Abstract
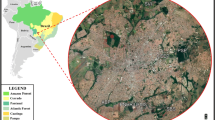
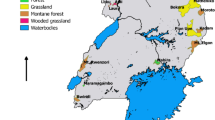
Small mammals are important hosts and/or reservoirs of Trypanosoma spp. This study aimed to verify the prevalence of Trypanosoma spp. in non-volant small mammals from the Brazilian Cerrado and to test the effects of T. lainsoni on the neutrophil/lymphocyte ratio (N/L) and body condition in rodent and marsupial populations. For this, we collected blood samples of 293 individuals captured in five forest fragments between 2019 and 2020. Blood was used to prepare the blood smears and packed on filter paper for DNA extraction. Generalized linear models were performed to test the effects of T. lainsoni on host health. The DNA was submitted to nested PCR targeting the Trypanosoma spp. 18S rRNA gene. From blood smears analyzed by microscopy, we obtained a positivity rate of 7.2% for Trypanosoma spp. About 31.1% of Gracilinanus agilis, Didelphis albiventris, and Rhipidomys macrurus samples were positive in nested PCR. From the obtained sequences, 83.3% were genetically identical to T. lainsoni and about 11% to T. cruzi TcI. In addition, we reported the infection of T. lainsoni in Hylaeamys megacephalus. We suggest that T. lainsoni does not influence the body condition and N/L ratio for either G. agilis or R. macrurus. Overall, our results expand the host list of T. lainsoni and demonstrate the infection of small mammals by T. cruzi TcI in peri-urban areas.



Similar content being viewed by others
Availability of data and materials
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Ahmed HA, MacLeod ET, Hide G, Welburn SC, Picozzi K (2011) The best practice for preparation of samples from FTA®cards for diagnosis of blood borne infections using African trypanosomes as a model system. Parasites & Vectors 4:1–7. https://doi.org/10.1186/1756-3305-4-68
Barros FDNL, Júnior FDS, Costa SM, De Farias DM, Moura MAO, Júnior PSB, Góes-Cavalcante G, Scofield A (2020) First report of natural infection by Trypanosoma cruzi in secretions of the scent glands and myocardium of Philander opossum (Marsupialia: Didelphidae): parasitological and clinicopathological findings. Vet Parasitol Reg Stud Reports 22:1–5. https://doi.org/10.1016/j.vprsr.2020.100463
Bezerra-Santos MA, Ramos RAN, Campos AK, Dantas-Torres F, Otranto D (2021) Didelphis spp. opossums and their parasites in the Americas: a One Heath perspective. Parasitol Res 120:4091–4111. https://doi.org/10.1007/s00436-021-07072-4
Brandão EMV, Xavier SCC, Carvalhaes JG, D’Andrea PS, Lemos FG, Azevedo FC, Cássia-Pires R, Jansen AM, Roque ALR (2019) Trypanosomatids in small mammals of an agroecosystem in Central Brazil: another piece in the puzzle of parasite transmission in an anthropogenic landscape. Pathogens 8:1–15. https://doi.org/10.3390/pathogens8040190
Bonvicino CR, De Oliveira JA, D’Andrea OS (2008) Guia dos roedores do Brasil, com chaves para gêneros baseadas em caracteres externos. Centro Pan-Americano de Febre Aftosa, Rio de Janeiro
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach, 2nd. edn. Springer-Verlag, New York
Claro HWP, Hannibal W (2022) Diet of small mammals in semi-deciduous forest fragments in Central Brazil. Austral Ecology 47:770–774. https://doi.org/10.1111/aec.13169
Costa BMA, Geise L, Pereira LG, Costa LP (2011) Phylogeography of Rhipidomys (Rodentia: Cricetidae: Sigmodontinae) and description of two new species from southeastern Brazilian. J Mammal 92:954–962. https://doi.org/10.1644/10-MAMM-A-249.1
Davis AK, Maney DL, Maerz JC (2008) The use of leukocyte profiles to measure stress in vertebrates: a review for ecologists. Funct Ecol 22:760–772. https://doi.org/10.1111/j.1365-2435.2008.01467.x
Díaz AG, Ragone PG, Rusman F, Floridia-Yapur N, Barquez RM, Díaz MM, Tomasini N, Diosque P (2020) A novel genotype and first record of Trypanosoma lainsoni in Argentina. Pathogens 9:1–11. https://doi.org/10.3390/pathogens9090731
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791. https://doi.org/10.2307/2408678
Ferreira JIGS, Da Costa AP, Nunes PH, Ramirez D, Fournier GFR, Saraiva D, Tonhosolo R, Marcili A (2017) New Trypanosoma species, Trypanosoma gennarii sp. nov., from South American marsupial in Brazilian Cerrado. Acta Tropica 176:249–255. https://doi.org/10.1016/j.actatropica.2017.08.018
Gottdenker NL, Chaves LF, Calzada JE, Saldaña A, Carroll RC (2012) Host life history strategy, species diversity, and habitat influence Trypanosoma cruzi vector infection in changing landscapes. PLoS Negl Trop Dis 6:e1884. https://doi.org/10.1371/journal.pntd.0001884
Gulas-Wroblewski BE, Kairis RB, Gorchakoc R, Wheless A, Murray KO (2021) Optimization of DNA extraction from field-collected mammalian whole blood on filter paper for Trypanosoma cruzi (Chagas Disease) detection. Pathogens 10:1–8. https://doi.org/10.3390/pathogens10081040
Hernandez SE, Strona ALS, Leiner NO, Suzán G, Romano MC (2018) Seasonal changes of faecal cortisol metabolite levels in Gracilinanus agilis (Didelphimorphia: Didelphidae) and its association to life histories variables and parasite loads. Conserv Physiol 6(1):coy021. https://doi.org/10.1093/conphys/coy021
Jansen AM, Leon L, Machado GM, Da Silva MH, Souza-Leão SM, Deane MP (1991) Trypanosoma cruzi in the opossum Didelphis marsupialis: parasitological and serological follow-up of the acute infection. Exp Parasitol 73:249–259. https://doi.org/10.1016/0014-4894(91)90096-F
Jansen AM, Xavier SCC, Roque ALR (2015) The multiple and complex and changeable scenarios of the Trypanosoma cruzi transmission cycle in the sylvatic environment. Acta Tropica 151:1–15. https://doi.org/10.1016/j.actatropica.2015.07.018
Jansen AM, Xavier SCC, Roque ALR (2018) Trypanosoma cruzi transmission in the wild and its most important reservoir hosts in Brazil. Parasit Vectors 11:1–25. https://doi.org/10.1186/s13071-018-3067-2
Jansen AM, Xavier SCC, Roque ALR (2020) Landmarks of the knowledge and Trypanosoma cruzi biology in the wild environment. Front Cell Infect Microbiol 10:1–15. https://doi.org/10.3389/fcimb.2020.00010
Kostygov AY, Karnkowska A, Votýpka J, Tashyreva D, Maciszewski K, Yurchenko V, Lukeš J (2021) Euglenozoa: taxonomy, diversity and ecology, symbioses and viroses. Open Biology 11(3):1–47. https://doi.org/10.1098/rsob.200407
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: Molecular Evolutionary Genetics Analysis across computing platforms. Mol Biol Evol 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Lessa LG, Geise L (2014) Food habits and carnivory by a small size opossum, Gracilinanus agilis (Didelphimorphia: Didelphidae). Mastozoología neotropica 21:139–143
Lopes GP, Leiner NO (2015) Semelparity in a population of Gracilinanus agilis (Didelphimorphia: Didelphidae) inhabiting the Brazilian cerrado. Mamm Bio 80:1–6. https://doi.org/10.1016/j.mambio.2014.08.004
Macedo J, Loretto D, Vieira MV, Cerqueira R (2006) Classes de desenvolvimento em marsupiais: Um método para animais vivos. Mastozoologia neotropical 13:133–136
Naiff RD, Barrett TV (2013) Trypanosoma (Megatrypanum) lainsoni n. sp. from Mesomys hispidus (Rodentia: Echimyidae) in Brazil: trypomastigotes described from experimentally infected laboratory mice. Parasite 20:1–5. https://doi.org/10.1051/parasite/2013049
Nantes WAG, Barreto WTG, Santos FM, De Macedo GC, Rucco AC, Assis WO, Porfírio GEO, De Andrade GD, Jansen AM, Gerrera HM (2019) The influence of parasitism by Trypanosoma cruzi in the hematological parameters of the white ear opossum (Didelphis albiventris) from Campo Grande, Mato Grosso do Sul, Brazil. Int J Parasitol Parasites Wildl 9:16–20. https://doi.org/10.1016/j.ijppaw.2019.03.015
Nantes WAG, Santos FM, Macedo GC, Barreto WTG, Gonçalves LR, Rodrigues MS, Chulli JVM, Rucco AC, Assis WO, Porfírio GEO, Oliveira CE, Xavier SCC, Herrera HM, Jansen AM (2021) Trypanosomatid species in Didelphis albiventris from urban forest fragments. Parasitol Res 120:223–231. https://doi.org/10.1007/s00436-020-06921-y
Noya BA, González ON (2015) An ecological overview on the factors that drives to Trypanosoma cruzi oral transmission. Acta Tropica 151:94–102. https://doi.org/10.1016/j.actatropica.2015.06.004
Noyes HA, Stevens JR, Teixeira M, Phelan J, Holz P (1999) A nested PCR for the ssrRNA gene detects Trypanosoma binneyi in the platypus and Trypanosoma sp. in wombats and kangaroos in Australia. Int J Parasitol 29:331–339. https://doi.org/10.1016/s0020-7519(98)00167-2
Olifiers N, Gentile R, Fiszon JT (2005) Relation between small-mammal species composition and anthropic variables in the Brazilian Atlantic Forest. Braz J Biol 65:495–501. https://doi.org/10.1590/S1519-69842005000300015
Ortiz PA, Garcia HA, Lima L, Da Silva FM, Campaner M, Pereira CL, Jittapalapong S, Neves L, Desquesnes M, Camargo EP, Teixeira MMG (2018) Diagnosis and genetic analysis of the worldwide distributed Rattus-borne Trypanosoma (Herpetosoma) lewisi and its allied species in blood and fleas of rodents. Infect Genet Evol 63:380–390. https://doi.org/10.1016/j.meegid.2017.09.001
Patton JL, Pardiñas UFJ, D’Elía G (eds) (2015) Mammals of South America, Vol. 2: Rodents. University Of Chicago Press, Chicago
Peig J, Green AJ (2009) New perspectives for estimating body condition from mass/length data: the scaled mass index as an alternative method. Oikos 118:1883–1891. https://doi.org/10.1111/j.1600-0706.2009.17643.x
Pires MM, Martins EG, Silva MNF, Dos Reis SF (2010) Gracilinanus microtarsus (Didelphimorphia: Didelphidae). Mamm Species 42:33–40. https://doi.org/10.1644/851.1
Pronovost H, Peterson AC, Chavez BG, Blum MJ, Dumonteil E, Herrera CP (2020) Deep sequencing reveals multiclonality and new discrete typing units of Trypanosoma cruzi in rodents from the southern United States. J Microbiol Immunol Infect 53:622–633. https://doi.org/10.1016/j.jmii.2018.12.004
R Core Team (2022) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rademaker V, Herrea HM, Raffel TR, D’Andrea PS, Freitas TPT, Abreu UGP, Hudson PJ, Jansen AM (2009) What is the role of small rodents in the transmission cycle of Trypanosoma cruzi and Trypanosoma evansi (Kinetoplastida Trypanosomatidae)? A study case in the Brazilian Pantanal. Acta Tropica 111:102–107. https://doi.org/10.1016/j.actatropica.2009.02.006
Rocha FL, Roque ALR, Arrais RC, Santos JP, Lima VS, Xavier SCC, Cordeir-Estrela P, D’Andrea PS, Jansen AM (2012) Trypanosoma cruzi TcI and TcII transmission among wild carnivores, small mammals and dogs in a conservation unit and surrounding areas, Brazil. Parasitology 140:160–170. https://doi.org/10.1017/S0031182012001539
Rodrigues MS, Lima L, Xavier SCC, Herrera HM, Rocha FL, Roque ALR, Teixeira MMG, Jansen AM (2019) Uncovering Trypanosoma spp. diversity of wild mammals by the use of DNA from blood clots. Int J Parasitol Parasites Wildl 8:171–181. https://doi.org/10.1016/j.ijppaw.2019.02.004
Roman F, Iñiguez AM, Yeo M, Jansen AM (2018) Multilocus sequence typing: genetic diversity in Trypanosoma cruzi I (TcI) isolates from Brazilian didelphids. Parasites & Vectors 11:1–13. https://doi.org/10.1186/s13071-018-2696-9
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Santos FM, Sano NY (2022) Parasite association in non-volant small mammals in Brazil. Community Ecol 23:129–136. https://doi.org/10.1007/s42974-022-00080-z
Santos FM, Sano NY, Liberal SC, Dario MA, Nantes WAG, Alves FM, Da Silva AR, De Oliveira CE, Roque ALR, Herrera HM, Jansen AM (2022) Kinetoplastid species maintained by a small mammal community in the Pantanal biome. Pathogens 11(10):1205. https://doi.org/10.3390/pathogens11101205
Sato H, Takano A, Kawabata H, Une Y, Watanabe H, Mukhtar MM (2009) Trypanosoma cf. varani in an imported ball python (Python reginus) from Ghana. J Parasitol 95:1029–1033. https://doi.org/10.1645/GE-1816.1
Schulte-Hostedde AI, Millar JS, Hickling GJ (2001) Evaluating body condition in small mammals. Can J Zool 79:1021–1029. https://doi.org/10.1139/z01-073
Smith LM, Burgoyne LA (2004) Collecting, archiving and processing DNA from wildlife samples using FTA® databasing paper. BMC Ecology 4:1–11. https://doi.org/10.1186/1472-6785-4-4
Smith A, Clark S, Averis S, Lymbery AJ, Wayne AF, Morris KD, Thompson RCA (2008) Trypanosomes in a declining species of threatened Australian marsupial, the brush-tailed bettong Bettongia penicillata (Marsupialia: Potoroidae). Parasitology 135:1329–1335. https://doi.org/10.1017/S0031182008004824
Strona ALS, Levenhagem M, Leiner NO (2015) Reproductive effort and seasonality associated with male-biased parasitism in Gracilinanus agilis (Didelphimorphia: Didelphidae) infected by Eimeria spp. (Apicomplexa: Eimeriidae) in the Brazilian cerrado. Parasitology 142(8):1086–1094. https://doi.org/10.1017/S0031182015000402
Tamura K, Nei M (1993) Estimation of the number of nucleotide substitutions in the control region of mitochondrial DNA in humans and chimpanzees. Mol Biol Evol 10:512–526. https://doi.org/10.1093/oxfordjournals.molbev.a040023
Urdaneta-Morales S, Nironi I (1996) Trypanosoma cruzi in the anal glands of urban opossums. I – Isolation and experimental infections. Memórias do Instituto Oswaldo Cruz 91:399–403. https://doi.org/10.1590/S0074-02761996000400002
Velásquez-Ortiz N, Ramírez JD (2020) Understanding the oral transmission of Trypanosoma cruzi as a veterinary and medical foodborne zoonosis. Res Vet Sci 132:448–461. https://doi.org/10.1016/j.rvsc.2020.07.024
Viola LB, Attias M, Takata CS, Campaner M, De Souza W, Camargo EP, Teixeira MMG (2009) Phylogenetic analyses based on small subunit rRNA and glycosomal glyceraldehyde-3-phosphate dehydrogenase genes and ultrastructural characterization of two snake trypanosomes: Trypanosoma serpentis n. sp. from Pseudoboa nigra and Trypanosoma cascavelli from Crotalus durissus terrificus. J Eukaryot Microbiol 56:594–602. https://doi.org/10.1111/j.1550-7408.2009.00444.x
Viola LB, Campaner M, Takata CS, Ferreira RC, Rodrigues AC, Freitas RA, Duarte MR, Grego KF, Barrett TV, Camargo EP, MMG T (2008) Phylogeny of snake trypanosomes inferred by SSU rDNA sequences, their possible transmission by phlebotomines, and taxonomic appraisal by molecular, cross-infection and morphological analysis. Parasitology 135:595–605. https://doi.org/10.1017/S0031182008004253
Voigt GL, Swist SL (2011) Hematology techniques and concepts for veterinary technicians, 2nd edn. John Wiley & Sons, Chichester
Weksler M, Percequillo AR (2011) Key to the genera of the tribe Oryzomyini (Rodentia: Cricetidae: Sigmodontinae). Mastozoologia Neotropical 18:281–292
Zingales B, Miles MA, Campbell DA, Tibayrenc M, Macedo AM, Teixeira MMG, Schijman AG, Llewellyn MS, Lages-Silva E, Machado CR, Andrade SG, Sturm NR (2012) The revised Trypanosoma cruzi subspecific nomenclature: rationale, epidemiological relevance and research applications. Infect Genet Evol 12:240–253. https://doi.org/10.1016/j.meegid.2011.12.009
Zuri I, Gottreich A, Terkel J (1998) Social stress in neighboring and encountering blind mole-rats (Spalax ehrenbergi). Physiol Behav 64:611–620. https://doi.org/10.1016/S0031-9384(98)00085-7
Acknowledgements
The authors are grateful to Prof. PhD. Alexandre Reis Percequillo (University of São Paulo) for the identification of rodents.
Funding
This research was financially supported by Programa de Pesquisas Ecológicas de Longa Duração do Triângulo Mineiro e Sudeste de Goiás (PELD-TMSG) of Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq 441225/2016-0 and 441142/2020-6). M. M. O. received a scholarship from CNPq (142532/2018-5).
Author information
Authors and Affiliations
Contributions
All the authors contributed to the study’s conception and design. Material preparation, data collection, and analysis were performed by M. M. O., C. P. R. F., C. G. H., K. R. O., I. A. C. A., P. V. A. R., and N. O. L. The first draft of the manuscript was written by M. M. O., and all the authors commented on previous versions of the manuscript. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
The procedures described here were approved by the Ethics Committee on the Use of Animals of Federal University of Uberlândia (CEUA/UFU, 41/2019). The license for capturing the small mammals was issued by the Biodiversity Authorization and Information System (SISBIO, 68066-1).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Section Editor: Vyacheslav Yurchenko
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
de Oliveira, M.M., Ferrando, C.P.R., Gómez-Hernández, C. et al. Prevalence of Trypanosoma lainsoni and its effects of parasitism on the health of non-volant small mammals from the Brazilian Cerrado. Parasitol Res 122, 1509–1518 (2023). https://doi.org/10.1007/s00436-023-07851-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-023-07851-1