Abstract
The genus Haemaphysalis Koch, 1844 (Acari: Ixodidae) is the second-largest genus, with more than 170 described species that primarily parasitize mammals and birds (Guglielmone et al. 2014, Guglielmone et al. 2020). Haemaphysalis species are three-host ticks, mainly distributed in southern and southeastern Asia and tropical Africa (Guglielmone et al. 2014). The present study identified a tick, Haemaphysalis hoodi Warburton & Nuttall, 1909, collected from a human in Yaoundé, Cameroon. This tick species feed on birds in sub-Saharan Africa. To the best of our knowledge, this is the second record of H. hoodi from humans. In addition, 16S ribosomal RNA and cytochrome oxidase I sequences were generated for this species for the first time. Screening pan-Rickettsia-PCR infection gave a negative result.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The genus Haemaphysalis Koch, 1844 (Acari: Ixodidae) is the second-largest genus, with more than 170 described species (Guglielmone et al. 2020). Haemaphysalis species are three-host ticks that primarily parasitize mammals and birds (Guglielmone et al. 2014). Species from this genus are mainly distributed in southern and southeastern Asia and tropical Africa, some species are known from Australia, and only a few species occur in the Americas (Guglielmone et al. 2014). Haemaphysalis species are reservoirs and vectors of many pathogenic microorganisms of animals and humans. For instance, Haemaphysalis leachi (Audouin, 1826) transmits Babesia rossi in dogs (Kamani 2021), as well as Rickettsia conorii, which causes human tick-bite fever, and Coxiella burnetii, the causative agent of Q fever (Hoogstraal 1956). Limited studies have been performed on Haemaphysalis species from wildlife that focused on their diversity and role as potential vectors and reservoirs of pathogens.
Out of 47 described Haemaphysalis species endemic to the Afrotropic region, eleven species are known from Cameroon, namely H. aciculifer Warburton, 1913, H. camicasi Tomlinson & Apanaskevich 2019, H. hoodi Warburton & Nuttall, 1909, H. houyi Nuttall & Warburton, 1915, H. leachi, H. moreli Camicas et al. 1972, H. paraleachi Camicas et al. 1983, H. parmata Neumann, 1905, H. princeps Tomlinson & Apanaskevich 2019, H. punctaleachi Camicas et al. 1973, and H. tauffliebi Morel 1965 (Morel and Mouchet 1958; Morel 1965; Camicas et al. 1972, 1973, 1983; Hoogstraal and El Kammah 1972; Apanaskevich et al. 2007; Tomlinson and Apanaskevich 2019). The haemaphysalid subgenus Ornithophysalis Hoogstraal & Wassef, 1973, comprises 19 species divided into five structural-biological groups (Hoogstraal and Wassef 1973; Camicas et al. 1998). Many of the species have not been adequately studied structurally, biologically, or epidemiologically (Hoogstraal and Wassef 1973). Haemaphysalis hoodi is one of the four species of the Haemaphysalis doenitzi group, which also includes H. doenitzi Warburton & Nuttall, 1909, H. phasiana Saito, Hoogstraal & Wassef, 1974, and H. madagascariensis Colas-Belcour & Millot, 1948 (Camicas et al. 1998). This species is broadly distributed in sub-Saharan Africa (Hoogstraal 1956; Hoogstraal and Wassef 1973). Adults, nymphs, and larvae of H. hoodi feed primarily on various groups of birds, while records from mammals are rare (Guglielmone et al. 2014). In Cameroon, H. hoodi was recorded from different ground-feeding bird species (Hoogstraal 1956; Santos Dias 1958). Here, we report for the first time a specimen of this species collected from a human in Yaoundé, Cameroon, and provide data on its mitochondrial (16S rRNA, cox I) genes.
Material and methods
In late October 2021, a light brown tick was removed manually from the shoulder of a woman in Nkozoa in the Mefou and Afamba Division of the center region of Cameroon (3°52′53.2″N 11°41′54.6″E). The collected tick was transferred to a 1.5 ml tube containing 600 µl absolute ethanol and sent to Bundeswehr Institute of Microbiology, Munich, Germany, for investigation. The tick specimen was first identified using morphological keys (Hoogstraal 1956), under a Keyence VHX‑900F microscope (Itasca, IL, USA). DNA was extracted using the QIAamp mini DNA extraction kit (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. As this is a rare tick species and no sequences from this species are available, 16S rRNA (Halos et al. 2004) and cox I (Apanaskevich et al. 2011) mitochondrial genes were sequenced, and the sequences obtained were edited and compared with the respective sequences deposited in GenBank using BLASTN and phylogenetic analysis. Sequences for each gene were aligned using MAFFT (Katoh and Standley 2013) with default parameters and phylogenetic analyses performed with IQ-Tree2 v1.6.12 (Minh et al. 2020). Optimal evolutionary models were calculated for each gene: 16S (K3Pu + F + I + G4) and Cox1 (TIM2 + F + I + G4). Nodal support was estimated using ultrafast bootstrap (n = 10,000) and the 50% consensus trees were reported. The partial sequences of the mitochondrial 16S rRNA and cox I genes generated in this study for H. hoodi species have been deposited in GenBank under the accession numbers ON189038 and ON191014. Additionally, Rickettsia spp. screening was performed using a previously published real-time PCR assay targeting a part of the gltA gene (Wölfel et al. 2008).
Results and discussion
The tick was identified as a female Haemaphysalis hoodi. The specific characteristics of the female include moderately dense punctations on scutum, broadly salient palpi, and absence of posterodorsal spur on palpal segment II (Fig. 1A,B) (Hoogstraal 1956; Morel 1965). Birds, especially ground feeder birds, are specific hosts for species within the Ornithophysalis subgenus; although some species parasitize birds and mammals, others only parasitize mammals (Hoogstraal 1956; Hoogstraal and Wassef 1973).
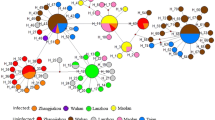
Haemaphysalis hoodi is a very rare parasite of humans (Guglielmone and Robbins 2018). Adults of H. hoodi have been found in three cases of human infestation in Ivory Coast although the exact localities were not reported (cited in Guglielmone et al. 2018). Our finding represents the second record of this tick species feeding on humans. Phylogenetic analysis indicated that the 16S rRNA sequence obtained (305 bp) from H. hoodi group in a moderately supported clade with H. bancrofti, H. doenitzi, and H. phasiana (Fig. 2). Of interest is that H. bancrofti is also part of this clade since it is classified in the subgenus Kaiseriana Dias, 1963, while H. hoodi and H. phasiana are classified in the Ornithophysalis Hoogstraal and Wassef, 1973, subgenus (Hoogstraal and Wassef 1973). Similarly, in the cox I analysis (636 bp), H. hoodi group in a well-supported clade with H. bancrofti, H. humerosa, and H. lagostrophi, the latter two species also belonging to the subgenus Ornithophysalis (Hoogstraal and Wassef 1973). While the overall support for the trees was weak, in each tree, several clades with good bootstrap support were obtained. No overwhelming support was found for any of the subgenera as monophyletic lineages. This may be due to limited phylogenetic signal due to the short sequences used in the analysis. It may, however, be noted that a recent analysis using 10 mitochondrial genes also resulted in a paraphyletic Haemaphysalis subgenus (Kelava et al. 2021). As such, more studies that focus on molecular systematics of the Haemaphysalis subgenera are needed to ascertain the validity of various subgenera. Even so, both 16S rRNA and cox I indicate that H. hoodi presents a unique genetic signature compared to other sequences available in the database that shows a genetic relationship to other members of the Ornithophysalis subgenus.
Maximum likelihood analysis of the 16S rRNA and cox I genes for the genus Haemaphysalis. Bootstrap support above 80% is indicated and the trees were rooted with Ixodes scapularis. The accession numbers used for the 16S rRNA and cox I genes are indicated behind the species names, respectively, and the tick sequenced in the current study is underlined. Subgenera are indicated in parentheses
Rickettsia spp. DNA was not amplified in the sample obtained from the H. hoodi female. It would have been expected to detect Rickettsia africae, which is responsible for the African tick-bite fever, mainly transmitted by Amblyomma species or Rickettsia aeschlimannii, a Hyalomma species related ricketsiae. Haemaphysalis species from Africa are not known as vectors for Rickettsia species. Haemaphysalis leachi was supposed to be a vector for Rickettsia conorii in southern Africa, but no isolates are available to confirm this. In Asia, especially in China, many Haemaphysalis species are vectors for Rickettsia spp., e.g., Rickettsia sibirica, Rickettsia heilongjiangensis, and Rickettsia japonica (Raoult and Parola 2007).
Data Availability
The sequences were submitted to GenBank under the following access numbers: ON189038, ON191014. The tick specimen is in the LCD collection at Bundeswehr Institute of Microbiology.
Change history
26 August 2022
A Correction to this paper has been published: https://doi.org/10.1007/s00436-022-07625-1
References
Apanaskevich DA, Horak IG, Camicas JL (2007) Redescription of Haemaphysalis (Rhipistoma) elliptica (Koch, 1844), an old taxon of the Haemaphysalis (Rhipisoma) leachi group from East and southern Africa, and of Haemaphysalis (Rhipistoma) leachi (Audouin, 1826) (Ixodida, Ixodidae). Onderstepoort J Vet Res 74:181–208
Apanaskevich DA, Horak IG, Matthee CA, Matthee S (2011) A new species of Ixodes (Acari: Ixodidae) from South African mammals. J Parasitol 97:389–398
Camicas JL, Hoogstraal H, El Kammah KM (1972) Notes on African Haemaphysalis ticks. VIII. H. (Rhipistoma) moreli sp. n., a carnivore parasite of the H. (R.) leachi group (Ixodoidea: Ixodidae). J Parasitol 58:1185–1196
Camicas JL, Hoogstraal H, El Kammah KM (1973) Notes on African Haemaphysalis ticks. XI. H. (Rhipistoma) punctaleachi sp. n., a carnivore parasite of West African forest carnivores (Ixodoidea: Ixodidae). J Parasitol 59:563–568
Camicas JL, Hoogstraal H, El Kammah KM (1983) Notes on African Haemaphysalis ticks. XIV. Description of adults of H. (Rhipistoma) paraleachi sp. n., a carnivore parasite of the H. (R.) leachi group (Ixodoidea: Ixodidae). J Parasitol 69:400–404
Camicas JL, Hervy JP, Adam F, Morel PC (1998) The ticks of the world (Acarida, Ixodida). Nomenclature, described stages, hosts, distribution. Paris: Orstom éditions, 233 pp
Guglielmone AA, Robbins RG, Apanaskevich DA, Petney TN, Estrada-Peña A Horak IG (2014) The hard ticks of the world (Acari: Ixodida: Ixodoidea). Springer, 738 pp
Guglielmone AA, Robbins RG (2018) Hard ticks (Acari: Ixodida: Ixodoidea) parasitizing humans. Springer, A global overview, p 313
Guglielmone AA, Petney TN, Robbins RG (2020) Ixodidae (Acari: Ixodoidea): descriptions and redescriptions of all known species from 1758 to December 31, 2019. Zootaxa 4871(1):001–322
Hoogstraal H (1956) African Ixodoidea, vol 1. Ticks of the Sudan (with special reference to Equatoria Province and with preliminary reviews of the genera Boophilus, Margaropus, Hyalomma). Research Report NM 005 050.29.07, US Dept. of the Navy, Bureau Med Surg
Halos L, Jamal T, Vial L, Maillard R, Suau A, Le Menach A, Boulouis H-J, Vayssier-Taussat M (2004) Determination of an efficient and reliable method for DNA extraction from ticks. Vet Res 35:709–713
Hoogstraal H, El Kammah KM (1972) Notes on African Haemaphysalis ticks X. H (Kaiseriana) aciculifer Warburton and H. (K) rugosa Santos Dias, the African representatives of the spinigera subgroup (Ixodoidea: Ixodidae). J Parasitol 58(5):960–978
Hoogstraal H, Wassef HY (1973) The Haemaphysalis ticks (Ixodoidea: Ixodidae) of birds. 3. H. (Ornithophysalis) Subgen. N.: definition, species, hosts, and distribution in the Oriental, Palearctic, Malagasy, and Ethiopian Faunal Regions. J Parasitol 59:1099–1117
Kamani J (2021) Molecular evidence indicts Haemaphysalis leachi (Acari: Ixodidae) as the vector of Babesia rossi in dogs in Nigeria. West Africa Ticks Tick-Borne Dis 12:101717
Katoh K, Standley DM (2013) MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol Biol Evol 30:772–780
Kelava S, Mans BJ, Shao R, Moustafa MAM, Matsuno K, Takano A, Kawabata H, Sato K, Fujita H, Ze C, Plantard O, Hornok S, Gao S, Barker D, Barker SC, Nakao R (2021) Phylogenies from mitochondrial genomes of 120 species of ticks: insights into the evolution of the families of ticks and of the genus Amblyomma. Ticks Tick Borne Dis 12:101577
Minh BQ, Schmidt HA, Chernomor O, Schrempf D, Woodhams MD, von Haeseler A, Lanfear R (2020) IQ-TREE 2: new models and efficient methods for phylogenetic inference in the genomic era. Mol Biol Evol 37:1530–1534
Morel PC (1965) Description de Haemaphysalis tauffliebi n. sp. d’Afrique centrale. Acarologia 7:281–285
Morel PC, Mouchet J (1958) Les tiques du Cameroun (Ixodidae et Argasidae). Ann Parasitol Hum Comp 33:69–111
Raoult D, Parola P (2007) Rickettsial diseases. Informa Healthcare USA, Inc. pp. 379
Santos Dias JAT (1958) Notes on various ticks (Acarina-Ixodoidea) in collections at some entomological Institutes in Paris and London. An Inst Med Trop 15:459–563
Tomlinson JA, Apanaskevich DA (2019) Two new species of Haemaphysalis Koch, 1844 (Acari: Ixodidae) in the H. (Rhipistoma) spinulosa subgroup, parasites of carnivores and hedgehogs in Africa. Syst Parasitol 96:485–509
Wölfel R, Essbauer S, Dobler G (2008) Diagnostics of tick-borne rickettsioses in Germany: a modern concept for a neglected disease. Int J Med Microbiol 298:368–374
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
A.P. and M.K. collected and provided the tick for investigation. B.M. did the genetic analysis, sequences submission, and prepared Fig. 2. D.A.A. contributed to the morphological identification. L.C.D. did the morphological identification and the further lab work (DNA extraction and all tests) and prepared Fig. 1. A.P., B.M., D.A.A., and L.C.D. wrote the manuscript. All authors read the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
All authors gave their consent for publication.
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Julia Walochnik.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: The authors regret that Figure 1 and Figure 2 were swapped incorrectly in the published version of the article.
The original article has been corrected.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Paguem, A., Mans, B.J., Kingsley, M. et al. Haemaphysalis hoodi (Acari: Ixodidae) on a human from Yaoundé, Cameroon, and its molecular characterization. Parasitol Res 121, 2887–2890 (2022). https://doi.org/10.1007/s00436-022-07613-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-022-07613-5