Abstract
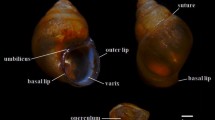
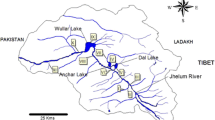
Indoplanorbis exustus, a freshwater pulmonate snail, is widely distributed in tropical and subtropical zones and plays a significant role as an intermediate host for trematode parasites. Various genetic markers have been used for species identification and phylogenetic studies of this snail. However, there are limited studies about their molecular genetics based on nuclear ribosomal DNA (rDNA) genes. A genetic analysis of I. exustus in Thailand was conducted based on the nuclear 18S rDNA (339 bp) and 28S rDNA (1036 bp) genes. Indoplanorbis snails were collected from 29 localities in 21 provinces covering six regions of Thailand. Nucleotide sequences from 44 snails together with sequences from the GenBank database were examined for phylogenetic relationships and genetic diversity. All sequences of the selected nucleotide regions exhibited a high level of similarity (99%) to the sequences of I. exustus in the GenBank database. The maximum likelihood tree based on the 18S and 28S rDNA fragment sequences of I. exustus in Thailand revealed only one group with clear separation from another genus in the family Planorbidae. The I. exustus 28S rDNA sequences showed intraspecific genetic divergence ranging from 0 to 0.78% and were classified into 8 different haplotypes. Conversely, the 18S rDNA data showed lower variation than the 28S rDNA data and revealed a single haplotype and intraspecific distances of zero among all sampled individuals. The haplotype network of 28S rDNA sequences of I. exustus in Thailand revealed six unique haplotypes and two haplotypes shared by at least two regions. Overall, both markers were successful in the identification of I. exustus. However, these markers, particularly the 18S rDNA, may not be suitable for genetic analysis within the species, particularly for population genetic studies, due to their limited variation as seen in this study. In summary, this study not only enhances understanding of genetic variation in I. exustus but is also useful for the selection of molecular markers in future genetic research.



Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the article.
References
Andrus PS, Stothard JR, Kabatereine NB, Wade CM (2023) Comparing shell size and shape with canonical variate analysis of sympatric Biomphalaria species within Lake Albert and Lake Victoria, Uganda. Zool J Linn Soc:zlad052. https://doi.org/10.1093/zoolinnean/zlad052
Ardpairin J, Dumidae A, Subkrasae C, Nateeworanart S, Thanwisai A, Vitta A (2022) Preliminary survey of larval trematodes in freshwater snails of Phitsanulok Province in lower Northern Thailand. Iran J Parasitol 17:268–276. https://doi.org/10.18502/ijpa.v17i2.9545
AVMA (American Veterinary Medical Association) (2020) AVMA guidelines for the euthanasia of animals: 2020 editions. Available online: https://www.avma.org/KB/Policies/Documents/euthanasia.pdf. Accessed 20 Jul 2020
Bandelt HJ, Forster P, Rohl A (1999) Median-joining networks for inferring intraspecific phylogenies. Mol Biol Evol 16(1):37–48. https://doi.org/10.1093/oxfordjournals.molbev.a026036
Bargues MD, Mas-Coma S (1997) Phylogenetic analysis of Lymnaeid snails based on 18S rDNA sequences. Mol Biol Evol 14(5):569–577. https://doi.org/10.1093/oxfordjournals.molbev.a025794
Bawm S, Khaing NHE, Win SY, Thein SS, Khaing Y, Thaw YN, Soe NC, Chel HM, Hmoon MM, Hayashi N, Htun LL, Katakura K, Nonaka N, Nakao R (2022) Morphological and molecular identification of trematode cercariae related with humans and animal health in freshwater snails from a lake and a dam in Myanmar. Parasitol Res 121(2):653–665. https://doi.org/10.1007/s00436-022-07428-4
Brandt RAM (1974) The non-marine aquatic Mollusca of Thailand. Arch Molluskenkd 105:1–423
Bunchom N, Saijuntha W, Vaisusuk K, Pilap W, Suksavate W, Suganuma N, Agatsuma T, Petney TN, Tantrawatpan C (2021) Genetic variation of a freshwater snail Hydrobioides nassa (Gastropoda: Bithyniidae) in Thailand examined by mitochondrial DNA sequences. Hydrobiologia 848:2965–2976. https://doi.org/10.1007/s10750-019-04013-2
Chai JY (2019) Human intestinal flukes from discovery to treatment and control. Springer Nature B.V, Dordrecht, Netherlands
Choudhary K, Verma AK, Swaroop S, Agrawal N (2015) A review on the molecular characterization of digenean parasites using molecular markers with special reference to ITS region. Helminthologia 52:167–187. https://doi.org/10.1515/helmin-2015-0031
Cowie RH, Dillon RT, Robinson DG, Smith JW (2009) Alien non-marine snails and slugs of priority quarantine importance in the United States: a preliminary risk assessment. Am Malacol Bull 27:113–132. https://doi.org/10.4003/006.027.0210
Demiaszkiewicz AW (2014) Migrations and the introduction of wild ruminants as a source of parasite exchange and emergence of new parasitoses. Ann Parasitol 60(1):25–30
Devkota R, Brant SV, Loker ES (2015) The Schistosoma indicum species group in Nepal: presence of a new lineage of schistosome and use of the Indoplanorbis exustus species complex of snail hosts. Int J Parasitol 45(13):857–870. https://doi.org/10.1016/j.ijpara.2015.07.008
Dumidae A, Janthu P, Subkrasae C, Polseela R, Mangkit B, Thanwisai A, Vitta A (2021) Population genetics analysis of a Pomacea snail (Gastropoda: Ampullariidae) in Thailand and its low infection by Angiostrongylus cantonensis. Zool Stud 60:31. https://doi.org/10.6620/ZS.2021.60-31
Dung BT, Doanh PN, The DT, Loan HT, Losson B, Caron Y (2013) Morphological and molecular characterization of lymnaeid snails and their potential role in transmission of Fasciola spp. in Vietnam. Korean J Parasitol 51:657–662. https://doi.org/10.3347/kjp.2013.51.6.657
Frandsen F (1983) A field guide to freshwater snails in countries of the WHO Eastern Mediterranean region. Danish Bilharziasis Laboratory, Copenhagen, Denmark
Gauffre-Autelin P, von Rintelen T, Stelbrink B, Albrecht C (2017) Recent range expansion of an intermediate host for animal schistosome parasites in the indo-Australian Archipelago: phylogeography of the freshwater gastropod Indoplanorbis exustus in south and Southeast Asia. Parasit Vectors 10(1):126. https://doi.org/10.1186/s13071-017-2043-6
Gow JL, Noble LR, Rollinson D, Tchuenté LAT, Jones CS (2007) Contrasting temporal dynamics and spatial patterns of population genetic structure correlate with differences in demography and habitat between two closely related African freshwater snails. Biol J Linn Soc 90:747–760. https://doi.org/10.1111/j.1095-8312.2007.00771.x
Habib MR, Lv S, Rollinson D, Zhou X (2021) Invasion and dispersal of Biomphalaria species: increased vigilance needed to prevent the introduction and spread of schistosomiasis. Front Med 8:614797. https://doi.org/10.3389/fmed.2021.614797
Hwang UW, Kim W (1999) General properties and phylogenetic utilities of nuclear ribosomal DNA and mitochondrial DNA commonly used in molecular systematics. Korean J Parasitol 37(4):215–228. https://doi.org/10.3347/kjp.1999.37.4.215
Ikeda T, Iwasaki K, Suzuki T, Wong LJ, Pagad S (2021) Global register of Introduced and Invasive Species- Japan. Version 1.2. Invasive Species Specialist Group ISSG. Checklist dataset. https://doi.org/10.15468/nt2yla
Jørgensen A, Madsen H, Nalugwa A, Nyakaana S, Rollinson D, Stothard JR, Kristensen TK (2011) A molecular phylogenetic analysis of Bulinus (Gastropoda: Planorbidae) with conserved nuclear genes. Zool Scr 40(2):126–136. https://doi.org/10.1111/j.1463-6409.2010.00458.x
Kelehear C, Brown GP, Shine R (2013) Invasive parasites in multiple invasive hosts: the arrival of a new host revives a stalled prior parasite invasion. Oikos 122:1317–1324. https://doi.org/10.1111/j.1600-0706.2013.00292.x
Kimura M (1980) A simple method for estimating evolutionary rate of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16(2):111–120. https://doi.org/10.1007/BF01731581
Krailas D, Namchote S, Komsuwan J, Wongpim T, Apiraksena K, Glaubrecht M, Sonthiporn P, Sansawang C, Suwanrit S (2022) Cercarial dermatitis outbreak caused by ruminant parasite with intermediate snail host: schistosome in Chana. South Thailand Evol Syst 6(2):151–173. https://doi.org/10.3897/evolsyst.6.87670
Kristensen TK, Ogunnowo O (1987) Indoplanorbis exustus (Deshaies, 1834), a freshwater snail new for Africa, found in Nigeria (Pulmonata: Planorbidae). J Molluscan Stud 53:245–246. https://doi.org/10.1093/mollus/53.2.245
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33(7):1870–1874. https://doi.org/10.1093/molbev/msw054
Leigh JW, Bryant D (2015) Popart: full-feature software for haplotype network construction. Methods Ecol Evol 6(9):1110–1116. https://doi.org/10.1111/2041-210X.12410
Librado P, Rozas J (2009) DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25(11):1451–1452. https://doi.org/10.1093/bioinformatics/btp187
Liu L, Mondal MM, Idris MA, Lokman HS, Rajapakse PJ, Satrija F, Diaz JL, Upatham ES, Attwood SW (2010) The phylogeography of Indoplanorbis exustus (Gastropoda: Planorbidae) in Asia. Parasit Vectors 3:57. https://doi.org/10.1186/1756-3305-3-57
Lu XT, Gu QY, Limpanont Y, Song LG, Wu ZD, Okanurak K, Lv ZY (2018) Snail-borne parasitic diseases: an update on global epidemiological distribution, transmission interruption and control methods. Infect Dis Poverty 7:28. https://doi.org/10.1186/s40249-018-0414-7
Lv S, Zhang Y, Liu HX, Hu L, Yang K, Steinmann P, Chen Z, Wang LY, Utzinger J, Zhou XN (2009) Invasive snails and an emerging infectious disease: results from the first national survey on Angiostrongylus cantonensis in China. PLoS Negl Trop Dis 3:e368. https://doi.org/10.1371/journal.pntd.0000368
Maes T, De Corte Z, Vangestel C, Virgilio M, Smitz N, Djuikwo-Teukeng FF, Papadaki MI, Huyse T (2022) Large-scale and small-scale population genetic structure of the medically important gastropod species Bulinus truncatus (Gastropoda, Heterobranchia). Parasit Vectors 15:328. https://doi.org/10.1186/s13071-022-05445-x
Matsuda T, Morishita M, Hinomoto N, Gotoh T (2014) Phylogenetic analysis of the spider mite sub-family Tetranychinae (Acari: Tetranychidae) based on the mitochondrial COI gene and the 18S and the 59 end of the 28S rRNA genes indicates that several genera are polyphyletic. PLoS One 9(10):e108672. https://doi.org/10.1371/journal.pone.0108672
Matumba TG, Oliver J, Barker NP, McQuaid CD, Teske PR (2020) Intraspecific mitochondrial gene variation can be as low as that of nuclear rRNA. F1000Res 9:339. https://doi.org/10.12688/f1000research.23635.2
Meyer JH, Fourdrigniez M, Soubeyran Y, Pagad S (2021) Global Register of Introduced and Invasive Species-French Polynesia. Version 1.2. Invasive Species Specialist Group ISSG. Checklist dataset. https://doi.org/10.15468/bi2dvk
Morgan JA, DeJong RJ, Jung Y, Khallaayoune K, Kock S, Mkoji GM, Loker ES (2002) A phylogeny of planorbid snails, with implications for the evolution of Schistosoma parasites. Mol Phylogenet Evol 25(3):477–488. https://doi.org/10.1016/s1055-7903(02)00280-4
Mouahid G, Clerissi C, Allienne JF, Chaparro C, Al Yafae S, Mintsa Nguema R, Ibikounlé M, Moné H (2018) The phylogeny of the genus Indoplanorbis (Gastropoda, Planorbidae) from Africa and the French West Indies. Zool Scr 47:558–564. https://doi.org/10.1111/zsc.12297
Mouchet F, Rey JL, Cunin P (1987) Decouverte d’Indoplanorbis exustus (Planorbidae; Bulininae) a Yamoussokro, Cote d’Ivoire. Bull Soc Pathol Exot Filiales 80:811–812
Neubauer TA, Mandic O, Harzhauser M, Jovanović G (2017) The discovery of Bulinus (Pulmonata: Planorbidae) in a Miocene palaeolake in the Balkan Peninsula. J Molluscan Stud 83(3):295–303. https://doi.org/10.1093/mollus/eyx015
Nithiuthai S, Anantaphruti MT, Waikagul J, Gajadhar A (2004) Waterborne zoonotic helminthiases. Vet Parasitol 126:167–193. https://doi.org/10.1016/j.vetpar.2004.09.018
Nyström V, Angerbjörn A, Dalén L (2006) Genetic consequences of a demographic bottleneck in the Scandinavian arctic fox. Oikos 114:84–94. https://doi.org/10.1111/j.2006.0030-1299.14701.x
Pagad S (2020) Protected areas - Global Register of Introduced and Invasive Species - Socotra Archipelago, Yemen. Invasive Species Specialist Group ISSG. Checklist dataset. https://doi.org/10.15468/n9mssm
Parashar BD, Rao KM (1982) Effect of desiccation on freshwater snail, Indoplanorbis exustus, intermediate host of schistosomiasis. Jpn J Med Sci Biol 35:243–247. https://doi.org/10.7883/yoken1952.35.243
Patwardhan A, Ray S, Roy A (2014) Molecular markers in phylogenetic studies-a review. J Phylogen Evolution Biol 2:2. https://doi.org/10.4172/2329-9002.1000131
Pawlowski J, Audic S, Adl S, Bass D, Belbahri L, Berney C, Bowser SS, Cepicka I, Decelle J, Dunthorn M, Fiore-Donno AM, Gile GH, Holzmann M, Jahn R, Jirků M, Keeling PJ, Kostka M, Kudryavtsev A, Lara E et al (2012) CBOL Protist Working Group: barcoding eukaryotic richness beyond the animal, plant, and fungal Kingdoms. PLoS Biol 10(11):e1001419. https://doi.org/10.1371/journal.pbio.1001419
Pointier JP, David P, Jarne P (2005) Biological invasions: the case of planorbid snails. J Helminthol 79:249–256
Ryland JS, Bishop JDD (1990) Prevalence of cross-fertilisation in the hermaphroditic compound ascidian Diplosoma listerianum. Mar Ecol Prog Ser 61:125–132. https://doi.org/10.3354/meps061125
Saijuntha W, Tantrawatpan C, Agatsuma T, Rajapakse R, Karunathilake KJK, Pilap W, Tawong W, Petney TN, Andrews RH (2021) Phylogeographic genetic variation of Indoplanorbis exustus (Deshayes, 1834) (Gastropoda: Planorbidae) in South and Southeast Asia. One Health 12:100211. https://doi.org/10.1016/j.onehlt.2021.100211
Shirk RY, Hamrick JL, Zhang C, Qiang S (2014) Patterns of genetic diversity reveal multiple introductions and recurrent founder effects during range expansion in invasive populations of Geranium carolinianum (Geraniaceae). Heredity 112:497–507. https://doi.org/10.1038/hdy.2013.132
Sil M, Mahveen J, Roy A, Karanth KP, Aravind NA (2022) Insight into the evolutionary history of Indoplanorbis exustus (Bulinidae: Gastropoda) at the scale of population and species. Biol J Linn Soc Lond 137(1):68–84. https://doi.org/10.1093/biolinnean/blac062
Stothard JR, Bremond P, Andriamaro L, Loxton NJ, Sellin B, Sellin E, Rollinson D (2000) Molecular characterization of the freshwater snail Lymnaea natalensis (Gastropoda: Lymnaeidae) on Madagascar with an observation of an unusual polymorphism in ribosomal small subunit genes. J Zool 252:303–315. https://doi.org/10.1111/j.1469-7998.2000.tb00625.x
Tamura K (1992) Estimation of the number of nucleotide substitutions when there are strong transition-transversion and G + C-content biases. 9(4):678-687. https://doi.org/10.1093/oxfordjournals.molbev.a040752
Taraschewski H (2006) Hosts and parasites as aliens. J Helminthol 80:99–128. https://doi.org/10.1079/joh2006364
Van Bocxlaer B, Strong EE, Richter R, Stelbrink B, Von Rintelen T (2018) Anatomical and genetic data reveal that Rivularia Heude, 1890 belongs to Viviparinae (Gastropoda: Viviparidae). Zool J Linnean Soc 182(1):1–23. https://doi.org/10.1093/zoolinnean/zlx014
van Leeuwen CHA, van der Velde G (2012) Prerequisites for flying snails: external transport potential of aquatic snails by waterbirds. Freshw Sci 31:963–972. https://doi.org/10.1899/12-023.1
Vences M, Thomas M, Bonett RM, Chiari Y, Vietes DR (2005) Deciphering amphibian diversity through DNA barcoding: chances and challenges. Philos Trans R Soc Lond Ser B Biol Sci 360(1462):1859–1868. https://doi.org/10.1098/rstb.2005.1717
Weber DS, Stewart BS, Lehman N (2004) Genetic consequences of a severe population bottleneck in the Guadalupe fur seal (Arctocephalus townsendi). J Hered 95:144–153. https://doi.org/10.1093/jhered/esh018
Wiroonpan P, Chontananarth T, Purivirojkul W (2021) Cercarial trematodes in freshwater snails from Bangkok, Thailand: prevalence, morphological and molecular studies and human parasite perspective. Parasitology 148(3):366–383. https://doi.org/10.1017/S0031182020002073
Won EJ, Lee YJ, Kim MJ, Chai JY, Na BK, Sohn WM (2020) Morphological and molecular characteristics of Clinostomid Metacercariae from Korea and Myanmar. Korean J Parasitol 58(6):635–645. https://doi.org/10.3347/kjp.2020.58.6.635
Zein-Eddine R, Djuikwo-Teukeng FF, Al-Jawhari M, Senghor B, Huyse T, Dreyfuss G (2014) Phylogeny of seven Bulinus species originating from endemic areas in three African countries, in relation to the human blood fluke Schistosoma haematobium. BMC Evol Biol 14:271. https://doi.org/10.1186/s12862-014-0271-3
Zein-Eddine R, Djuikwo-Teukeng FF, Dar Y, Dreyfuss G, Van den Broeck F (2017) Population genetics of the Schistosoma snail host Bulinus truncatus in Egypt. Acta Trop 172:36–43. https://doi.org/10.1016/j.actatropica.2017.04.002
Acknowledgements
We would like to thank the Department of Microbiology and Parasitology, Faculty of Medical Science, Naresuan University, for providing the necessary support and access to facilities.
Funding
This study was supported by Naresuan University (NU) and the National Science, Research and Innovation Fund (NSRF), Thailand (grant no. R2566B046). This funding sponsor had no role in the design of the study.
Author information
Authors and Affiliations
Contributions
AD performed experiments, analyzed data, and wrote the paper. CS performed experiments and wrote the paper. JA performed experiments and wrote the paper. SP performed experiments and wrote the paper. CH performed experiments and wrote the paper. AT provided materials and wrote the paper. AV performed experiments, analyzed data, provided materials, and wrote the paper.
Corresponding author
Ethics declarations
Ethical approval
The experiments involving invertebrate animals (snails) were approved from the Center for Animal Research at Naresuan University (Project Ethics No: NU-AQ640803).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Handling Editor: Julia Walochnik
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dumidae, A., Subkrasae, C., Ardpairin, J. et al. Genetic variation of the freshwater snail Indoplanorbis exustus (Gastropoda: Planorbidae) in Thailand, inferred from 18S and 28S rDNA sequences. Parasitol Res 123, 93 (2024). https://doi.org/10.1007/s00436-024-08120-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00436-024-08120-5