description of Strigea falconis by Dubois (1938, 1968) in the presence of a cup-shaped prosoma with the holdfast organ slightly protruding from its opening, well-developed suckers and pharynx, large, lobed testes, vitelline follicles evenly distributed in both parts of body, in prosoma never covering pharynx and oral sucker and terminating near the copulatory bursa in opisthosoma, and well-pronounced copulatory bursa with a well-delimited genital cone. Our morphometric data fall within the dimensions provided by Dubois (1938, 1968), only the oral sucker and pharynx show lower minima, while testes are wider. When comparing our dimensions to those of Heneberg et al. (2018), who provided measurements of 30 individuals ex Buteo buteo linked to sequences, our material overlaps as well; however, it exhibits lower minima for prosoma, suckers, pharynx, anterior ovary width and anterior testis length, while on the other side, the sucker length ratio shows slightly higher maxima (1:1.52–2.62 vs. 1:1.3–2.3) (see also Supplementary Information, Table S1). Our material of S. falconis can be well distinguished from S. falconis brasiliana Szidat, 1929, considered a subspecies by Dubois (1968). Our specimens of S. falconis differ in size relation of the genital cone to the ovary (genital cone of similar size as ovary vs. genital cone larger than ovary), in larger eggs (83–111 × 55–66 vs. 67–91 × 42–55), and in geographical distribution, as S. falconis brasiliana is restricted to Cuba and South America (see Dubois 1968; Drago et al. 2015; and Table S1). Based on phylogenetic analyses, S. falconis is most close to S. magnirostris López-Jiménez et al., 2023 and S. macrobursa (Drago & Lunaschi, 2011) from Mexico and South America. Our material of S. falconis differs from S. magnirostris in absence of papillae on oral sucker, absence of neck region in opisthosoma, tegument being devoid of spines, in the opisthosoma to prosoma ratio (3.3 vs. 3.3–5.3) and in possessing larger testes. From S. macrobursa, our material differs in larger body (3313 vs. 957–2880), lobed testes (vs. not lobed), presence of a muscular ring (vs. muscular ring absent), absence of tegumental spines and very shallow genital atrium (vs. very deep). From S. macropharynx described from a gyrfalcon in Alaska by Dubois and Rausch (1965), our material differs in a larger body (3313 vs. 790–2110) and larger prosoma (766–903 × 666–827 vs. 450–700 × 320–480), in the presence of a much smaller pharynx (74–123 × 61–90 vs. 135–165 × 110–145) being substantially smaller than oral sucker (vs. pharynx larger than oral sucker), and a short, inconspicuous hermaphroditic duct (vs. long, muscular). From the North American S. macroconophora Dubois & Rausch, 1950 ex Buteo jamaicensis borealis with similarly distributed vitelline follicles (see Dubois and Rausch 1965), our material differs in possessing larger testes, a smaller genital cone (452 × 288 vs. 340–660 × 405–510) and a smaller copulatory bursa (taking c. 1/5 of opisthosoma length vs. taking 1/3 of opisthosoma length) and a wider extent of vitelline follicles in prosoma (extending up to its three quarters or more vs. extending to its half) (see also Table S1).
Molecular identification and phylogenetic analyses
The Bayesian inference and phylogenetic analyses (NJ, ML and ME) of the ribosomal (D2) and mitochondrial (ND1 and cox1) are congruent with previous studies and confirmed the separation of the Strigeidae in two major clades, corresponding to the two tribes of Strigeinae Railliet, 1919, i.e. Strigeini Dubois, 1936 and Cotylurini Dubois, 1936 (Fig. 2).
Phylogenetic tree based on the D2 domain of rDNA of sequences of Strigeidae constructed using the maximum likelihood (ML) method. The scale shows the number of nucleotide substitutions per site between DNA sequences. Nodal support from Bayesian inference (BI) and neighbour joining (NJ), maximum likelihood and Minimum evolution (ME) analyses indicated as BI/(NJ-ML-ME), with values > 0.90 (BI) and ‘*’ for bootstrap values higher than 85% in ML, NJ and ME
Molecular analyses and comparisons of the D2 domain of the large ribosomal subunit (rDNA) were conducted based on 55 sequences available in GenBank from 9 genera and 30 species or lineages of Strigeidae and sequences obtained in the present study. No variation was observed between our sequences isolated from gyrfalcons in Iceland and the sequence of Strigea sp. (Acc. N. KT362372) of a mesocercaria isolated from Pelophylax sp. in France, and only one variation (i.e. a homology of 99.4%) with the sequence of Strigea sp. (Acc. N. KT362373) also of a mesocercaria from Pelophylax sp. in France and the sequence of Strigeidae sp. (Acc. N. LC011455) of a metacercaria from Turdus naumanni in Japan was observed. The phylogenetic analysis of this domain (Fig. 2) shows that the genus Strigea is split into two separate clades (confirmed by intraspecific and interspecific variations, see Tables S2 and S3): (i) the first clade includes the type-species Strigea strigis, as well as the European species Strigea robusta (syn. Parastrigea robusta); (ii) the second clade contains the South American species Strigea magnirostris, Strigea macrobursa (syn. P. macrobursa), the two haplotypes of Strigea sp. and Strigeidae sp. and sequences from this study. These two clades of Strigea spp. are separated by another clade comprising the genera Parastrigea Szidat, 1928 and Apharyngostrigea Ciurea, 1927 (see Fig. 2).
Molecular analyses and comparisons of the ND1 and partial cox1 domains of the mDNA were used at the generic and specific level. For the ND1 (395 bp, 35 sequences), intraspecific variations (4 to 6 variations, i.e. homology of 99.2%) between haplotypes STR2 and STR3 and sequences of Strigea falconis isolated from Accipiter nisus, Aquila pomarina, Aquila heliaca, Buteo buteo and Circus aeruginosus from the Czech Republic (Acc. Ns. MF628024–MF628029 and KT074969–KT074970), and interspecific variations (73 to 88 variations) with other species of Strigea confirmed the affiliation of our adults from gyrfalcons to the European S. falconis.
Molecular and phylogenetic analyses of the partial cox1 (236 bp, 64 sequences) show the presence of two clades within the Strigea spp.: (i) a clade comprising the type-species Strigea strigis, as well as the species S. robusta, S. macrobursa, S. sphaerula and S. vandenbrokae; (ii) a second clade comprising the species S. magnirostris and S. falconis. Intraspecific variations (6 variations, 99.9% of homology) confirmed that the haplotypes of adults isolated from gyrfalcon in Iceland belong to the species Strigea falconis (see Table S4). Interspecific variability with other species of Strigea spp. varies from 12.9% (29 variations—e.g. S. falconis vs. S. magnirostris) to 14.9% (50 variations—e.g. S. falconis vs. S. robusta) (see Table S5).
For both ND1 and partial cox1 domains, all of the well-known species represent distinct taxa well supported by BI and bootstrap values (see Figs. 3 and 4).
Phylogenetic tree based on the ND1 domain of mDNA of sequences of Strigeidae constructed using the maximum likelihood (ML) method. The scale shows the number of nucleotide substitutions per site between DNA sequences. Nodal support from Bayesian inference (BI) and neighbour joining (NJ), maximum likelihood and minimum evolution (ME) analyses indicated as BI/(NJ-ML-ME), with values > 0.98 (BI) and ‘*’ for bootstrap values higher than 95% in ML, NJ and ME
Phylogenetic tree based on the cox1 domain of mDNA of sequences of Strigeidae constructed using the maximum likelihood (ML) method. The scale shows the number of nucleotide substitutions per site between DNA sequences. Nodal support from Bayesian inference (BI) and neighbour joining (NJ), maximum likelihood and minimum evolution (ME) analyses indicated as BI/(NJ-ML-ME), with values > 0.95 (BI) and ‘*’ for bootstrap values higher than 95% in ML, NJ and ME
Discussion
In the present study, we provide the first record of S. falconis from Iceland, and at the same time, the first record of S. falconis from a gyrfalcon host supported by molecular genetic characterisation combined with morphology. From Iceland, there were reports on Strigea sp. by Christensen (2013) and Christensen et al. (2015). The species-level identification of their record is accomplished in the present study, supporting the wide geographical distribution of S. falconis and proving that it extends to the sub-Arctic.
The first record of S. falconis from F. rusticolus islandus was provided by Baylis (1939) from Argyllshire, Scotland; however, without any further details, therefore we can only speculate about the origin of the Icelandic gyrfalcon in Scotland. Apart from the finding of Baylis (1939), there is only one more record of S. falconis from gyrfalcons from a kennel in Tuva, Russia, in South Siberia (Davydova et al. 2016); however, this identification should be viewed with caution, as it is based on eggs only and is not supported by molecular genetic data. The only other strigeid from a gyrfalcon is S. macropharynx described by Dubois and Rausch (1965) from Alaska, USA. This scarcity of trematode records from gyrfalcon is most probably because of the birds’ restricted geographical distribution (Arctic, sub-Arctic) and their trophic specialisation on rock and willow ptarmigan, which are not the permissive intermediate hosts for transmission of metacercariae to gyrfalcons. Therefore, we assume that gyrfalcons are rather uncommon hosts of trematodes. Although gyrfalcons prey upon a wide variety of birds other than ptarmigan, it seems that the trematodes so far recorded in Iceland could have been ingested rather accidentally with prey (Cryptocotyle spp., L. propinqua, M. pygmaeus and P. elegans; see Christensen 2013; Christensen et al. 2015).
The present case of S. falconis in Iceland is probably the most northern record of this species, and it is beyond doubt unusual. Based on the information we have on the life cycle of S. falconis, it is impossible to be completed in Iceland. The Icelandic gyrfalcon population is fully sedentary, i.e. not migrating southwards to Europe for wintering (Nielsen and Cade 1990a), where it could get a trematode infection as it happens, e.g. to waterfowl acquiring bird schistosomes in wintering places (Kolářová et al. 2006; 2013; Jouet et al. 2015). There are no appropriate snail intermediate hosts present in Iceland, although there is a recent single record of P. planorbis from central Iceland (Academy of Natural Sciences (ANS) 2024), which is unusual as this snail species is not known to expand to higher latitudes (Glöer 2019). And even though there are small planorbid snails (Gyraulus parvus (Say) and Bathyomphalus contortus (L.)) (Faltýnková et al., 2023; own observation), which potentially could act as alternative first intermediate hosts, other organisms serving as second intermediate hosts (amphibians and reptiles) are missing completely. The only plausible explanation is that the Icelandic gyrfalcons acquired their infection by preying upon migratory birds (infected with metacercariae) arriving from Europe where they got infected during wintering by ingesting amphibians or reptiles carrying mesocercariae of S. falconis.
As mentioned above, the main diet of gyrfalcons is ptarmigan, but this type of prey can be supplemented by other birds, such as waterfowl, waders, shorebirds, gulls, auks, passerines and even other raptors in summer and winter, of which many are migratory (Nielsen 2003, 2011; Nielsen and Cade 1990b, Table S6). This spectrum might overlap with the wide array of bird species recorded as hosts of metacercariae of S. falconis (previously also reported under the name Tetracotyle ardeae Mataré, 1910) including Ciconiiformes, Passeriformes, Podicipediformes, Ralliformes, Pelecaniformes, Anserifores, Charadriiformes, Falconiformes, Coraciiformes, Galliformes, Columbiformes, Strigiformes and Piciformes (Sudarikov 1959, 1984; Bykhovskaya-Pavlovskaya 1962; Dubois 1968). Several species of these bird families were recorded as prey items of gyrfalcons in Iceland, indicating mainly species of Anseriformes and Charadriiformes as the most likely candidates (see Table S6). As the location of the metacercariae in birds, the subcutaneous tissue, the area near oesophagus or trachea, body cavity and adipose tissue was reported (Odening 1967; Dubois 1968; Krone and Streich 2000; Syrota et al. 2021). In more recent time, in Germany, Buteo buteo was reported to host metacercariae of S. falconis by Krone and Streich (2000), while in Ukraine, Ardea purpurea and Nycticorax nycticorax were recorded (Syrota et al. 2021); in Korea, Egretta alba modesta Gray was recorded by Ryang et al. (1991). Certainly, the reports are scarce now because it is nearly impossible to obtain birds for parasitological examination, but the few reports document the presence S. falconis in the ecosystems. On the other hand, the fairly wide host spectrum recorded in the older literature casts some doubt if only S. falconis was involved, particularly because metacercariae are the life cycle stage with least characters for identification. The birds act as third intermediate and most likely as paratenic hosts, accumulating metacercariae over time (long-time survival of metacercariae in their hosts is known) and ensuring their dispersion. As Iceland lies on the East Atlantic bird flyway, we can assume that a transfer of metacercariae by migratory birds is highly likely. The western part of Iceland (the Reykjanes Peninsula and the Westfjords) is used by a wide array of bird species for breeding, and some species stop in this region while migrating to Greenland (Wilson 1981; Garðarsson 1999; Doyle et al. 2021).
The taxonomy of the Strigeinae is still controversial, due to the absence of easily identifiable morphological characters, or variations depending on the stage or maturity of the parasites, and the hosts from which they have been isolated, leading to misidentifications in the past (Heneberg et al. 2018). The recent use of an integrative approach combining morphology, molecular biology and the study of parasites and their hosts is essential and has made it possible to confirm or place species in synonymy and to understand the taxonomy within several genera of this family (Blasco-Costa et al. 2016; Achatz et al. 2020; Locke et al. 2021; Pyrka et al. 2021; Sokolov et al. 2021). Regarding the genus Strigea, López-Jiménez et al. (2023) analysed the South American species based on different nuclear and mitochondrial domains. Heneberg et al. (2018) showed the separation of the European species into three clades (Strigea sensu stricto, Amphistoma and Cryptostrigea), proved the well-defined status of S. falconis and assumed that it might belong to a separate genus (Cryptostrigea). The new species described since then and the results obtained in this study confirm this hypothesis, and another recently described species, Strigea magnirostris, falls in the Cryptostrigea clade. However, there is still not enough evidence to put the transfer to Cryptostrigea into effect because more molecular data for other species and genera are needed to reinforce this hypothesis. Records supported by molecular genetic data are still scarce for S. falconis, apart from Heneberg et al. (2018), the only other finding is that of mesocercariae ex Natrix natrix from Poland by Bełcik et al. (2022), whose molecular data concern only the small subunit of ribosomal RNA gene, making a comparison with our data impossible. Moreover, concerning haplotypes of the adults isolated from the gyrfalcons in Iceland, our material corresponds morphologically to those found in Central Europe in Buteo buteo of which Heneberg et al. (2018) provided DNA sequences. Our analyses also show a high degree of similarity in D2 between our haplotypes of S. falconis and the sequences of mesocercariae (Strigea sp.) isolated from frogs in France (Patrelle et al. 2015). This could indicate the circulation of S. falconis in wetlands in France, with frogs being the source of infection for aquatic birds. Given the conserved nature of 28S ribosomal DNA, and the absence of sequences for other mitochondrial domains for these taxa, a comparison with other markers is necessary to confirm their identity. For the D2 (28S), our haplotypes are also close to the sequence of a metacercaria (Strigeidae sp.) from Turdus naumanni in Japan (Iwaki et al. 2014, unpublished). Unfortunately, the lack of sequences for other genetic domains and of additional data on this taxon means that we are unable to identify whether it could be the same species. Finally, for a better phylogenetic resolution, more genetic data from other continents than Europe are needed (only seven species are available out of 47 described), particularly from South America. Obtaining sequences especially of Strigea falconis var. brasiliana is of interest because this sub-species, which has been frequently found, shows substantial morphological differences (see ‘Remarks’) to the Holarctic species, suggesting that it could be a well-defined, separate species.
Strigea falconis illustrates that a complex life cycle with multiple intermediate hosts (including paratenic hosts) can be advantageous for a parasite (see Poulin 2007; Parker et al. 2015; Benesh et al. 2021), particularly in combination with low host specificity, and can lead to its wide dispersion. This is indicated by the large-scale distribution of S. falconis and its common occurrence in birds of prey. At the same time, this case study shows one of the pitfalls of such a complex life cycle when a parasite is introduced to a new area with no suitable intermediate hosts. Thus, gyrfalcons in Iceland are apparently dead-end hosts for S. falconis.
Data availability
Data are available from the authors upon reasonable request; sequences are made available via GenBank.
References
Academy of Natural Sciences (ANS) (2024) Malacology collection at the Academy of Natural Sciences of Philadelphia. Occurrence dataset https://doi.org/10.15468/xp1dhx accessed via GBIF.org on 2024-01-27. https://www.gbif.org/occurrence/215859593
Achatz TJ, Pulis EE, González-Acuña D, Tkach VV (2020) Phylogenetic relationships of Cardiocephaloides spp. (Digenea, Diplostomoidea) and the genetic characterization of Cardiocephaloides physalis from Magellanic penguin, Spheniscus magellanicus, in Chile. Acta Parasitol 65:525–534. https://doi.org/10.2478/s11686-019-00162-5
Achatz TJ, Bell JA, Melo FTV, Fecchio A, Tkach VV (2021) Phylogenetic position of Sphincterodiplostomum Dubois, 1936 (Digenea: Diplostomoidea) with description of a second species from Pantanal. Brazil J Helminthol 95(e6):1–8. https://doi.org/10.1017/S0022149X21000018
Baylis HA (1939) Further records of parasitic worms from British vertebrates. Ann Mag Nat Hist, Ser 11(23):473–498. https://doi.org/10.1080/00222933908527013
Bełcik A, Różycki M, Korpysa-Dzirba W, Marucci G, Fafiński Z, Fafińska P, Karamon J, Kochanowski M, Cencek T, Bilska-Zając E (2022) Grass snakes (Natrix natrix) as a reservoir of Alaria alata and other parasites. Pathogens 11(2):156. https://doi.org/10.3390/pathogens11020156
Benesh DP, Parker G, Chubb JC (2021) Life-cycle complexity in helminths: what are the benefits? Evolution 75:1936–1952. https://doi.org/10.1111/evo.14299
Bengtson S-A (1971) Hunting methods and choice of prey of gyrfalcons Falco rusticolus at Mývatn in Northeast Iceland. Ibis 113:468–476
Bennett J, Presswell B, Poulin R (2023) Tracking life cycles of parasites across a broad taxonomic scale in a marine ecosystem. Int J Parasitol 53:285–303. https://doi.org/10.1016/j.ijpara.2023.02.004
Blasco-Costa I, Poulin R, Presswell B (2016) Species of Apatemon Szidat, 1928 and Australapatemon Sudarikov, 1959 (Trematoda: Strigeidae) from New Zealand: linking and characterising life cycle stages with morphology and molecules. Parasitol Res 115:271–289. https://doi.org/10.1007/s00436-015-4744-0
Bowles J, Blair D, McManus DP (1995) A molecular phylogeny of the human schistosomes. Mol Phylogenet Evol 4:103–109. https://doi.org/10.1006/mpev.1995.1011
Bykhovskaya-Pavlovskaya IE (1962) The trematode fauna of birds of the USSR. An ecological and geographical account. Izdatelstvo Akademii Nauk SSSR, Moscow, Leningrad. (in Russian)
Christensen ND (2013) Parasitic fauna of the gyrfalcon (Falco rusticolus) in Iceland. MSc Thesis, University of Copenhagen, Faculty of Science, Copenhagen
Christensen ND, Skírnisson K, Nielsen ÓK (2015) The parasite fauna of the gyrfalcon (Falco rusticolus) in Iceland. J Wildl Dis 51:929–933. https://doi.org/10.7589/2015-01-022
Clausen B, Gudmundsson F (1981) Causes of mortality among free-ranging gyrfalcons in Iceland. J Wildl Dis 17:105–109. https://doi.org/10.7589/0090-3558-17.1.105
Cramp S, Simmons KEL (1980) Handbook of the birds of Europe, the Middle East and North Africa. The birds of the Palearctic., vol. II, Hawks and buzzards. Oxford University Press, Oxford
Davydova OE, Vasilevitch FI, Pimenov NV (2016) Clinical and helminthological characteristics of Falconiformes birds contained in the conditions of kennel. Russ J Agricult Soc-Econ Sci 54:58–69. https://doi.org/10.18551/rjoas.2016-06.08
de Buron I, Hill-Spanik KM, Baker T, Fignar G, Broach J (2023) Infection of Atlantic tripletail Lobotes surinamensis (Teleostei: Lobotidae) by brain metacercariae Cardiocephaloides medioconiger (Digenea: Strigeidae). PeerJ 11:e15365. https://doi.org/10.7717/peerj.15365
Dereeper A, Guignon V, Blanc G, Audic S, Buffet S, Chevenet F, Dufayard JF, Guindon S, Lefort V, Lescot M, Claverie JM, Gascuel O (2008) Phylogeny. fr: robust phylogenetic analysis for the non-specialist. Nucleic Acids Res 36(Web Server):W465-9. https://doi.org/10.1093/nar/gkn180
Dereeper A, Audic S, Claverie JM, Blanc G (2010) BLAST-EXPLORER helps you building datasets for phylogenetic analysis. BMC Evol Biol 12(10):8. https://doi.org/10.1186/1471-2148-10-8
Doyle S, Cabot D, Griffin L, Kane A, Colhoun K, Bearhop S, McMahon BJ (2021) Spring and autumn movements of an Arctic bird in relation to temperature and primary production. J Avian Biol e02830. https://doi.org/10.1111/jav.02830
Drago FB, Lunaschi LI, Cabrela NE, Barbieri L (2015) Helminth parasites of four species of strigiform birds from Central and Northeastern Argentina. Rev Arg Parasitol 4:15–23
Dubois G (1938) Monographie des Strigeida (Trematoda). Mém Soc Neuchateloise Sci Nat 6:1–503
Dubois G (1968) Synopsis des Strigeidae et des Diplostomatidae (Trematoda). Mém Soc Neuchateloise Sci Nat 10:1–259
Dubois G, Rausch RL (1965) Studies on the helminth fauna of Alaska. XLIII. Strigea macropharynx sp. n., a trematode parasite of Falco rusticolus L. J Parasitol 51:770–772. https://doi.org/10.2307/3276154
Faltýnková A, Kudlai O, Pantoja C, Jouet D, Skírnisson K (2023) Prey‑mimetism in cercariae of Apatemon (Digenea, Strigeidae) in freshwater in northern latitudes. Par Res 122(3):815–831. https://doi.org/10.1007/s00436-023-07779-6
Garðarsson A (1999) The density of seabirds west of Iceland. Rit Fiskideildar 16:155–169
Glöer P (2019) The freshwater gastropods of the West-Palaearctis. Volume 1. Fresh and brackish waters except spring and subterranean snails. Muchowdruck, Neustadt
Hammoud C, Kayenbergh A, Tumusiime J, Verschuren D, Albrecht C, Huyse T, Van Bocxlaer B (2022) Trematode infection affects shell shape and size in Bulinus tropicus. Int J Parasitol Par Wildl 18:300–311. https://doi.org/10.1016/j.ijppaw.2022.07.003
Heneberg P, Sitko J, Bizos J (2016) Molecular and comparative morphological analysis of central European parasitic flatworms of the superfamily Brachylaimoidea Allison, 1943 (Trematoda: Plagiorchiida). Parasitology 143:455–474. https://doi.org/10.1017/S003118201500181X
Heneberg P, Sitko J, Těšínský M, Rząd I, Bizos J (2018) Central European Strigeidae Railliet, 1919 (Trematoda: Strigeidida): molecular and comparative morphological analysis suggests the reclassification of Parastrigea robusta Szidat, 1928 into Strigea Abildgaard, 1790. Parasitol Int 67:688–701. https://doi.org/10.1016/j.parint.2018.07.003
Hernández-Mena DI, García-Varela M, Pérez-Ponce de León G (2017) Filling the gaps in the classification of the Digenea Carus, 1863: systematic position of the Proterodiplostomidae Dubois, 1936 within the superfamily Diplostomoidea Poirier, 1886, inferred from nuclear and mitochondrial DNA sequences. Syst Parasitol 94:833–848. https://doi.org/10.1007/s11230-017-9745-1
Huelsenbeck JP, Ronquist F (2001) MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics 17:754–755. https://doi.org/10.1093/bioinformatics/17.8.754
Icelandic Institute of Natural History (IINH) 2000. Red data list 2 – birds. https://www.ni.is/en/research/monitoring-and-research/voktun-falka (Nielsen OK)
Jantošková H, Halán M (2014) The helminth fauna of captive and wild birds of prey in Prievidza district, Central Slovakia. 57th Student Scientific Conference, Košice, 9 April, 2014. Folia Vet 28:89–91
Jouet D, Ferté H, Hologne C, Kaltenbach ML, Depaquit J (2009) Avian schistosomes in French aquatic birds: a molecular approach. J Helminthol 83:181–189. https://doi.org/10.1017/S0022149X09311712
Jouet D, Kolářová L, Patrelle C, Ferté H, Skírnisson K (2015) Trichobilharzia anseri n. sp. (Schistosomatidae: Digenea), a new visceral species of avian schistosomes isolated from greylag goose (Anser anser L.) in Iceland and France. Infect Genet Evol 34:298–306. https://doi.org/10.1016/j.meegid.2015.06.012
Kolářová L, Rudolfová J, Hampl V, Skírnisson K (2006) Allobilharzia visceralis gen. nov., sp. nov. (Schistosomatidae-Trematoda) from Cygnus cygnus (L.) (Anatidae). Parasitol Int 55:179–186. https://doi.org/10.1016/j.parint.2005.10.009
Kolářová L, Skírnisson K, Ferté H, Jouet D (2013) Trichobilharzia mergi sp. nov. (Trematoda: Digenea: Schistosomatidae), a visceral schistosome of Mergus serrator (L.) (Aves: Anatidae). Parasitol Int 62:300–308. https://doi.org/10.1016/j.parint.2013.03.002
Komorová P, Sitko J, Špakulová M, Hurníková Z (2016) Intestinal and liver flukes of birds of prey (Accipitriformes, Falconiformes, Strigiformes) from Slovakia: uniform or diverse compound? Parasitol Res 115:2837–2844. https://doi.org/10.1007/s00436-016-5034-1
Komorová P, Sitko J, Špakulová M, Hurníková Z, Sałamatin R, Chovancová G (2017) New data on helminth fauna of birds of prey (Falconiformes, Accipitriformes, Strigiformes) in the Slovak Republic. Helminthologia 54:314–321. https://doi.org/10.1515/helm-2017-0038
Koskimies P, Sulkava S (2011) Diet of the gyrfalcon (Falco rusticolus) in northern Fennoscandia. In: Watson RT, Cade TJ, Fuller M, Hunt G, Potapov E (eds) Gyrfalcons and Ptarmigan in a Changing World. The Peregrine Fund, Boise, Idaho, USA, pp 177–190. https://doi.org/10.4080/gpcw.2011.0400
Kostadinova A, Herniou E, Barrett J, Littlewood DTJ (2003) Phylogenetic relationships of Echinostoma Rudolphi, 1809 (Digenea: Echinostomatidae) and related genera re-assessed via DNA and morphological analyses. Syst Parasitol 54:159–176. https://doi.org/10.1023/A:1022681123340
Krone O, Streich WJ (2000) Strigea falconispalumbi in Eurasian buzzards from Germany. J Wildl Dis 36:559–561. https://doi.org/10.7589/0090-3558-36.3.559
Lebedeva DI, Yakovleva GA (2016) Trematoda Nematostrigea serpens (Nitzsch 1819) Sandground 1934, a new species in the parasite fauna of birds in Karelia. Biol Bull Russ Acad Sci 43:1018–1023. https://doi.org/10.1134/S1062359016090065
Locke SA, Van Dam A, Caffara M, Pinto HA, López-Hernández D, Blanar CA (2018) Validity of the Diplostomoidea and Diplostomida (Digenea, Platyhelminthes) upheld in phylogenomic analysis. Int J Parasitol 48:1043–1059. https://doi.org/10.1016/j.ijpara.2018.07.001
Locke SA, Drago FB, López-Hernández D, Chibwana FD, Núñez V, Van Dam A, Achinelly MF, Johnson PTJ, de Assis JCA, de Melo AL, Pinto HA (2021) Intercontinental distributions, phylogenetic position and life cycles of species of Apharyngostrigea (Digenea, Diplostomoidea) illuminated with morphological, experimental, molecular and genomic data. Int J Parasitol 51:667–683. https://doi.org/10.1016/j.ijpara.2020.12.006
López-Jiménez A, González-García MT, García-Varela M (2022) Molecular and morphological evidence suggests the reallocation from Parastrigea brasiliana (Szidat, 1928) Dubois, 1964 to Apharyngostrigea Ciurea, 1927 (Digenea: Strigeidae), a parasite of boat-billed heron (Cochlearius cochlearius) from the Neotropical region. Parasitol Int 86:102468. https://doi.org/10.1016/j.parint.2021.102468
López-Jiménez A, González-García M, Andrade-Gómez L, García-Varela M (2023) Phylogenetic analyses based on molecular and morphological data reveal a new species of Strigea Abildgaard, 1790 (Digenea: Strigeidae) and taxonomic changes in strigeids infecting Neotropical birds of prey. J Helminthol 97:E35. https://doi.org/10.1017/S0022149X23000196
Mollaret I, Jamieson BG, Adlard RD, Hugall A, Lecointre G, Chombard C, Justine JL (1997) Phylogenetic analysis of the Monogenea and their relationships with Digenea and Eucestoda inferred from 28S rDNA sequences. Mol Biochem Parasitol 90:433–438. https://doi.org/10.1016/s0166-6851(97)00176-x
Nielsen ÓK (2003) The impact of food availability on gyrfalcon (Falco rusticolus) diet and timing of breeding. In: Thompson DBA, Redpath SM, Fielding AH, Marquiss M, Galbraith CA (eds) Birds of prey in a changing environment. Scottish Natural Heritage, The Stationery Office Limited, Edinburg, 283–302
Nielsen ÓK (2011) Gyrfalcon population and reproduction in relation to rock ptarmigan numbers in Iceland. In: Watson T, Cade TJ, Fuller M, Hunt G, Potapov E (eds) Gyrfalcons and ptarmigan in a changing world. II. Boise: The Peregrine Fund 21–48
Nielsen ÓK, Cade TJ (1990a) Annual cycle of the gyrfalcon in Iceland. Natl Geogr Res 6:41–62
Nielsen ÓK, Cade TJ (1990b) Seasonal changes in food habits of gyrfalcons in NE-Iceland. Ornis Scand 21:202–211. https://doi.org/10.2307/3676780
Niewiadomska K (2002) Family Strigeidae Railliet, 1919. In: Gibson DI, Jones A, Bray RA (eds) Keys to the Trematoda. Volume 1. CAB International and Natural History Museum London, Wallingford, pp 231–241.
Odening K (1964) Drei neue Furcocercarien aus dem Raum Berlin. Monatsb Dtsch Akad Wiss Berlin 6:739–743
Odening K (1967) Die Lebenszyklen von Strigea falconispalumbi (Viborg), S. trigis (Schrank) und S. sphaerula (Rudolphi) (Trematoda, Strigeida) im Raum Berlin. Zool Jahrb Syst 94:1–67
Okulewicz J, Sitko J, Mellin M (1993) Trematodes parasitising in the white-tailed eagle (Haliaeetus albicilla L.). Wiad Parazytol 39:257–263 (in Polish)
Olson PD, Cribb TH, Tkach VV, Bray RA, Littlewood DT (2003) Phylogeny and classification of the Digenea (Platyhelminthes: Trematoda). Int J Parasitol 33:733–755. https://doi.org/10.1016/s0020-7519(03)00049-3
Parker GA, Ball MA, Chubb JC (2015) Evolution of complex life cycles in trophically transmitted helminths. I. Host incorporation and trophic ascent. J Evol Biol 28:267–291. https://doi.org/10.1111/jeb.12575
Patrelle C, Portier J, Jouet D, Delorme D, Ferté H (2015) Prevalence and intensity of Alaria alata (Goeze, 1792) in water frogs and brown frogs in natural conditions. Parasitol Res 114:4405–4412. https://doi.org/10.1007/s00436-015-4680-z
Pleijel F, Jondelius U, Norlinder E, Nygren A, Oxelman B, Schander C, Sundberg P, Thollesson M (2008) Phylogenies without roots? A plea for the use of vouchers in molecular phylogenetic studies. Mol Phylogenet Evol 48:369–371. https://doi.org/10.1016/j.ympev.2008.03.024
Poulin R (2007) Evolutionary ecology of parasites. Princeton University Press, Princeton
Pulis EE, Tkach VV, Newman RA (2011) Helminth parasites of the wood frog, Lithobates sylvaticus, in prairie pothole wetlands of the Northern Great Plains. Wetlands 31:675–685. https://doi.org/10.1007/s13157-011-0183-6
Pyrka E, Kanarek G, Zaleśny G, Hildebrand J (2021) Leeches as the intermediate host for strigeid trematodes: genetic diversity and taxonomy of the genera Australapatemon Sudarikov, 1959 and Cotylurus Szidat, 1928. Parasit Vectors 14:44. https://doi.org/10.1186/s13071-020-04538-9
Rochat EC, Blasco-Costa I, Scholz T, Unmack PJ (2020) High diversity of metazoan parasites in carp gudgeons (Eleotridae: Hypseleotris spp.) from Eastern Australia. J Helminthol 94:e146. https://doi.org/10.1017/S0022149X20000280
Ryang Y-S, Ahn Y-K, Yoon M-B (1991) Trematode infections in the small intestine of Egretta alba modesta in Kangwon-do. Korean J Parasitol 29:227–233 (in Korean)
Sanmartín ML, Alvarez F, Barreiro G, Leiro J (2004) Helminth fauna of falconiform and strigiform birds of prey in Galicia, Northwest Spain. Parasitol Res 92:255–263. https://doi.org/10.1007/s00436-003-1042-z
Santoro M, Tripepi M, Kinsella JM, Panebianco A, Mattiucci S (2010) Helminth infestation in birds of prey (Accipitriformes and Falconiformes) in Southern Italy. Vet J 186:119–122. https://doi.org/10.1016/j.tvjl.2009.07.001
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675. https://doi.org/10.1038/nmeth.2089
Sinsch U, Heneberg P, Těšínský M, Balczun C, Scheid P (2019) Helminth endoparasites of the smooth newt Lissotriton vulgaris: linking morphological identification and molecular data. J Helminthol 93:332–341. https://doi.org/10.1017/S0022149X18000184
Sitko J (1998) Trematodes of birds of prey (Falconiformes) in the Czech Republic. Helminthologia 35:131–146
Sitko J, Faltýnková A, Scholz T (2006) Checklist of the trematodes (Digenea) of birds of the Czech and Slovak Republics. Academia Press, Praha
Sokolov SG, Lebedeva DI, Khasanov FK, Gordeev II (2021) First description of the metacercaria of Nematostrigea serpens serpens (Nitzsch, 1819) (Trematoda, Strigeidae) and phylogenetic affiliation of Nematostrigea vietnamiensis Zhokhov & Mishina, 2011. Acta Parasitol 66:664–672. https://doi.org/10.1007/s11686-020-00275-2
Soldánová M, Georgieva S, Roháčová J, Knudsen R, Kuhn JA, Henriksen EH, Siwertsson A, Shaw JC, Kuris AM, Amundsen PA, Scholz T, Lafferty KD, Kostadinova A (2017) Molecular analyses reveal high species diversity of trematodes in a sub-Arctic lake. Int J Parasitol 47:327–345. https://doi.org/10.1016/j.ijpara.2016.12.008
Sudarikov V E (1959) Order Strigeidida (La Rue, 1926) Sudarikov, 1959. In: Skrjabin KI (ed) Trematodes of animals and man. Osnovy trematodologii, Izdatelstvo AN SSSR, Moscow, 16: 217–631. (in Russian)
Sudarikov VE (1984) Trematodes of the fauna of the USSR. Izdatelstvo Nauka, Moscow (in Russian)
Svinin AO, Bashinskiy IV, Litvinchuk SN, Ermakov OA, Ivanov AY, Neymark LA, Vedernikov AA, Osipov VV, Drobot GP, Dubois A (2020) Strigea robusta causes polydactyly and severe forms of Rostand’s anomaly P in water frogs. Parasit Vectors 13:381. https://doi.org/10.1186/s13071-020-04256-2
Svinin AO, Matushkina KA, Dedukh DV, Bashinskiy IV, Ermakov OA, Litvinchuk SN (2022) Strigea robusta (Digenea: Strigeidae) infection effects on the gonadal structure and limb malformation in toad early development. J Exp Zool A Ecol Integr Physiol 337:675–686. https://doi.org/10.1002/jez.2599
Svinin AO, Chikhlyaev IV, Bashinskiy IW, Osipov VV, Neymark LA, Ivanov AY, Stoyko TG, Chernigova PI, Ibrogimova PK, Litvinchuk SN, Ermakov OA (2023) Diversity of trematodes from the amphibian anomaly P hotspot: role of planorbid snails. PLoS ONE 18:e0281740. https://doi.org/10.1371/journal.pone.0281740
Syrota Y, Lisitsyna O, Greben O, Kornyushin V, du Preez L (2021) The helminth infracommunities in three species of herons (Aves: Ardeidae) from Ukraine. Parasitol Int 85:102442. https://doi.org/10.1016/j.parint.2021.102442
Tamura K, Stecher G, Kumar S (2021) MEGA11: molecular evolutionary genetics analysis version 11. Mol Biol Evol 38:3022–3027. https://doi.org/10.1093/molbev/msab120
Tkach VV (2001) Molecular phylogeny of the suborder Plagiorchiata and its position in the system of Digenea. In: Littlewood DTJ, Bray RA (eds) Interrelationships of Platyhelminthes. Taylor & Francis London, pp 186–193
Vermaak A, Smit NJ, Kudlai O (2021) Molecular and morphological characterisation of the metacercariae of two species of Cardiocephaloides (Digenea: Strigeidae) infecting endemic South African klipfish (Perciformes: Clinidae). Folia Parasitol 68:007. https://doi.org/10.14411/fp.2021.007
Wilson JR (1981) The migration of High Arctic shorebirds through Iceland. Bird Study 28:21–32. https://doi.org/10.1080/00063658109476695
Zafra R, Martínez-Moreno FJ, Rufino-Moya PJ, Gutiérrez PN, Martínez-Cruz S, Buffoni L, Martínez-Moreno A, Acosta I (2022) Study of the helminth fauna in eagle owl (Bubo bubo) in the south of Spain. Int J Parasitol Par Wildl 18:260–265. https://doi.org/10.1016/j.ijppaw.2022.06.011
Acknowledgements
We are grateful to Blanka Škoríková for scanning of the drawing.
Funding
Open access publishing supported by the National Technical Library in Prague. The present study was funded by the research fund from the University of Iceland.
Author information
Authors and Affiliations
Contributions
Conceptualisation: AF, DJ, OKN and KS. Developing methods and data analysis: AF and DJ. Preparation of figures and tables: AF, DJ and OKN. Conducting the research, data interpretation and writing: AF, DJ, OKN and KS.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
All authors have read and agreed to the published version of the manuscript.
Competing interests
The authors declare no competing interests.
Additional information
Section Editor: David Bruce Conn
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Faltýnková, A., Jouet, D., Nielsen, Ó.K. et al. First species record of Strigea falconis Szidat, 1928 (Trematoda, Strigeidae) from gyrfalcon Falco rusticolus in Iceland—pros and cons of a complex life cycle. Parasitol Res 123, 147 (2024). https://doi.org/10.1007/s00436-024-08161-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00436-024-08161-w