Abstract
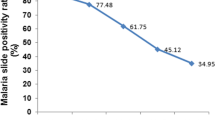
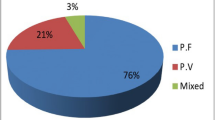
Evidence on the trends of the proportion of malaria infections detected by routine passive case detection at health facilities is important for public health decision making especially in areas moving towards elimination. The objective was to assess nine years of trends on clinical malaria infections detected at health facility and its associated climate factors, in the water resource development set up of Wonji sugar estate, Oromia, Ethiopia. Retrospective data were collected from malaria-suspected patient recording logbook at Wonji sugar factory’s primary hospital. Monthly average meteorological data were obtained from the estate meteorological station. Data were collected from April through June 2018 and January 2022. The data were analyzed using Stata version 16.0 software for Chi-square and regression analysis. Over the last nine years, 34,388 cases were legible for analysis with complete data. Of these, 11.75% (4039/34388) were positive for clinical malaria. Plasmodium vivax test positivity was the highest proportion (8.2%, n = 2820) followed by Plasmodium falciparum (3.48%, n = 1197) and mixed infections (P. falciparum and P. vivax, 0.06%, n = 21). The odds of being positive for malaria was highest in males (AOR = 1.46; 95%CI = 1.36–1.52; P < 0.001) compared to females and in older individuals of above 15 years old (AOR = 4.55, 95%CI = 4.01–5.17, P < 0.001) followed by school-aged children (5–15 years old) (AOR = 2.16; 95%CI = 1.88–2.49, P < 0.001). There was no significant variation in the proportion of malaria-positive cases in the dry and wet seasons (P = 0.059). Malaria test positivity rates were associated with average monthly rainfall (AdjIRR = 1.00; 95%CI = 1.00–1.001, P < 0.001) while negatively associated with average monthly minim temperature (adjIRR = 0.94; 95%CI = 0.94–0.95; P < 0.001) and average monthly relative humidity (adjIRR = 0.99, 95%CI = 0.99–1.00, P = 0.023). There was year-round malaria transmission, adults especially males and school children frequently tested malaria positive. Hence, alternative vector management tools like larval source management have to be deployed besides ITNs and IRS in such water development areas to achieve the malaria elimination goal.



Similar content being viewed by others
Data availability
The raw data used for this study are available from the corresponding author up on reasonable request.
Change history
11 August 2023
A Correction to this paper has been published: https://doi.org/10.1007/s00436-023-07938-9
References
Abeku T et al (2004) Effects of meteorological factors on epidemic malaria in Ethiopia: a statistical modelling approach based on theoretical reasoning. Parasitology 128(6):585–593
Aregawi M et al (2014) Time series analysis of trends in malaria cases and deaths at hospitals and the effect of antimalarial interventions, 2001–2011, Ethiopia. PLoS One 9(11):e106359
Farajzadeh M et al (2015) Spatiotemporal Anopheles population dynamics, response to climatic conditions: the case of Chabahar, South Baluchistan Iran. Annals of Global Health 81(5):694–704
File T, Dinka H, Golassa L (2019) A retrospective analysis on the transmission of Plasmodium falciparum and Plasmodium vivax: the case of Adama City, East Shoa Zone, Oromia, Ethiopia. Malar J 18(1):193
FMOH (2020) Federal Ministry of Health Ethiopia 2020, Ethiopia malaria elimination strategic plan: 2021–2025. Addis Ababa, Ethhiopia
Gari T, Lindtjørn B (2018) Reshaping the vector control strategy for malaria elimination in Ethiopia in the context of current evidence and new tools: opportunities and challenges. Malar J 17:454. https://doi.org/10.1186/s12936-018-2607-8
Gemechu T, Samuel A, Yewhalaw D (2015) Ten years trend analysis of malaria prevalence and its correlation with climatic variables in Sibu Sire District, east Wollega zone, Oromia regional state, Western Ethiopia: a retrospective study. Sci, Technol Arts Res J 4(4):99–105
Getachew and Teshome S (2022) Ten years trend analysis of malaria prevalence in Ziway Dugda District, Arsi Zone, Ethiopia. Int J Biomed Eng Clin Sci 8(1):8–11
Getachew D, Balkew M, Tekie H (2020) Anopheles larval species composition and characterization of breeding habitats in two localities in the Ghibe River Basin, southwestern Ethiopia. Malar J 19(1):1–13
Girum T, Shumbej T, Shewangizaw M (2019) Burden of malaria in Ethiopia, 2000–2016: findings from the Global Health Estimates 2016. Trop Dis, Travel Med Vaccines 5(1):1–7
Haileselassie W, Zemene E, Lee M-C, Zhong D, Zhou G, Taye B, Dagne A, Deressa W, Kazura JW, Yan G, Yewhalaw D (2021) The effect of irrigation on malaria vector bionomics and transmission intensity in western Ethiopia. Parasites Vectors 14(516):1–11
Hawaria D et al (2019) Ten years malaria trend at Arjo-Didessa sugar development site and its vicinity, Southwest Ethiopia: a retrospective study. Malar J 18(1):1–11
Kajeguka D, Tarmo S (2017) Meteorological influence in pattern of malaria cases in North-Eastern Tanzania: five years analysis of malaria incidence and climate condition. J Pathol Microbiol 2:1–5
Karunamoorthi K, Bekele M (2012) Changes in malaria indices in an Ethiopian health centre: a five year retrospective analysis. Health Scope 1(3):118–126
Kenea O, Balkew M, Gebre-Michael T (2011) Environmental factors associated with larval habitats of anopheline mosquitoes (Diptera: Culicidae) in irrigation and major drainage areas in the middle course of the Rift Valley, central Ethiopia. J Vector Borne Dis 48(2):85
Kenea O et al (2016) Human-biting activities of Anopheles species in south-central Ethiopia. Parasit Vectors 9(1):527
Kibret S et al (2010) The impact of a small-scale irrigation scheme on malaria transmission in Ziway area. Central Ethiopia Trop Med Int Health 15(1):41–50
Kibret S et al (2019) Malaria around large dams in Africa: effect of environmental and transmission endemicity factors. Malar J 18(1):1–12
Kulkarni MA et al (2016) 10 years of environmental change on the slopes of Mount Kilimanjaro and its associated shift in malaria vector distributions. Front Public Health 4:281
Murdock C, Sternberg E, Thomas M (2016) Malaria transmission potential could be reduced with current and future climate change. Sci Rep 6(1):1–7
Ogwang R et al (2018) The 2015–2016 malaria epidemic in Northern Uganda; what are the implications for malaria control interventions? Acta Trop 188:27–33
PMI (2020) U.S. President’s malaria initiative ethiopia malaria operational plan FY 2020. Retrieved from www.pmi.gov
PMI (2022) U.S. President’s malaria initiative ethiopia malaria operational plan FY 2022. Retrieved from www.pmi.gov
Price RN et al (2020) Plasmodium vivax in the era of the shrinking P. falciparum map. Trends Parasitol 36(6):560–570
Robinson LJ et al (2015) Strategies for understanding and reducing the Plasmodium vivax and Plasmodium ovale hypnozoite reservoir in Papua New Guinean children: a randomised placebo-controlled trial and mathematical model. PLoS Med 12(10):e1001891
Sani Dedgeba HM (2017) Malaria trends in Silt’i district from 2009–2015 and current childhood malaria in K’ibbet hospital, south-central Ethiopia. Malar World J 8(22):1–6
Sena LD, Deressa WA, Ali AA (2014) Analysis of trend of malaria prevalence in south-west Ethiopia: a retrospective comparative study. Malar J 13:188
Shamebo T, Petros B (2019) Trend analysis of malaria prevalence in Halaba special district Southern Ethiopia. BMC Res Notes 12(1):1–6
Shapiro LL, Whitehead SA, Thomas MB (2017) Quantifying the effects of temperature on mosquito and parasite traits that determine the transmission potential of human malaria. PLoS Biol 15(10):e2003489
Taffese HS et al (2018) Malaria epidemiology and interventions in Ethiopia from 2001 to 2016. Infect Dis Poverty 7(06):1–9
Tesfa H, Bayih AG, Zeleke AJ (2018) A 17-year trend analysis of malaria at Adi Arkay, north Gondar zone, Northwest Ethiopia. Malar J 17(155):6
Taye B, Lelisa K, Emana D, Asale A, Yewhalaw D (2016) Seasonal dynamics, longevity, and biting activity of anopheline mosquitoes in southwestern Ethiopia. J Insect Sci 16(1):6
Villena OC, Ryan SJ, Murdock CC, Johnson LR (2020) Temperature impacts the transmission of malaria parasites by Anopheles gambiae and Anopheles stephensi mosquitoes. https://doi.org/10.1101/2020.07.08.194472
Wang X et al (2016) Effects of a malaria elimination program: a retrospective study of 623 cases from 2008 to 2013 in a Chinese county hospital near the China-Myanmar border. Emerg Microbes Infect 5:e6
WHO (2020) World Health Organization (WHO). Basic malaria microscopy. World Health Organization, 2010
WHO (2021) World malaria report 2017. World Health Organization, Geneva
Xiang J et al (2018) Association between malaria incidence and meteorological factors: a multi-location study in China, 2005–2012. Epidemiol Infect 146(1):89–99
Yamana TK, Eltahir EA (2013) Incorporating the effects of humidity in a mechanistic model of Anopheles gambiae mosquito population dynamics in the Sahel region of Africa. Parasit Vectors 6(1):1–10
Yewhalaw D et al (2014) Water resource developments in Ethiopia: potential benefits and negative impacts on the environment, vector-borne diseases, and food security. Environ Rev 22(4):364–371
Acknowledgements
We are grateful to all staff members of Wonji Sugar estate’s: Primary Hospital and Metrology office for providing us with valuable data in realizing this study. We are so grateful for all the data collectors. Finally, Mr. Gezahgn Negash deserves appreciation for mapping the study site.
Funding
Addis Ababa and Dilla University have financially supported the study.
Author information
Authors and Affiliations
Contributions
KL conceived the study design, undertook the field study, analyzed the data, and drafted the manuscript. EH took part in data analysis and manuscript preparation. DB participated in study design and manuscript revision. SD involved in the study design, revision of the manuscript, and facilitation of administrative issues. All authors read the manuscript.
Corresponding authors
Ethics declarations
Ethical considerations
Ethical clearances were obtained from Addis Ababa University, Aklilu Lemma Institute of Pathobiology, Institutional Review Board (IRB) (reference number: ALIPB/IRB/012/2017/2018) and from Oromia Regional Health Bureau Ethical Clearance Committee (reference number: BEFO/DBTHL/1–8/422).
Consent to participate
This is not applicable.
Consent for publication
This is not applicable.
Conflict of interest
The authors declare no competing interests.
Disclaimer
The organizations have no role in the design, data collection, analysis, interpretation of the data, and writing the manuscript.
Additional information
Section Editor: Tobili Sam-Yellowe
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: The authors regret that the Figure 2 that appears in the original article is incorrect.
The original article has been corrected.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lelisa, K., Hailemeskel, E., Bekele, D. et al. Malaria positivity rate trend analysis at water resources development project of Wonji Sugar Estate Oromia, Ethiopia. Parasitol Res 122, 2259–2266 (2023). https://doi.org/10.1007/s00436-023-07923-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-023-07923-2