Abstract
Snails are fascinating molluscs with unique morphological and physiological adaptive features to cope with various environments. They have traditionally been utilized as food and feed sources in many regions of the world. The future exploitation of alternative nutrient sources, like snails, is likely to increase further. Snails, however, also serve as an intermediate host for several zoonotic parasites. A category of parasitic infections, known as snail-transmitted parasitic diseases (STPDs), is harmful to humans and animals and is mainly driven by various trematodes, cestodes, and nematodes. The environment plays a crucial role in transmitting these parasites, as suitable habitats and conditions can facilitate their growth and proliferation in snails. In light of diverse environmental settings and biologically categorized snail species, this review evaluates the dynamics of significant STPDs of zoological importance. Additionally, possible diagnostic approaches for the prevention of STPDs are highlighted. One-health measures must be considered when employing snails as an alternative food or feed source to ensure the safety of snail-based products and prevent any adverse effects on humans, animals, and the environment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Molluscs, such as snails, slugs, and mussels, are a distinct group of spineless animals and play vital roles in the ecosystem (Fortunato 2015; Rosenberg 2014; Vaughn and Hakenkamp 2001). The snail species have been used as a food source in different regions worldwide (Hill et al. 2015; Ngenwi et al. 2010). Many production animals, such as poultry and fish, naturally feed on snails (Amobi et al. 2019; Pertiwi and Saputri 2020). In light of finding novel animal feed ingredients to ensure future food security, snails have been recognized as a potential alternative feed source for production animals that can undergo commercial-scale production (Adeyeye et al. 2020; Munonye and Moses 2019). Snails are considered a valuable supplement to conventional feedstuffs because they contain high levels of protein, calcium, and other essential nutrients (Wright et al. 2018) (Table 1). In addition, snail farming can be operated under relatively low-cost animal husbandry practices that can be profitable in areas where snails are abundant (Mvodo Meyo et al. 2021). Snails have several applications in multiple sectors, such as agriculture, medicine, the food industry, cosmetics, biotechnology, and environmental monitoring, due to their unique biological characteristics and their ability to adapt to changing environments (Akan et al. 2020; Cilia and Fratini 2018; Wang et al. 2020; Yuan et al. 2019) (Table 2). While having a valuable component for various applications, it is noteworthy that they act as agents for serious parasitic disease transmission.
Snail-transmitted parasitic diseases (STPDs) are a significant public health concern in many parts of the world, particularly in tropical and sub-tropical regions (Lu et al. 2018). Snails serve as intermediate hosts in the transmission of various parasites, primarily three classes of helminths: trematodes, nematodes, and cestodes (Chakraborty and Joy 2020; Lopatek et al. 2022). Snails act as single intermediate hosts for certain parasites, such as Angiostrongylus and Schistosoma mansoni, whereas in others, e.g., Clonorchis sinensis, Paragonimus westermani, Fasciolopsis buski, and Fasciola hepatica, snails can be only first intermediate hosts and then the parasitic larva invade other second intermediate hosts like fish, crab, or encysted in the aquatic vegetations for more asexual reproductions (Ansari et al. 2021; Mehmood et al. 2017). After invading snails, the parasites go through some developmental stages. For example, in the case of most trematodes, miracidium penetrates the snails, develops further into sporocyst and redia, and then releases cercaria from the snails (Galaktionov and Dobrovolskij 2013). Then, the cercaria encysts and develops into metacercaria that can enter into livestock and human bodies through the consumption of feed, fodder, food, and water, or cercaria in the case of schistosomes can directly penetrate skins through contaminated water bodies (Lv et al. 2018). It is important to note that not all snail species are suitable for use as animal feed, and it is necessary to choose snail species that are safe, nutritious, and affordable for the intended animal food or feed applications. In this context, understanding the biology of snails and their interactions with parasites and pathogens is crucial for ensuring their safe and sustainable use within the food and feed sector (Colgan 2020; Imathiu 2020; Pissia et al. 2021; Ponder et al. 2019a).
The biology of snails in relation to parasitic disease transmission
Snails are essential hosts for many parasites, including those that cause significant morbidity and mortality in humans and animals. Understanding the physiological processes of snails is crucial to getting insights into the complex interactions between parasites and snail hosts and in developing effective prevention and control measures and management of snail-transmitted parasitic diseases (Lu et al. 2018; Malek 2018; Ponder et al. 2019b).
Characteristics of snail species
Snails are invertebrates belonging to the phylum, Mollusca, which is the second-largest animal group and is quite diverse (Ponder et al. 2019b). Under this phylum, gastropods are the largest, highly diversified class of molluscs consisting of snails and slugs present in saltwater, freshwater, and land. They are characterized by a single spiral shell and a foot for movement (Fig. 1) (Bouchet et al. 2017). Snails are further categorized into multiple orders based on certain morphological features and habitats, each containing several families (Table 3). The families including Achatinidae, Bradybaenidae, Camaenidae, Cyclophoridae, Helicarionidae, Subulinidae, and Succineidae belong to land snails, whereas the freshwater snails include Ampullariidae, Ancyclidae, Acroloxidae, Bithyniidae, Physidae, Planorbidae, Lymnaeidae, Viviparidae, Thiaridae, and Pomatiopsidae (Bouchet et al. 2017; Saadi et al. 2020; Soldatenko and Petrov 2019). Both land and water snail species are susceptible to being infected with at least one parasite during the life cycle from their surrounding environments, and the parasite can surpass the snails’ structural and biochemical barriers. However, the nature or species of the parasite infecting them depends on the body of snails and their physiological status, along with feeding and habitat diversity.
Three different snail species were collected from Rampur, Chitwan, Nepal (latitude: 27.658734, longitude: 84.35105; 4th September 2022; 28.7 °C). The morphological identification and comparison of published databases (Glöer and Bössneck 2013; Raheem et al. 2010) may suggest the following species: a Lissachatina spp. (Achatinidae), b Gyraulus spp. (Planorbidae), and c Radix spp. (Lymnaeidae)
Anatomical and physiological features of snails
The visceral mass is the main body cavity of the snail that contains digestive, respiratory, and reproductive organs (Fig. 2). The viscera are protected and supported by a species-specific shell containing calcium carbonate (Larsson et al. 2020; Parveen et al. 2020). The one pair of snail’s tentacles helps in sensory perception during the retraction of the head region (Brenzinger et al. 2021). The penetration of the first larval stage (miracidium) of Schistosoma species occurs from the outer surface of the head, foot, and tentacles, near the buccal mass esophagus, whereas the Fasciola species penetrates the mantle collar and anterior wall of the pulmonary sac (Malek 2018). The anatomical features and physiological activities of snails are crucial for the development, proliferation, and spread of pathogens in the body, as discussed below.
The major physiological systems of the snail. Nervous system in blue, circulatory and respiratory systems in pink, excretory system in green, reproductive system in yellow, digestive system in black, and digestive glands in red color. Adapted and modified from published literature (Ponder et al. 2019b)
The digestive system of snails
Parasites can induce physiological changes and pathology in their hosts, affecting metabolism, immune response, growth, development, and (Marcogliese). In natural environments and also in experimental investigations, fecundity
Parasites can induce systemic physiological changes and pathological lesions in the different organs of snails. Snails with different natures or feeding habits and life stages are prone to infection with a wide range of pathogens via the oral route through contaminated feeding materials (Marcogliese and Pietrock 2011). Land snails possess an esophagus, and the crop for temporary storage and digestion of ingesta, but freshwater snails have a gizzard, which is a strong muscular stomach that helps in the final digestion of ingesta (Fig. 3) (Ponder et al. 2019a; Pouil et al. 2020). Herbivores possess an oral cavity that employs amylase to process food material and eliminate harmful pathogens, allowing both to pass along with ingesta (Escobar-Correas et al. 2019; GHOSE 1961). Conversely, carnivores lack any amylase activity in their oral cavity. Nutrients derived from the digestion of food, such as amino acids, glucose, and fatty acids, can be utilized by the larval stage of parasites like redia and sporocysts (Malek 2018). Any interferences in the digestion of food, absorption, and metabolism of nutrients lead to the loss of tolerance by snails against environmental stress (Ponder et al. 2019b). Once nutrients are absorbed from the gastrointestinal tract of snails, they enter the circulatory system of snails.
The digestive system contains organs and digestive glands (in red). Adapted and modified from published literature (Ponder et al. 2019b)
The circulatory and respiratory systems of snails
Snails have an open circulatory system with a heart bag serving as the central organ for the pumping of haemolymph (blood) into the body cavity of snails (Fig. 4). Within the cavity, haemolymph bathes various organs and tissues (Ponder et al. 2019a). However, the majority of the larval stages of trematodes, like daughter sporocysts and cercariae, migrate posteriorly or to the distal end opposite to the flow of direction of the haemolymph infecting ovotestis and albumen glands (Malek 2018). Such trematode infections in the respiratory organs of snails lead to inflammatory reactions, congestion, and spread to various organs (Blair 2022; Malek 2018). Hence, the visceral organs of snails are prone to parasitic lodgement, infestation, and inflammatory responses. Thus, snails have adapted their lifestyle to efficiently fulfill their growth and reproductive functions in response to parasitic infections.
Representing different parts of the excretory in green, circulatory in pink, and respiratory systems in pink of the snail. Adapted and modified from published literature (Ponder et al. 2019b)
The reproductive system of snails
Most snail species are hermaphrodites, as they contain both male and female reproductive organs (Fig. 5). However, cross-fertilization occurs in snails to fertilize their eggs. After fertilization, snails lay eggs under soil or leaves, and the eggs hatch into the small juvenile stage (Fig. 6) (Ponder et al. 2019b). The reproductive system of snails does not appear to have a direct role in the growth and proliferation of infecting parasites, unlike digestive or circulatory systems. The duration of life stages and the development of body structures of snails can vary depending on environmental factors and their nutrient supply (Erkano 2021). As discussed below, the overall reproductive performance and weight gain of snails are affected by the interplay of biotic and abiotic factors surrounding them.
Representing the reproductive system of a snail: male in yellow and female in orange. Adapted and modified from published literature (Ponder et al. 2019b)
Environmental factors affecting snails’ growth and parasitic disease transmission
Environmental conditions lead to changes in both the physiology and morphology of snails. External abiotic factors, such as temperature, salinity, oxygen, and pH, influence the physiological processes of snails (Bula et al. 2017; Donham et al. 2022; Saallah et al. 2020).
Temperature
Temperature plays a vital role in the physiochemical activities of snails. For example, in Pila globosa, higher lipid peroxidation (51.8%) and antioxidative activity (26.4%) were reported in the summer than in other seasons (Pati et al. 2023). During environmental stressful situations, heat shock proteins interact with chaperone-assisted proteins to maintain cellular homeostasis and the survival of land snails, Helix pomatia Linnaeus (Idczak-Figiel et al. 2023). During summer, higher physical activities of snails lead to their migration, breeding, and growth. In the freshwater snails (Bulinus glolosus), at the temperatures of 15.5 °C and 36.0 °C, snails were unable to produce eggs, but egg mass output was found to be the highest at 25.8 °C (Kalinda et al. 2017). The optimum temperature for snail hosts and their infecting parasites is around 27 °C. The relative abundance of snail parasites can decrease by 8 to 17%, with the rise in temperature above the optimum level by 1 to 2 °C, respectively (Kalinda et al. 2018). These studies indicate that both snails and infecting parasites require a unique temperature range for their interaction, proliferation, and survival (Nguyen et al. 2020).
Oxygen, salinity, and pH
The abundance of snail species in certain habitats is also determined by their capacity to utilize oxygen either from the air or dissolved in surrounding environments, and this factor is more critical for aquatic snail species. For example, a higher abundance of Bulinus globosus (31.7%) followed by Lymnaea natalensis (21.6%), Lymnaea truncatula (15.1%), and Biomphalaria pfeifferi (14.6%) in the Ethiopian Rift Valley region was associated with variations in the oxygen concentrations of water bodies and the oxygen utilizing abilities of snails (Olkeba et al. 2020). Embryonic survival and development of snails were negatively affected by increased environmental salinity even though embryos reside inside their protective egg mass (Barrios-Figueroa and Urbina 2023). The optimum pH for snails is 7.8 to 8.5. Thus, a rise in acidity can lead to disturbances in the enzymatic activity of snails, and their shells may become more transparent and thinner and crack (O'Sullivan et al. 2011).
There is a lack of comprehensive research on the cellular signaling pathways of snails in response to variations in environmental factors. Understanding the impacts and interactions of various environmental parameters on the growth and physiology of snails and infecting parasites is critical while aiming to exploit snails for various commercial applications.
Snail-transmitted parasitic diseases in animals and humans
STPDs are a significant problem in veterinary and public health, causing heavy economic losses. Based on the class of parasites of snails, STPDs can be divided into three groups: trematode diseases (by infection of flukes such as liver flukes, intestinal flukes, blood flukes, and lung flukes), nematode diseases (rat lungworm infection), and cestode diseases (Davainea infection). The majority of STPDs are responsible for clinical manifestations and lesions in livestock and humans and belong to digenetic trematodes, metastrongyle nematodes, and a few tapeworms (Table 4) (Lu et al. 2018). As discussed in the following, the dietary habits of humans, animals, and birds can influence the prevalence of snail-associated parasitic diseases.
Fasciolosis
The two species of parasites that cause fasciolosis in humans and animals are Fasciola hepatica and Fasciola gigantica, commonly known as liver flukes. Fasciolosis due to F. hepatica is widely distributed worldwide, whereas F. gigantica is found more in Africa, the Middle East, and Asia, with limited distribution of suitable intermediate lymnaeid snail hosts (Mehmood et al. 2017). In the global scenario, fasciolosis has susceptibility to more than 180 million people, whereas more than 35–72 million individuals are considered to be infected with liver fluke (Sabourin et al. 2018). Fasciolosis causes significant economic losses estimated annually at over US$3 billion worldwide (Sabourin et al. 2018).
Liver flukes have a complex life cycle. They involve humans or animals as the definitive host and snails as the intermediate host belonging to the Lymnaeidae family and are responsible for harboring the intermediate stages of liver fluke (miracidium, sporocyst, redia, and cercaria). The ingestion of encysted infective metacercaria stage found on aquatic vegetation is the means of transmission to the final host (Ansari et al. 2021; Caldeira et al. 2016; Hu et al. 2021; Lalor et al. 2021). Preventive measures may include grazing restriction in the areas where snails are abundant or sunlight treatment of grasses from such areas. To prevent human infections, freshwater vegetables should not be consumed raw, because water sources such as artificial irrigation channels are known to harbor snails (Stein 2018). The use of molluscicides or biological means (duck or poultry, plants) can prevent the snail population. One-health approach with multidisciplinary collaborative activities like vaccination of livestock, proper management of freshwater bodies, and improved diagnosis and treatment approaches are crucial steps in mitigating the zoonotic fascioliasis risk (Ali et al. 2017; Mas-Coma et al. 2023). Using triclabendazole to treat human fascioliasis highlights the need to focus on a one-health approach to animal reservoirs for better control (Bargues et al. 2020). Moreover, collaborative efforts from various international organizations, such as the World Health Organization (WHO), the Food and Agriculture Organization (FAO), the World Organization for Animal Health (OIE), and the United Nations Environment Programme (UNEP), could play a pivotal role in controlling and preventing fascioliasis in endemic areas of the world through awareness and education, animal health management, and meat inspection (Corning 2014; Mas-Coma et al. 2023).
Fasciolopsiasis
Fasciolopsiasis is a foodborne zoonotic parasitic disease caused by the infection of Fasciolopsis buski (intestinal fluke) and is transmitted by snails of the Planorbidae family (Lu et al. 2018). Fasciolopsiasis is categorized as a neglected disease by the WHO, and it is considered endemic in specific regions of the Far East and Southeast Asia, especially where pig farming, freshwater habitats, and open-air defecation are common (Siles-Lucas et al. 2021; Toledo et al. 2019). The life cycle of Fasciolopsis buski closely resembles that of Fasciola hepatica and begins with the hatching of eggs into miracidia, and then the miracidia invade snails of the Planorbidae family (Guerrant et al. 2011; Siles-Lucas et al. 2021). The infected snails, e.g., Planorbis spp. and Segmentina spp., with miracidia, often feed on plants fertilized with human night soil. The cercariae of Fasciolopsis buski then emerge and encyst on the tubers or nuts of these plants, and when humans or animals consume these infected plants and their raw derivatives, it can lead to infection (Siles-Lucas et al. 2021). Infections with Fasciolopsis buski are often asymptomatic but can lead to ulcers or abscesses in the duodenum and jejunum, causing epigastric pain resembling peptic ulcer disease (Mas-Coma et al. 2019). Severe cases of fasciolopsiasis may result in eosinophilia, anemia, and systemic allergic symptoms in children (Kimberlin et al. 2022; Long et al. 2012). Preventive measures for both fasciolosis and fasciolopsiasis should include proper sewage disposal, environmental sanitation, education, and personal hygiene (Mas-Coma et al. 2021; Zhou 2022).
Paragonimiasis
Paragonimiasis occurs in humans and animals by eating raw or inadequately cooked crabs or crayfish infected with metacercaria of Paragonimus spp. (Ahn et al. 2021). Several Paragonimus spp. have been reported to cause human infections globally. However, these infections are more common in Asian enclaves, including Korea, Japan, China, Taiwan, the Philippines, Thailand, Vietnam, and India (Blair 2019). Species of snail-like Melanoides and Tarebia act as the first intermediate host (Paller et al. 2021). This disease mainly infects livestock and humans, leading to pathological lesions, most commonly of the pulmonary system, although extra-pulmonary infections in the central nervous system also occur (Ahn et al. 2021). Effective anthelmintic drugs that can be used against paragonimiasis are praziquantel and triclabendazole (Richter 2022). In addition, the application of a one-health approach, such as raising awareness, implementing diagnostics, and emphasizing the importance of preventive measures for parasitic infections, is essential. Moreover, dietary recommendations include avoiding consuming raw or undercooked aquatic meat like freshwater crabs and crayfish and ensuring proper cooking to prevent Paragonimiasis effectively (Farag et al. 2023; Sadhukhan 2022).
Schistosomiasis
The infection of blood fluke of various species of Schistosoma causes schistosomiasis. The WHO has reported that approximately 770 million people are at risk of schistosomiasis, and it is spread over more than 78 countries in Africa, Asia, and Latin America, especially in poor communities (WHO 2022). Biomphalaria, Lanistes, Planorbarius, Neotricula, Oncomelania, Robertsiella, and Melanoides species of snails are responsible for acting as intermediate hosts for these blood flukes (Ovando and Marchi 2021; Saito 2022). The infective stage is either schistosomula or cercarial penetration on the skin (Anderson and Enabulele 2021; Li et al. 2021; Limpanont et al. 2020). The use of night soil (human waste) as fertilizers has been applied since ancient times (Kawa et al. 2019), and it has acted as a potential facilitator for parasites excreted by feces. In China, in 2007, half of the farmers in 36 selected villages applied night soil to crops, and its usage was highly associated with S. japonicum infections (Carlton et al. 2015). A preventive measure for schistosomiasis could be the breaking of the life cycle of the parasite in the endemic areas. The WHO employs several strategies to combat schistosomiasis. One key method is preventive treatment using the drug praziquantel, given as mass drug administration (MDA) (King et al. 2020). This approach targets high-risk populations, like school-age children and adults living in endemic areas (like Sub-Saharan Africa and Yemen), to reduce schistosomiasis infection and related health problems (Deol et al. 2019). The WHO also conducts health education campaigns to inform communities about the disease’s risks, transmission, and prevention, such as creating awareness toward clean and safe water sources in endemic regions. In non-endemic areas (like Europe and North America), strategies have focused on targeting the risks and safety measures for imported cases (Gabrielli and Garba Djirmay 2023). Surveillance systems help monitor disease prevalence and program impact while research improves diagnostics and treatment methods. Such integrated one-health approaches ultimately play a significant role in reducing the burden of schistosomiasis worldwide.
Echinostomiasis
Echinostomiasis is one of the most neglected trematode infections in humans (Toledo et al. 2019). Humans become infected after ingesting raw or insufficiently cooked molluscs, fish, crustaceans, amphibians, or aquatic vegetables (Toledo et al. 2022). Echinostomiasis is endemic in Southeast Asia, the Middle East, and East Africa (Siles-Lucas et al. 2021). Adult Echinostoma spp. of parasites commonly inhabit the intestines of a wide range of birds and mammals, including humans (also recorded in reptiles and fishes) (Pham et al. 2022; Seo et al. 2020; Toledo et al. 2019). The prevalence of echinostomiasis in endemic areas, especially in Sub-Saharan African and Asian countries, remained unchanged despite environmental measures and education (Yigezu et al. 2018). Cercarial infection rates of Echinostome spp. in snail species decreased from 58 to 3.6% between 2007 and 2016 in South-West Ethiopia, which could be due to increased resistance development by snails against the parasitic load (Mengistu et al. 2011; Mereta et al. 2019; Toledo and Esteban 2016). Protecting fishponds from night soil fertilization or fecal contamination, proper waste management, and education campaigns are important measures to protect potential hosts from this parasitic infection (Toledo and Esteban 2016).
Clonorchiasis and opisthorchiasis
Clonorchiasis and opisthorchiasis are parasitic diseases caused by the infections of Clonorchis sinensis, Opisthorchis viverrini, and Opisthorchis felineus flukes (Pozio and Morales 2022). These parasitic diseases are major public health problems affecting over 15 million in the trans-national region of the Mekong River basin in Asian countries: Cambodia, Yunnan Province and Guangxi Zhuang of China, Lao, Myanmar, Thailand, and Vietnam (Sripa et al. 2021). Snails of the Bithyniidae family have a major role in the transmission cycle as the first intermediate host, while fish act as the secondary/paratenic host (Charoensuk et al. 2022; Ovando and Marchi 2021). There should be proper selection, handling, and treatment of food of aquatic origin before consumption, as consumption of undercooked fish is the main route of infection in humans. Education, improving sanitary conditions, and treatment can be effective in preventing infections, especially in areas where human night soil and pig feces have been applied as nourishment for the fish.
Angiostrongyliasis
Angiostrongyliasis has been reported increasingly in parts of Asia, the Pacific islands, Australia, the USA, the Caribbean (initially Cuba), the Caribbean islands, Mallorca, and the Canary Islands. It is now widely distributed across many tropical/sub-tropical and a few more temperate localities, with the parasite found in numerous host species (Turck et al. 2022). It is estimated that more than 7000 cases of neuro-angiostrongyliasis have been reported in humans (Niebuhr et al. 2019). Angiostrongylus cantonensis (rat lungworm), the causative agent for angiostrongyliasis, is a nematode parasite having a complex life cycle where rats are the final hosts, gastropods (snails) act as intermediate hosts, and with several paratenic hosts (freshwater prawns/shrimp, crabs, flatworms, fish, frogs, toads, lizards, and centipedes) (Turck et al. 2022). The wide range of snail species (more than 60) belonging to gastropods is responsible for harboring the intermediate larval stage of this nematode (Brozzo et al. 2020; Segeritz et al. 2021; Turck et al. 2022). Since the wide range of infections occurs in different snail species and humans and other animals, it is crucial to raise awareness of the associated dangers of consuming snails as food and feeding materials. These food preferences are regarded as customs in, for example, China and Thailand, where undercooked snails have been consumed for generations (Wang et al. 2012). Changing such preferences may be challenging regardless of educational efforts. In Thailand, studies have shown that Angiostrongylus parasites, responsible for human angiostrongyliasis, were present in up to 33.3% of snail species, including Pila virescens and Pomacea canaliculata (Watthanakulpanich et al. 2021). For better prevention and control of angiostrongyliasis, regular screening and monitoring of its associated parasite in food, meat, and environment are needed, potentially applying one-health principles for the presence of the infective parasite (Sears et al. 2021; Turck et al. 2022).
Application of diagnostic techniques to detect the STPDs
The successful application of diagnostic techniques or tools for detecting the presence of parasites in snails, environment/habitats (water, soil), paratenic hosts, and the definitive hosts requires accurate knowledge of species of parasites and their snail vectors. Such techniques should be rapid, sensitive, and robust for detecting and identifying different stages of parasites (Hammoud et al. 2022). These diagnostic tools are crucial for adequately managing STPDs, limiting their spread from the environment to snails, humans, and animals (Table 5).
Direct shedding and microscopy are the traditional techniques used for the detection/identification of parasites in snails, where the snail tissues and their shedding materials are examined under a microscope to visualize the parasites (Joof et al. 2020). Serology-based techniques, for example, enzyme-linked immunosorbent assay (ELISA) and immunochromatographic assays (ICA), can be sensitive and specific for detecting parasitic antigens in snail tissues or body fluids (Yuan et al. 2023). The polymerase chain reaction (PCR) techniques were found to be more sensitive than the conventional shedding techniques for the detection of infection of S. mansoni in the Biomphalaria species of snails (Joof et al. 2020).
Multiplex PCR (simultaneous detection of multiple species of parasites and snails) and qPCR (amenable to high-throughput and multiplexing, sensitive, faster, cheaper) are methods of choice coupled with environmental DNA (eDNA). The eDNA detection technique can be a potential tool for monitoring of parasitic agents in water/soil samples (Jones et al. 2018). The limit of detection of the eDNA assays for the detection of Fasciola hepatica and snails (Austropeplea tomentosa) was observed to be 14 fg and 50 fg genomic DNA, respectively, in water samples (Rathinasamy et al. 2021). Thus, such eDNA-based techniques can be useful in detecting snail-borne parasites in various environmental samples (Rathinasamy et al. 2021).
Loop-mediated isothermal amplification (LAMP) is another molecular technique that is simple and rapid and can be used for the detection of parasites in snails (Tran et al. 2022). Recently, it has been suggested that LAMP results for certain STPDs, such as schistosomiasis, appear to be comparable to or better than commonly used diagnostic techniques (García-Bernalt Diego et al. 2021). PCR is a widely used molecular technique used for the detection of DNA or RNA either from parasites or from snails.
Future perspectives
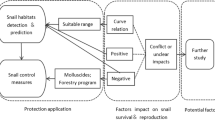
It is noteworthy that STPDs are not only a concern of public health, but they also have the potential to be cross-transmitted between animals and birds. STPDs, like schistosomiasis, fascioliasis, and angiostrongyliasis, pose a significant threat to human and animal health globally (Hammoud et al. 2022). In the future, proper prevention and control of STPDs appears to be a significant challenge for public health systems and veterinary medicine. The one-health approach to most STPDs requires broader collaborative efforts between human and animal health professionals, environmental experts, and public health authorities. Promoting surveillance, health education, veterinary care, and environmental management can mitigate the risks and minimize the transmission of these STPDs. With advancements in diagnostic and treatment methods, there is hope for better management of these diseases, especially with the application of one-health approaches (Fig. 7).
The dynamics of STPDs are likely to be affected by several factors, climate change, modified temperature, and rainfall patterns, which can affect the distribution and abundance of snail species and, consequently, the transmission of the diseases (Bula et al. 2017; Donham et al. 2022; Saallah et al. 2020). Likewise, urbanization and globalization can facilitate the spread of snails and parasites across borders and increase human exposure through travel and trade (Celis-Ramírez et al. 2022). While snails can serve as hosts for disease-causing parasites, it is important to note that they also play important roles in ecosystems as food sources for other animals and as decomposers. Therefore, any efforts to control snail populations and STPDs should be implemented in a naturally responsible manner and considered in conjunction with the current epidemiological situation in the endemic areas.
Snails are high in protein content, making them a nutritious food source for livestock and other animals (Uboh et al. 2014). Moreover, they efficiently convert feed into biomass, requiring fewer resources than traditional livestock (Mvodo Meyo et al. 2021). However, it is noted that snails can serve as hosts for disease-causing parasites, and there is a risk that these parasites could be transmitted to animals consuming snail-based feed. In addition, if snails are not raised under proper conditions, they may accumulate harmful substances in their tissues that could be toxic to animals (Bula et al. 2017; Donham et al. 2022; Saallah et al. 2020). Therefore, the establishment of appropriate protocols and methods for snail farming is essential to producing safe and nutritious snails that lead to minimal risks associated with the spread of STPDs. Moreover, strict regulations and monitoring should be put in place to ensure that snails used for animal feed are free from harmful substances and parasites.
Looking forward, the potential of snails as an alternative feed source should be explored further while ensuring that any hazards associated with their use are minimized. With careful planning and management, snails have the potential to be commercially utilized as food or feed sources that are sustainable, safe, and nutritious for humans and animals.
Conclusions
Snails are natural scavengers and play a vital role in the ecosystem. Both land and water snail species are prone to be infected with parasites from their surrounding environments. The physiological systems and visceral organs of snails are prone to parasitic lodgement, infestation, and inflammatory responses, and such parasitic infections in snails can compromise the growth and development of snails. As snails appear to be increasingly exploited for food or feed applications in the future, parasites invading snails can transmit to humans and animals. Such transmission can increase the risks of various parasitic diseases in humans and animals, including but not limited to fasciolosis, fasciolopsiasis, schistosomiasis, and angiostrongyliasis. These STPDs pose significant threats to human and animal health globally. Understanding the impacts and interactions of various abiotic factors on the growth and physiology of snails and infecting parasites is critical while aiming to exploit snails for various commercial applications. In this context, the application of a one-health concept is necessary to maximize the commercial utilization of snails as food and feed while minimizing the risks of snail-associated parasitic diseases. Various diagnostic tools can assist in detecting snail-infecting parasites in food, feed, animals, and the environment. The integrated use of more sensitive, specific, and cheaper diagnostic tools allows simultaneous monitoring of disease prevalence for better control and prevention of STPDs. Moreover, appropriate farming methods and protocols for snails must be developed, and regulations should be put in place to minimize the associated hazards of using snails as an alternative nutrient source. As the potential of snails as an alternative source of feed for animals is promising, future research efforts are necessary to ensure the safe and responsible use of snails for the benefit of human, animal, and environmental health.
Data availability
Not applicable.
Abbreviations
- AC:
-
Angiostrongylus cantonensis
- C:
-
Cercaria
- Ca:
-
Calcium
- CF:
-
Crude fiber
- CHO:
-
Carbohydrates
- CS:
-
Clonorchis sinensis
- DH:
-
Definitive host
- IH:
-
Intermediate host
- FB:
-
Fasciolopsis buski
- Fe:
-
Iron
- DM:
-
Dry matter
- FG:
-
Fasciola gigantica
- FH:
-
Fasciola hepatica
- L1:
-
1st larval stage
- L2:
-
2nd larval stage
- L3:
-
3rd larval stage
- Mc:
-
Metacercaria
- Mi:
-
Miracidium
- OF:
-
Opisthorchis felineus
- OV:
-
Opisthorchis viverrini
- PW:
-
Paragonimus westermani
- R:
-
Redia
- Sc:
-
Sporocyst
- SH:
-
Schistosoma haematobium
- SJ:
-
Schistosoma japonicum
- SM:
-
Schistosoma mansoni
- SMa:
-
Schistosoma malayensis
- SMe:
-
Schistosoma mekongi
- STPDs:
-
Snail transmitted parasitic diseases
- TP:
-
Total protein
References
Adema CM et al (2017) Whole genome analysis of a schistosomiasis-transmitting freshwater snail. Nat Commun 8(1):15451. https://doi.org/10.1038/ncomms15451
Adeyeye SAO, Bolaji OT, Abegunde TA, Adesina TO (2020) Processing and utilization of snail meat in alleviating protein malnutrition in Africa: a review. Nutr Food Sci 50(6):1085–1097. https://doi.org/10.1108/NFS-08-2019-0261
Ahn CS et al (2021) Spectrum of pleuropulmonary paragonimiasis: an analysis of 685 cases diagnosed over 22 years. J Inf Secur 82(1):150–158. https://doi.org/10.1016/j.jinf.2020.09.037
Akan M, Michling F, Matti K, Krause S, Muno-Bender J, Wendland J (2020) Snails as taxis for a large yeast biodiversity. Fermentation 6(3):90. https://doi.org/10.3390/fermentation6030090
Ali SM et al (2017) Field use of molluscicides in schistosomiasis control programmes: an operational manual for programme managers. World Health Organization
Amobi M, Ebenebe C, Nwobodo J, Ezenwaeya G, Okereke N, Okpoko V (2019) Effects of snail offal meal on performance of broiler chickens. Int J Livest Prod 10(2):43–48. https://doi.org/10.5897/IJLP2018.0552
Anderson TJ, Enabulele EE (2021) Schistosoma mansoni. Trends Parasitol 37(2):176–177. https://doi.org/10.1016/j.pt.2020.06.003
Ansari B et al (2021) Molecular phylogenetics of Physa acuta (Pulmonata: Basommatophora): an invasive species in Central Punjab Pakistan. Braz J Biol 83. https://doi.org/10.1590/1519-6984.246984
Bargues MD et al (2020) Genetic uniformity, geographical spread and anthropogenic habitat modifications of lymnaeid vectors found in a One Health initiative in the highest human fascioliasis hyperendemic of the Bolivian Altiplano. Parasit Vectors 13:1–19. https://doi.org/10.1186/s13071-020-04045-x
Barrios-Figueroa R, Urbina M (2023) Behavioural and physiological responses to salinization and air exposure during the ontogeny of a freshwater South American snail. Conserv Physiol 11(1):coac089. https://doi.org/10.1093/conphys/coac089
Blair D (2019) Paragonimiasis. In: Digenetic Trematodes, pp 105–138. https://doi.org/10.1007/978-3-030-18616-6_5
Blair D (2022) Lung flukes of the genus Paragonimus: ancient and re-emerging pathogens. Parasitology 149(10):1286–1295. https://doi.org/10.1017/S0031182022000300
Bouchet P et al (2017) Revised classification, nomenclator and typification of gastropod and monoplacophoran families. Malacologia 61(1-2):1–526. https://doi.org/10.4002/040.061.0201
Brenzinger B, Schrödl M, Kano Y (2021) Origin and significance of two pairs of head tentacles in the radiation of euthyneuran sea slugs and land snails. Sci Rep 11(1):21016. https://doi.org/10.1038/s41598-021-99172-5
Brozzo A et al (2020) Molecular phylogeny and trait evolution of Madeiran land snails: radiation of the Geomitrini (Stylommatophora: Helicoidea: Geomitridae). Cladistics 36(6):594–616. https://doi.org/10.1111/cla.12440
Bula W, Leiwakabessy F, Rumahlatu D (2017) The influence of environmental factors on the diversity of gastropods in Marsegu Island, Maluku. Biosaintifika 9(3):483–491
Caldeira RL, Teodoro TM, Jannotti-Passos LK, Lira-Moreira PM, Goveia CDO, Carvalho ODS (2016) Characterization of South American snails of the genus Biomphalaria (Basommatophora: Planorbidae) and Schistosoma mansoni (Platyhelminthes: Trematoda) in molluscs by PCR-RFLP. Biomed Res Int 2016. https://doi.org/10.1155/2016/1045391
Carlton EJ, Liu Y, Zhong B, Hubbard A, Spear RC (2015) Associations between schistosomiasis and the use of human waste as an agricultural fertilizer in China. PLoS Negl Trop Dis 9(1):e0003444. https://doi.org/10.1371/journal.pntd.0003444
Celis-Ramírez M, Quintero-Angel M, Varela-M RE (2022) Control of invasive alien species: The Giant African snail (Lissachatina fulica) a difficult urban public management challenge. J Environ Manag 322:116159. https://doi.org/10.1016/j.jenvman.2022.116159
Chakraborty K, Joy M (2020) High-value compounds from the molluscs of marine and estuarine ecosystems as prospective functional food ingredients: an overview. Food Res Int 137:109637. https://doi.org/10.1016/j.foodres.2020.109637
Chang Z et al (2020) Snail promotes the generation of vascular endothelium by breast cancer cells. Cell Death Dis 11(6):457. https://doi.org/10.1038/s41419-020-2651-5
Charoensuk L, Ribas A, Chedtabud K, Prakobwong S (2022) Infection rate of Opisthorchis viverrini metacercariae in cyprinoid fish from the markets and its association to human opisthorchiasis in the local community in the Northeast Thailand. Acta Trop 225:106216. https://doi.org/10.1016/j.actatropica.2021.106216
Cilia G, Fratini F (2018) Antimicrobial properties of terrestrial snail and slug mucus. J Complement Integr Med 15(3). https://doi.org/10.1515/jcim-2017-0168
Colgan D (2020) Molluscan Research and the sustainable development goals, vol 40. Taylor & Francis, pp 283–285. https://doi.org/10.1080/13235818.2020.1795965
Corning S (2014) World Organisation for Animal Health: strengthening Veterinary Services for effective One Health collaboration. Rev Sci Tech 33(2):639–650. https://doi.org/10.20506/rst.33.2.2305
Deol AK et al (2019) Schistosomiasis-assessing progress toward the 2020 and 2025 global goals. N Engl J Med 381(26):2519–2528. https://doi.org/10.1056/NEJMoa1812165
Dewi V, Sato S, Yasuda H (2017) Effects of a mud snail Cipangopaludina chinensis laeta (Architaenioglossa: Viviparidae) on the abundance of terrestrial arthropods through rice plant development in a paddy field. Appl Entomol Zool 52:97–106. https://doi.org/10.1007/s13355-016-0458-8
Dhiman V, Pant D (2021) Human health and snails. J Immunoass Immunochem 42(3):211–235. https://doi.org/10.1080/15321819.2020.1844751
Di Filippo MF et al (2022) Effectiveness of snail slime in the green synthesis of silver nanoparticles. Nanomaterials 12(19):3447. https://doi.org/10.3390/nano12193447
Ding HL et al (2016) Revision of sinistral land snails of the genus Camaena (Stylommatophora, Camaenidae) from China based on morphological and molecular data, with description of a new species from Guangxi, China. Zookeys 584:25. https://doi.org/10.3897/zookeys.584.7173
Djikeng FT et al (2022) Effect of different processing methods on the proximate composition, mineral content, and functional properties of snail (Archachatina marginata) meat. J Agric Food Res 8:100298. https://doi.org/10.1016/j.jafr.2022.100298
Donham EM, Strope LT, Hamilton SL, Kroeker KJ (2022) Coupled changes in pH, temperature, and dissolved oxygen impact the physiology and ecology of herbivorous kelp forest grazers. Glob Chang Biol 28(9):3023–3039. https://doi.org/10.1111/gcb.16125
Dummee V, Kruatrachue M, Trinachartvanit W, Tanhan P, Pokethitiyook P, Damrongphol P (2012) Bioaccumulation of heavy metals in water, sediments, aquatic plant and histopathological effects on the golden apple snail in Beung Boraphet reservoir, Thailand. Ecotoxicol Environ Saf 86:204–212. https://doi.org/10.1016/j.ecoenv.2012.09.018
Edison A, Hendra P, Sidharta BBR, Herwiyanti S, Jacub C (2021) Inhibitory activity of snail (Achatina Fulica (Lam.) Bowdich) mucus on growth of mammary cancer in rat induced with DMBA (7, 12-Dimethylbenz (Α) Anthracene). J Pharm Sci Commun 18(2):102–111. https://doi.org/10.24071/jpsc.002525
Erkano WK (2021) Impacts of environmental parameters on the infectivity of freshwater Snail. Update on Malacology. Intech Open. https://doi.org/10.5772/intechopen.99829
Escobar-Correas S, Mendoza-Porras O, Dellagnola FA, Colgrave ML, Vega IA (2019) Integrative proteomic analysis of digestive tract glycosidases from the invasive golden apple snail, Pomacea canaliculata. J Proteome Res 18(9):3342–3352. https://doi.org/10.1021/acs.jproteome.9b00282
Farag MA, Mansour ST, Nouh RA, Khattab AR (2023) Crustaceans (shrimp, crab, and lobster): a comprehensive review of their potential health hazards and detection methods to assure their biosafety. J Food Saf 43(1):e13026. https://doi.org/10.1111/jfs.13026
Federspiel F, Skovmand S, Skarphedinsson S (2020) Eosinophilic meningitis due to Angiostrongylus cantonensis in Europe. Int J Infect Dis 93:28–39. https://doi.org/10.1016/j.ijid.2020.01.012
Fedorova OS et al (2020) Opisthorchis felineus infection, risks, and morbidity in rural Western Siberia, Russian Federation. PLoS Negl Trop Dis 14(6):e0008421. https://doi.org/10.1371/journal.pntd.0008421
Fortunato H (2015) Mollusks: tools in environmental and climate research. Am Malacol Bull 33(2):310–324. https://doi.org/10.4003/006.033.0208
Gabrielli AF, Garba Djirmay A (2023) Schistosomiasis in Europe. Curr Trop Med Rep:1–9. https://doi.org/10.1007/s40475-023-00286-9
Galaktionov KV, Dobrovolskij A (2013) The biology and evolution of trematodes: an essay on the biology, morphology, life cycles, transmissions, and evolution of digenetic trematodes. Springer Science & Business Media. https://doi.org/10.1007/978-94-017-3247-5
García-Bernalt Diego J, Fernández-Soto P, Febrer-Sendra B, Crego-Vicente B, Muro A (2021) Loop-mediated isothermal amplification in schistosomiasis. J Clin Med 10(3):511. https://doi.org/10.3390/jcm10030511
Ghose KC (1961) Observations on the digestive enzymes and cellulolytic bacteria of the giant land snail Achatina fulica and their occurrence in the Gastropoda. Proc Zool Soc Lond 137(1):127–133. https://doi.org/10.1111/j.1469-7998.1961.tb06165.x
Ghosh S, Jung C, Meyer-Rochow VB (2016) Snail farming: an Indian perspective of a potential tool for food security. Ann Aquac Res 3(3):1–6. https://doi.org/10.47739/2379-0881/1024
Glöer P, Bössneck U (2013) Freshwater molluscs from Nepal and North India with the description of seven new species. Arch Molluskenkd 142(1):137–156. https://doi.org/10.1127/arch.moll/1869-0963/142/137-156
Guerrant RL, Walker DH, Weller PF (2011) Tropical infectious diseases: principles, pathogens and practice e-book. Elsevier Health Science. https://doi.org/10.1345/aph.1Q302
Gugliandolo E et al (2021) Protective effect of snail secretion filtrate against ethanol-induced gastric ulcer in mice. Sci Rep 11(1):3638. https://doi.org/10.1038/s41598-021-83170-8
Haase M, Grego J, Erőss ZP, Farkas R, Fehér Z (2021) On the origin and diversification of the stygobiotic freshwater snail genus Hauffenia (Caenogastropoda: Hydrobiidae) with special focus on the northern species and the description of two new species. Eur J Tax 775:143-184-143-184. https://doi.org/10.5852/ejt.2021.775.1555
Hailegebriel T, Nibret E, Munshea A (2020) Prevalence of Schistosoma mansoni and S. haematobium in snail intermediate hosts in Africa: a systematic review and meta-analysis. J Trop Med. https://doi.org/10.1155/2020/8850840
Hammoud C et al (2022) Simultaneous genotyping of snails and infecting trematode parasites using high-throughput amplicon sequencing. Mol Ecol Resour 22(2):567–586. https://doi.org/10.1111/1755-0998.13492
Hill EA, Hunt CO, Lucarini G, Mutri G, Farr L, Barker G (2015) Land gastropod piercing during the Late Pleistocene and early Holocene in the Haua Fteah, Libya. J Archaeol Sci Rep 4:320–325. https://doi.org/10.1016/j.jasrep.2015.09.003
Horsáková V, Nekola JC, Horsák M (2020) Integrative taxonomic consideration of the Holarctic Euconulus fulvus group of land snails (Gastropoda, Stylommatophora). Syst Biodivers 18(2):142–160. https://doi.org/10.1080/14772000.2020.1725172
Hu R-S et al (2021) Differential expression of microRNAs and tRNA fragments mediate the adaptation of the liver fluke Fasciola gigantica to its intermediate snail and definitive mammalian hosts. Int J Parasitol 51(5):405–414. https://doi.org/10.1016/j.ijpara.2020.10.009
Idczak-Figiel PA, Ostrowski M, Nowakowska A (2023) The influence of environmental stressful conditions on the interaction between heat shock proteins and chaperone-assisted proteins in land snails, Helix pomatia L. Can J Zool. https://doi.org/10.1139/cjz-2023-0118
Imathiu S (2020) Benefits and food safety concerns associated with consumption of edible insects. Nutri Food Sci J 18:1–11. https://doi.org/10.1016/j.nfs.2019.11.002
Jackiewicz M (2020) Phylogeny and relationships within European species of the family Lymnaeidae (Gastropoda: Pulmonata: Basommatophora). Folia Malacol 5(1):61–95. https://doi.org/10.12657/folmal.005.003
Jin W et al (2022) Chromosome-level genome assembly of the freshwater snail Bellamya purificata (Caenogastropoda). Zool Res 43(4):683. https://doi.org/10.24272/j.issn.2095-8137.2022.118
Jones RA et al (2018) Detection of Galba truncatula, Fasciola hepatica and Calicophoron daubneyi environmental DNA within water sources on pasture land, a future tool for fluke control? Parasit Vectors 11(1):1–9. https://doi.org/10.1186/s13071-018-2928-z
Joof E, Andrus PS, Sowunmi K, Onyango VM, Wade CM (2020) Comparing PCR techniques against conventional cercarial shedding methods for detecting Schistosoma mansoni infection in Biomphalaria snails. Acta Trop 212:105716. https://doi.org/10.1016/j.actatropica.2020.105716
Kalinda C, Chimbari MJ, Grant WE, Wang H-H, Odhiambo JN, Mukaratirwa S (2018) Simulation of population dynamics of Bulinus globosus: effects of environmental temperature on production of Schistosoma haematobium cercariae. PLoS Negl Trop Dis 2(8):e0006651. https://doi.org/10.1371/journal.pntd.0006651
Kalinda C, Chimbari MJ, Mukaratirwa S (2017) Effect of temperature on the Bulinus globosus — Schistosoma haematobium system. Infect Dis Poverty 6(1):57. https://doi.org/10.1186/s40249-017-0260-z
Kawa NC et al (2019) Night soil. Ethnobiol Lett 10(1):40–49. https://doi.org/10.14237/ebl.10.1.2019.1351
Kimberlin D, Fischer M, Long SS, Prober CG (2022) Principles and practice of pediatric infectious diseases E-Book. Elsevier Health Sciences
King CH et al (2020) Impact of different mass drug administration strategies for gaining and sustaining control of Schistosoma mansoni and Schistosoma haematobium infection in Africa. Am J Trop Med Hyg 103(1 Suppl):14. https://doi.org/10.4269/ajtmh.19-0829
Lalor R et al (2021) Pathogenicity and virulence of the liver flukes Fasciola hepatica and Fasciola gigantica that cause the zoonosis Fasciolosis. Virulence 12(1):2839–2867. https://doi.org/10.1080/21505594.2021.1996520
Larsson J, Westram AM, Bengmark S, Lundh T, Butlin R (2020) A developmentally descriptive method for quantifying shape in gastropod shells. J R Soc Interface 17(163):20190721. https://doi.org/10.1098/rsif.2019.0721
Lee SY, Lee HJ, Kim YK (2019) Comparative analysis of complete mitochondrial genomes with Cerithioidea and molecular phylogeny of the freshwater snail, Semisulcospira gottschei (Caenogastropoda, Cerithioidea). Int J Biol Macromol 135:1193–1201. https://doi.org/10.1016/j.ijbiomac.2019.06.036
Li HM et al (2021) Nucleic acid amplification techniques for the detection of Schistosoma mansoni infection in humans and the intermediate snail host: a structured review and meta-analysis of diagnostic accuracy. Int J Infect Dis 112:152–164. https://doi.org/10.1016/j.ijid.2021.08.061
Limpanont Y, Phuphisut O, Reamtong O, Adisakwattana P (2020) Recent advances in Schistosoma mekongi ecology, transcriptomics and proteomics of relevance to snail control. Acta Trop 202:105244. https://doi.org/10.1016/j.actatropica.2019.105244
Llanwarne F, Helmby H (2021) Granuloma formation and tissue pathology in Schistosoma japonicum versus Schistosoma mansoni infections. Parasite Immunol 43(2):e12778. https://doi.org/10.1111/pim.12778
Long SS, Pickering LK, Prober CG (2012) Principles and practice of pediatric infectious disease. Elsevier Health Sciences
Lopatek M, Wieczorek K, Osek J (2022) Prevalence and antimicrobial resistance of bacterial foodborne pathogens isolated from raw bivalve molluscs subjected to consumption in Poland during a ten-year period. Foods 11(21):3521. https://doi.org/10.3390/foods11213521
Lopes AS, Pulido-Murillo EA, López-Hernández D, de Melo AL, Pinto HA (2021) First report of Melanoides tuberculata (Mollusca: Thiaridae) harboring a xiphidiocercaria in Brazil: a new parasite introduced in the Americas? Parasitol Int 82:102284. https://doi.org/10.1016/j.parint.2021.102284
Lu XT et al (2018) Snail-borne parasitic diseases: an update on global epidemiological distribution, transmission interruption and control methods. Infect Dis Poverty 7:1–16. https://doi.org/10.1186/s40249-018-0414-7
Lv S et al (2018) Invasive Pomacea snails as important intermediate hosts of Angiostrongylus cantonensis in Laos, Cambodia and Vietnam: Implications for outbreaks of eosinophilic meningitis. Acta Trop 183:32–35. https://doi.org/10.1016/j.actatropica.2018.03.021
Lydeard C, Cummings KS (2019) Freshwater mollusks of the world: a distribution atlas. JHU Press. https://doi.org/10.1353/book.66164
Malek EA (2018) Snail transmitted parasitic diseases: volume I. CRC Press. https://doi.org/10.1201/9781351076654
Marcogliese DJ, Pietrock M (2011) Combined effects of parasites and contaminants on animal health: parasites do matter. Trends Parasitol 27(3):123–130. https://doi.org/10.1016/j.pt.2010.11.002
Mas-Coma S, Valero MA, Bargues MD (2019) Fascioliasis. In: Digenetic Trematodes, pp 71–103. https://doi.org/10.1007/978-3-030-18616-6_4
Mas-Coma S, Valero MA, Bargues MD (2023) One Health for fascioliasis control in human endemic areas. Trends Parasitol. https://doi.org/10.1016/j.pt.2023.05.009
Mas-Coma S et al (2021) Domestic pig prioritized in one health action against fascioliasis in human endemic areas: experimental assessment of transmission capacity and epidemiological evaluation of reservoir role. One Health 13:100249. https://doi.org/10.1016/j.onehlt.2021.100249
Mehmood K et al (2017) A review on epidemiology, global prevalence and economical losses of fasciolosis in ruminants. Microb Pathog 109:253–262. https://doi.org/10.1016/j.micpath.2017.06.006
Mengistu M, Shimelis T, Torben W, Terefe A, Kassa T, Hailu A (2011) Human intestinal schistosomiasis in communities living near three rivers of Jimma town, southwestern Ethiopia. Ethiop J Health Sci 21(2):111–118. https://doi.org/10.4314/ejhs.v21i2.69051
Mereta ST et al (2019) Environmental determinants of distribution of freshwater snails and trematode infection in the Omo Gibe River Basin, southwest Ethiopia. Infect Dis Poverty 8(1):1–10. https://doi.org/10.1186/s40249-019-0604-y
Munonye J, Moses P (2019) Profitability analysis of snail farming in Owerri agricultural zone Imo state. AES. https://doi.org/10.22004/ag.econ.289661
Mvodo Meyo ES, Nkemasong ZA, Shu G, Ngono JPN, Ngosong C (2021) Snail farming as an alternative profitable livestock system for sustainable development sustainable development in Africa: fostering sustainability in one of the world’s most promising continents. Springer, pp 477–490. https://doi.org/10.1007/978-3-030-74693-3_26
Naveen Y, Singh A (2019) Shell morphometric traits showing normal distribution pattern and positive correlation in freshwater snail, Lymnaea luteola (Hygrophila: Lymnaeidae). J Exp Zoology India 22(1):273–278
Ngenwi A, Mafeni J, Etchu K, Oben F (2010) Characteristics of snail farmers and constraints to increased production in West and Central Africa. Afr J Environ Sci Technol 4(5):274–278
Nguyen K, Gemmell B, Rohr J (2020) Effects of temperature and viscosity on miracidial and cercarial movement of Schistosoma mansoni: ramifications for disease transmission. Int J Parasitol 50(2):153–159. https://doi.org/10.1016/j.ijpara.2019.12.003
Niebuhr C, Jarvi SI, Siers SR (2019) A review of rat lungworm infection and recent data on its definitive hosts in Hawaii. Hum Wildl Interact 13(2):238–249
Noothuan N, Apitanyasai K, Panha S, Tassanakajon A (2021) Snail mucus from the mantle and foot of two land snails, Lissachatina fulica and Hemiplecta distincta, exhibits different protein profile and biological activity. BMC Res Notes 14(1):1–7. https://doi.org/10.1186/s13104-021-05557-0
Olkeba BK et al (2020) Environmental and biotic factors affecting freshwater snail intermediate hosts in the Ethiopian Rift Valley region. Parasit Vectors 13(1):1–13. https://doi.org/10.1186/s13071-020-04163-6
O'Sullivan C, Fried B, Sherma J (2011) Studies on the pH tolerance of freshwater snails. Trends Comp Biochem Physiol 15:63–66
Ovando XM, Marchi GF (2021) An annotated checklist of Planorbinae (Heterobranchia, Hygrophila, Planorbidae) from northwestern Argentina. Check List 17(6):1493–1507. https://doi.org/10.15560/17.6.1493
Palasio RGS, Zanotti-Magalhães EM, Tuan R (2018) Genetic diversity of the freshwater snail Biomphalaria tenagophila (d'Orbigny, 1835) (Gastropoda: Hygrophila: Planorbidae) across two coastal areas of southeast Brazil. Folia Malacol 26(4). https://doi.org/10.12657/folmal.026.019
Paller VG et al (2021) Paragonimus westermani infection of freshwater crab Sundathelphusa philippina and melaniid snails in Cadacan River in Irosin, Sorsogon, Philippines. J Parasit Dis:1–9. https://doi.org/10.1007/s12639-020-01340-3
Parveen S, Chakraborty A, Chanda DK, Pramanik S, Barik A, Aditya G (2020) Microstructure analysis and chemical and mechanical characterization of the shells of three freshwater snails. ACS Omega 5(40):25757–25771. https://doi.org/10.1021/acsomega.0c03064
Pati SG, Panda F, Samanta L, Paital B (2023) Spatio-temporal changes in oxidative stress physiology parameters in apple snail Pila globosa as a function of soil Mg, Ca, organic carbon and aquatic physico-chemical factors. Environ Geochem Health 45:2591–2610. https://doi.org/10.1007/s10653-022-01376-9
Pertiwi MP, Saputri DD (2020) Golden apple snail (Pomacea canaliculata) as an alternative protein source in Pasupati catfish (Pangasius sp.) fish feed. Nus Biosci 12(2). https://doi.org/10.13057/nusbiosci/n120212
Pham LTK, Saijuntha W, Lawton SP, Le TH (2022) Mitophylogenomics of the zoonotic fluke Echinostoma malayanum confirms it as a member of the genus Artyfechinostomum Lane, 1915 and illustrates the complexity of Echinostomatidae systematics. Parasitol Res 121(3):899–913. https://doi.org/10.1007/s00436-022-07449-z
Pissia MΑ, Matsakidou A, Kiosseoglou V (2021) Raw materials from snails for food preparation. Future Foods 3:100034. https://doi.org/10.1016/j.fufo.2021.100034
Ponder WF, Lindberg DR, Ponder JM (2019a) Biology and evolution of the mollusca, volume 1. CRC Press. https://doi.org/10.1201/9781351115667
Ponder WF, Lindberg DR, Ponder JM (2019b) Biology and evolution of the mollusca, volume 2. CRC Press. https://doi.org/10.1201/9781351115254-9
Pouil S, Jones NJ, Smith JG, Mandal S, Griffiths NA, Mathews TJ (2020) Comparing trace element bioaccumulation and depuration in snails and mayfly nymphs at a coal ash-contaminated site. Environ Toxicol Chem 39(12):2437–2449. https://doi.org/10.1002/etc.4857
Pozio E, Morales MAG (2022) Clonorchiasis and Opisthorchiasis helminth infections and their impact on global public health. Springer, pp 221–256. https://doi.org/10.1007/978-3-031-00303-5_7
Raheem D, Budha P, Naggs F, Preece R (2010) An illustrated guide to the land snails of Nepal. The Natural History Museum, London
Rathinasamy V et al (2021) Towards understanding the liver fluke transmission dynamics on farms: detection of liver fluke transmitting snail and liver fluke-specific environmental DNA in water samples from an irrigated dairy farm in Southeast Australia. Vet Parasitol 291:109373. https://doi.org/10.1016/j.vetpar.2021.109373
Rey O et al (2021) Population genetics of African Schistosoma species. Infect Genet Evol 89:104727. https://doi.org/10.1016/j.meegid.2021.104727
Richter J (2022) Current status of the treatment of paragonimiasis. Lung Cancer 8:10
Rosenberg G (2014) A new critical estimate of named species-level diversity of the recent Mollusca. Am Malacol Bull 32(2):308–322. https://doi.org/10.4003/006.032.0204
Saadi AJ, Davison A, Wade CM (2020) Molecular phylogeny of freshwater snails and limpets (Panpulmonata: Hygrophila). Zool J Linnean Soc 190(2):518–531. https://doi.org/10.1093/zoolinnean/zlz177
Saallah S, Ishak N, Sarbon N (2020) Effect of different molecular weight on the antioxidant activity and physicochemical properties of golden apple snail (Ampullariidae) protein hydrolysates. Food Res 4(4):1363–1370. https://doi.org/10.26656/fr.2017.4(4).348
Sabourin E, Alda P, Vázquez A, Hurtrez-Boussès S, Vittecoq M (2018) Impact of human activities on fasciolosis transmission. Trends Parasitol 34(10):891–903. https://doi.org/10.1016/j.pt.2018.08.004
Sadhukhan SK (2022) Prevention and control of parasitic zoonoses textbook of parasitic zoonoses. Springer, pp 83–90. https://doi.org/10.1007/978-981-16-7204-0_9
Saito T (2022) First record of the non-indigenous freshwater snail Galba humilis (Say, 1822) (Mollusca: Hygrophila: Lymnaeidae) in Japan. Bioinvasions Rec 11(2). https://doi.org/10.3391/bir.2022.11.2.16
Schultz JH, Bu L, Adema CM (2018) Comparative immunological study of the snail Physella acuta (Hygrophila, Pulmonata) reveals shared and unique aspects of gastropod immunobiology. Mol Immunol 101:108–119. https://doi.org/10.1016/j.molimm.2018.05.029
Schwertz CI et al (2015) Eurytrematosis: an emerging and neglected disease in South Brazil. World J Exp Med 5(3):160. https://doi.org/10.5493/wjem.v5.i3.160
Sears WJ et al (2021) AcanR3990 qPCR: a novel, highly sensitive, bioinformatically informed assay to detect Angiostrongylus cantonensis infections. Clin Infect Dis 73(7):e1594–e1600. https://doi.org/10.1093/cid/ciaa1791
Segeritz L, Cardona A, Taubert A, Hermosilla C, Ruiz A (2021) Autochthonous Angiostrongylus cantonensis, Angiostrongylus vasorum and Aelurostrongylus abstrusus infections in native terrestrial gastropods from the Macaronesian Archipelago of Spain. Parasitol Res 120:2671–2680. https://doi.org/10.1007/s00436-021-07203-x
Seo M et al (2020) Ancient echinostome eggs discovered in archaeological strata specimens from a Baekje Capital Ruins of South Korea. J Parasitol 106(1):184–187. https://doi.org/10.1645/19-55
Siles-Lucas M, Becerro-Recio D, Serrat J, González-Miguel J (2021) Fascioliasis and fasciolopsiasis: current knowledge and future trends. Res Vet Sci 134:27–35. https://doi.org/10.1016/j.rvsc.2020.10.011
Soldatenko EV, Petrov AA (2019) Musculature of the penial complex: a new criterion in unravelling the phylogeny of Hygrophila (Gastropoda: Pulmonata). J Morphol 280(4):508–525. https://doi.org/10.1002/jmor.20960
Squires RL (2022) The earliest Ancistrolepis (Gastropoda: Buccinidae) and its geologic implications. Paleo Bios 39(2). https://doi.org/10.5070/P939257077
Sripa B, Suwannatrai AT, Sayasone S, Do DT, Khieu V, Yang Y (2021) Current status of human liver fluke infections in the Greater Mekong Subregion. Acta Trop 224:106133. https://doi.org/10.1016/j.actatropica.2021.106133
Stein E (2018) Microbiologically safe foods. Scientific e-Resources
Swart EM, Starkloff NC, Ypenburg S, Ellers J, van Straalen NM, Koene JM (2020) The effect of mating on female reproduction across hermaphroditic freshwater snails. Invertebr Biol 139(1):e12275. https://doi.org/10.1111/ivb.12275
Toledo R, Álvarez-Izquierdo M, Esteban JG, Muñoz-Antoli C (2022) Neglected food-borne trematodiases: echinostomiasis and gastrodiscoidiasis. Parasitol 149(10):1319–1326. https://doi.org/10.1017/S0031182022000385
Toledo R, Alvárez-Izquierdo M, Muñoz-Antoli C, Esteban JG (2019) Intestinal trematode infections. In: Digenetic Trematodes, pp 181–213. https://doi.org/10.1007/978-3-030-18616-6_7
Toledo R, Esteban J (2016) An update on human echinostomiasis. Trans R Soc Trop Med Hyg 110(1):37–45. https://doi.org/10.1093/trstmh/trv099
Tran L, Toet H, Beddoe T (2022) Environmental detection of Fasciola hepatica by loop-mediated isothermal amplification. PeerJ 10:e13778. https://doi.org/10.7717/peerj.13778
Turck HC, Fox MT, Cowie RH (2022) Paratenic hosts of Angiostrongylus cantonensis and their relation to human neuroangiostrongyliasis globally. One Health:100426. https://doi.org/10.1016/j.onehlt.2022.100426
Uboh FE, Williams IO, Essien NC (2014) Effect of processing on the proximate and mineral composition of Archachatina marginata and Achatina achatina. Food Public Health 4(1):10–14
Uehara H, Murakami H, Yusa Y (2021) Predation by the carrion crow Corvus corone (Passeriformes: Corvidae) on the apple snail Pomacea canaliculata (Architaenioglossa: Ampullariidae) in different locations in Japan. Appl Entomol Zool 56(3):385–392. https://doi.org/10.1007/s13355-021-00747-5
Vaughn CC, Hakenkamp CC (2001) The functional role of burrowing bivalves in freshwater ecosystems. Freshw Biol 46(11):1431–1446. https://doi.org/10.1046/j.1365-2427.2001.00771.x
Wang J, Lu X, Zhang J, Ouyang Y, Qin Z, Zhao B (2020) Using golden apple snail to mitigate its invasion and improve soil quality: a biocontrol approach. Environ Sci Pollut Res 27:14903–14914. https://doi.org/10.1007/s11356-020-07998-9
Wang QP, Wu ZD, Wei J, Owen R, Lun ZR (2012) Human Angiostrongylus cantonensis: an update. Eur J Clin Microbiol Infect Dis 31:389–395. https://doi.org/10.1007/s10096-011-1328-5
Watthanakulpanich D et al (2021) Co-occurrence of Angiostrongylus malaysiensis and Angiostrongylus cantonensis DNA in cerebrospinal fluid: Evidence from human eosinophilic meningitis after ingestion of raw snail dish in Thailand. Food Waterborne Parasitol 24:e00128. https://doi.org/10.1016/j.fawpar.2021.e00128
WHO (2022) WHO guideline on control and elimination of human schistosomiasis. World Health Organization
Wiggering B, Neiber MT, Krailas D, Glaubrecht M (2019) Biological diversity or nomenclatural multiplicity: the Thai freshwater snail Neoradina prasongi Brandt, 1974 (Gastropoda: Thiaridae). Syst Biodivers 17(3):260–276. https://doi.org/10.1080/14772000.2019.1606862
Wright AC, Fan Y, Baker GL (2018) Nutritional value and food safety of bivalve molluscan shellfish. J Shellfish Res 37(4):695–708. https://doi.org/10.2983/035.037.0403
Yamazaki T, Sonoda T, Nobetsu T, Goshima S (2018) Contribution to the knowledge of the taxonomy of the Japanese species of Volutharpa (Gastropoda: Buccinidae). Venus 76(1-4):1–18
Yigezu G et al (2018) Habitat suitability modelling for predicting potential habitats of freshwater snail intermediate hosts in Omo-Gibe river basin, Southwest Ethiopia. Ecol Inform 45:70–80. https://doi.org/10.1016/j.ecoinf.2018.04.002
Yuan L et al (2019) Resveratrol inhibits the invasion and metastasis of colon cancer through reversal of epithelial-mesenchymal transition via the AKT/GSK-3β/Snail signaling pathway Corrigendum in/10.3892/mmr. 2022.12870. Mol Med Rep 20(3):2783–2795. https://doi.org/10.3892/mmr.2019.10528
Yuan X et al (2023) The 22nd chromatography component of the Fasciola gigantica excretory-secretory products decreased the proliferation of peripheral blood mononuclear cells from buffalo. Animals 13(4):564. https://doi.org/10.3390/ani13040564
Zhang S, Zhang S (2018) Three species of Siphonalia Adams, 1863 (Gastropoda, Buccinidae) from China seas, with descriptions of two new species. J Oceanol Limnol 36(6):2333–2336. https://doi.org/10.1007/s00343-019-7218-x
Zhou X-N (2022) Infectious Diseases of Poverty: progress achieved during the decade gone and perspectives for the future. Infec Dis Poverty 11:1–4. https://doi.org/10.1186/s40249-021-00931-3
Acknowledgements
The authors would like to thank the members of the Khanal Laboratory (KLab) for their scientific input and discussion.
Funding
Open access funding provided by Nord University. This study was supported by the CEER project (Project number: 2021/10345) funded by the Norwegian Agency for International Cooperation and Quality Enhancement in Higher Education (HK-dir) under the Norwegian Partnership Program for Global Academic Cooperation (NORPART) with support from the Norwegian Ministry of Education and Research (MER).
Author information
Authors and Affiliations
Contributions
CRP: Conceptualization, Visualization, Validation, Writing – Original Draft; Review and Editing; HL: Project administration, Validation, Supervision, Review & Editing; KSU: Validation, Review and Editing; PK: Conceptualization, Visualization, Validation, Supervision, Funding, Project administration, Resources, Validation, Review and Editing. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Section Editor: Pengfei Cai
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pathak, C.R., Luitel, H., Utaaker, K.S. et al. One-health approach on the future application of snails: a focus on snail-transmitted parasitic diseases. Parasitol Res 123, 28 (2024). https://doi.org/10.1007/s00436-023-08021-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00436-023-08021-z