Abstract
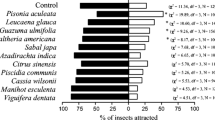
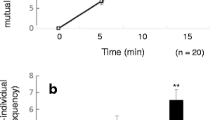
The aim of this study was to identify the aggregation sites and transmission characteristics of Gasterophilus pecorum, the dominant pathogen of endangered equines in desert steppe. Therefore, we tested with a four-arm olfactometer the olfactory response of the G. pecorum adults to the odors that have a great impact on their life cycle, and also investigated the occurrence sites of the adults in the area where the Przewalski’s horse (Equus przewalskii) roam frequently during the peak period of G. pecorum infection. The results of four-directional olfactory test showed that the fresh horse feces had a stronger attraction rate on both male (50.4%) and female flies (38.2%). Stipa caucasica, the only oviposition plant where G. pecorum lay eggs, had a better attraction effect on females than that on males. And the attraction rates of S. caucasica to G. pecorum females in the early growth stage (Stipa I) and mid-growth stage (Stipa II) were 32.8% and 36.8%, respectively. In addition, the two-directional olfactory test showed that the attraction rate of males to fresh horse feces (68.90%) was higher than that to Stipa II (31.10%), and females also showed similar olfactory responses. Moreover, in our field investigation, 68.29% of G. pecorum adults were collected from around the horse feces. The results of laboratory test and field investigation implied that the location mechanism of G. pecorum aggregation for mating is related to the orientation of horse feces. The horse feces and the vicinity are the key contamination areas of G. pecorum, and it is also the areas where horses are seriously infected with G. pecorum. Those fresh feces, which gather abundant information about the host, naturally had the greatest chance of contacting with the host; G. pecorum adults create the opportunity to enter directly into the host’s mouth and infect the host by laying eggs on S. caucasica, which is the most favorite plant of the host in this area. These characteristics are one of the main reasons why G. pecorum has become the dominant species under the condition of sparse vegetation in desert steppe.






Similar content being viewed by others
Abbreviations
- KNR:
-
Kalamaili Ungulate Nature Reserve
- CK:
-
Control check
References
Albuquerque TA, Zurek L (2014) Temporal changes in the bacterial community of animal feces and their correlation with stable fly oviposition, larval development, and adult fitness. Front Microbiol 5:590. https://doi.org/10.3389/fmicb.2014.00590
Brevault T, Quilici S (2010) Flower and fruit volatiles assist host-plant location in the Tomato fruit fly Neoceratitis cyanescens. Physiol Entomol 35:9–18. https://doi.org/10.1111/j.1365-3032.2009.00704.x
Catts EP (1979) Hilltop aggregation and mating behavior by Gasterophilus intestinalis (Diptera: Gasterophilidae). J Med Entomol 16:461–464. https://doi.org/10.1093/jmedent/16.6.461
Cheng F et al (2014) Isolation and characterization of microsatellite loci from Gasterophilus pecorum, (Diptera: Gasterophilidae). Conserv Genet Resour 6:487–489. https://doi.org/10.1007/s12686-013-0136-x
Chu HJ, Jiang ZG, Lan WX, Wang C, Tao YS, Jiang F (2008) Dietary overlap among kulan Equus hemionus, goitered gazelle Gazella subgutturosa and livestock. Acta Zool Sin 54:941–954 (in Chinese with English abstract)
Coapio GG, Cruz-López L, Guerenstein P, Malo EA, Rojas JC (2018) Oviposition preference and larval performance and behavior of Trichoplusia ni (Lepidoptera: Noctuidae) on host and nonhost plants. Arthropod-Plant Inte 12:267–276. https://doi.org/10.1007/s11829-017-9566-9
Cogley TP, Cogley MC (1999) Inter-relationship between Gasterophilus larvae and the horse’s gastric and duodenal wall with special reference to penetration. Vet Parasitol 86:127–142. https://doi.org/10.1016/S0304-4017(99)00119-3
Cogley TP, Cogley MC (2000) Field observations of the host-parasite relationship associated with the common horse bot fly, Gasterophilus intestinalis. Vet Parasitol 88:93–105. https://doi.org/10.1016/S0304-4017(99)00191-0
Colwell DD, Hall MJR, Scholl PJ (2006) The Oestrid flies: biology, host-parasite relationships, impact and management. Oxford University Press, London, UK, pp 140–166
Cope SE, Catts EP (1991) Parahost behavior of adult Gasterophilus intestinalis (Diptera: Gasterophilidae) in Delaware. J Med Entomol 28:67–73. https://doi.org/10.1093/jmedent/28.1.67
Dekker T, Geier M, Cardé RT (2005) Carbon dioxide instantly sensitizes female yellow fever mosquitoes to human skin odours. J Exp Biol 208(15):2963–2972. https://doi.org/10.1242/jeb.01736
Galvez-Marroquin Z, Cruz-López L, Malo EA, Ramsey JM, Rojas JC (2017) Behavioural and electrophysiological responses of Triatoma dimidiata nymphs to conspecific faecal volatiles. Med Vet Entomol 32:102–110. https://doi.org/10.1111/mve.12271
Glasser S, Farzan S (2016) Host-associated volatiles attract parasitoids of a native solitary bee, Osmia lignaria Say (Hymenoptera, Megachilidae). J Hymenopt Res 51:249–2560 10.3897/jhr.51.9727
Gouinguené SP, Turlings TCJ (2002) The effects of abiotic factors on induced volatile emissions in corn plants. Plant Physiol 129:1296–1307. https://doi.org/10.1104/pp.001941
Grenacher S, Kröber T, Guerin PM, Vlimant M (2001) Behavioural and chemoreceptor cell responses of the tick, Ixodes ricinus, to its own faeces and faecal constituents. Exp Appl Acarol 25:641–660. https://doi.org/10.1023/A:1016145805759
Harris MO, Galanihe LD, Sandnayake M (1999) Adult emergence and reproductive behavior of the leafcurling midge Dasineura mali (Diptera: Cecidomyiidae). Ann Entomol Soc Am 92:748–757. https://doi.org/10.1093/aesa/92.5.748
Herrebout WM, Veer J (2010) Habitat selection in Eucarcelia rutilla Vill. (Diptera: Tachinidae). J Appl Entomol 64:55–61. https://doi.org/10.1111/j.1439-0418.1967.tb02071.x
Hoseini SM, Zaheri BA, Adibi MA, Ronaghi H, Moshrefi AH (2017) Histopathological study of esophageal infection with Gasterophilus pecorum (Diptera: Oestridae) in Persian Onager (Equus hemionus onager). J Arthropod Borne Dis 11(3):441–445. https://pubmed.ncbi.nlm.nih.gov/29322061/
Huang HQ, Zhang BR, Chu HJ, Zhang D, Li K (2016) Gasterophilus (Diptera, Gasterophilidae) infection of equids in the Kalamaili Nature Reserve, China. Parasite 23: 36. https://doi.org/10.1051/parasite/2016036
Huang HQ et al (2017) Distribution of Gasterophilus (Diptera: Gasterophilidae) myiasis foci in arid desert steppe: a case study of Kalamaili Mountain Ungulate Nature Reserve. Sci Silvae Sin 53:142–149 (in Chinese with English abstract)
Huang HQ et al (2021) Spatial distribution of Gasterophilus pecorum (Diptera) eggs in the desert steppe of the Kalamaili Nature Reserve (Xinjiang, China). BMC Ecol Evo 21:169. https://doi.org/10.1186/s12862-021-01897-4
Huang HQ et al (2022) Analysis on the relationship between winter precipitation and the annual variation of horse stomach fly community in arid desert steppe, Northwest China (2007–2019). Integr Zool 17(1):128–138. https://doi.org/10.1111/1749-4877.12578
Ibrayev B, Lider L, Bauer C (2015) Gasterophilus spp. infections in horses from northern and central Kazakhstan. Vet Parasitol 207:94–98. https://doi.org/10.1016/j.vetpar.2014.11.015
Jost DJ, Pitre HN (2002) Soybean looper (Lepidoptera: Noctuidae) oviposition on cotton and soybean of different growth stages: influence of olfactory stimuli. J Econ Entomol 95:286–293. https://doi.org/10.1603/0022-0493-95.2.286
Kolosova N, Gorenstein N, Kish CM, Dudareva N (2001) Regulation of circadian methyl benzoate emission in diurnally and nocturnally emitting plants. Plant Cell 13:2333–2347. https://doi.org/10.1105/tpc.010162
Krecek RC, Reinecke RK, Horak IG (1989) Internal parasites of horses on mixed grassveld and bushveld in Transval, Republic of South Africa. Vet Parasitol 34:135–143. https://doi.org/10.1016/0304-4017(89)90173-8
Li XY, Chen YO, Wang QK, Li K, Pape T, Zhang D (2018) Molecular and morphological characterization of third instar Palaearctic horse stomach bot fly larvae (Oestridae: Gasterophilinae, Gasterophilus). Vet Parasitol 262:56–74. https://doi.org/10.1016/j.vetpar.2018.09.011
Li XY, Pape T, Zhang D (2019) Taxonomic review of Gasterophilus (Oestridae, Gasterophilinae) of the world, with updated nomenclature, keys, biological notes and distributions. ZooKeys 891:119–156. https://pubmed.ncbi.nlm.nih.gov/31802974/
Liu SH, Hu DF, Li K (2015) Oviposition site selection by Gasterophilus pecorum (Diptera: Gasterophilidae) in its habitat in Kalamaili Nature Reserve, Xinjiang, China. Parasite 22:34. https://doi.org/10.1051/parasite/2015034
Liu SH, Li K, Hu DF (2016) The incidence and species composition of Gasterophilus (Diptera, Gasterophilidae) causing equine myiasis in northern Xinjiang, China. Vet Parasitol 217:36–38. https://doi.org/10.1051/parasite/2015034
Lyons ET, Tolliver SC, Collins SS (2006) Prevalence of large endoparasites at necropsy in horses infected with Population B small strongyles in a herd established in Kentucky in 1966. Parasitol Res 99:114–118. https://doi.org/10.1007/s00436-005-0116-5
Mauchline AL, Osborne JL, Martin AP, Poppy GM, Powell W (2005) The effects of non-host plant essential oil volatiles on the behaviour of the pollen beetle Meligethes aeneus. Entomol Exp Appl 114:181–188. https://doi.org/10.1111/j.1570-7458.2005.00237.x
Moshaverinia A, Baratpour A, Abedi V, Mohammadi-Yekta M (2016) Gasterophilosis in Turkmen horses caused by Gasterophilus pecorum (Diptera, Oestridae). Sci Parasitol 17:49–52. https://profdoc.um.ac.ir/paper-abstract-1057918.html
Mukbel R, Torgerson PR, Abo-Shehada M (2001) Seasonal variations in the abundance of Gasterophilus spp. larvae in donkeys in northern Jordan. Trop Anim Health Prod 33:501–509. https://doi.org/10.1023/A:1012732613902
Natale D, Mattiacci L, Pasqualini E, Dorn D (2004) Apple and peach fruit volatiles and the apple constituent butyl hexanoate attract female oriental fruit moth, Cydia molesta, in the laboratory. J Appl Entomol 128(1):22–27. https://doi.org/10.1046/j.1439-0418.2003.00802.x
Olival KJ et al (2013) Lack of population genetic structure and host specificity in the bat fly, Cyclopodia horsfieldi, across species of Pteropus bats in Southeast Asia. Parasit Vectors 6:231. https://doi.org/10.1186/1756-3305-6-231
Otranto D, Milillo P, Capelli G, Colwell DD (2005) Species composition of Gasterophilus spp. (Diptera: Oestridae) causing equine gastric myiasis in southern Italy: parasite biodiversity and risks for extinction. Vet Parasitol 133:111–118. https://doi.org/10.1016/j.vetpar.2005.05.015
Pacheco A, Blanco-Metzler H, Mora R (2012) Modification of four-arm olfactometers to be used with the coffee borer Hypothenemus hampei Ferrari (Coleoptera: Curculionidae). Agron Costarric 6:69–78 (in Spanish with English abstract)
Pawlas M, Sołtysiak Z, Nicpoń J (2007) Existence and pathomorhological picture of gasterophilosis in horses from north-east Poland. Med Weter 63:1377–1380. https://www.cabdirect.org/cabdirect/abstract/20073258397
Peñaflor MFGV, Erb M, Miranda LA, Werneburg AG, Bento JMS (2011) Herbivore-induced plant volatiles can serve as host location cues for a generalist and a specialist egg parasitoid. J Chem Ecol 37:1304–1313. https://doi.org/10.1007/s10886-011-0047-9
Piñero JC, Dorn S (2009) Response of female oriental fruit moth to volatiles from apple and peach trees at three phenological stages. Entomol Exp Appl 131:67–74. https://doi.org/10.1111/j.1570-7458.2009.00832.x
Ranjith AM (2007) An inexpensive olfactometer and wind tunnel for Trichogramma chilonis Ishii (Trichogrammatidae: Hymenoptera). J Trop Agric 45:63–65. https://www.mendeley.com/catalogue/7566d466-32a9-3e67-a96e-f3676b5ee5e9/
Rastegaev YM (1985) Ecological characteristics of the warble and bot flies attacking horses (Diptera: Oestridae, Gasterophilidae) in the desert zone of the Caspian Region. Entomol Rev 64:35–39. http://pascal-francis.inist.fr/vibad/index.php?action=getRecordDetail&idt=8455857
Rehbein S, Visser M, Winter R (2013) Prevalence, intensity and seasonality of gastrointestinal parasites in abattoir horses in Germany. Parasitol Res 112:407–413. https://doi.org/10.1007/s00436-012-3150-0
Reshetnikov AD, Barashkova AI, Prokopyev ZS (2014) Infestation of horses by the causative agents of gasterophilosis (Diptera: Gasterophilidae): the species composition and the north-eastern border of the area in the Republic (Sakha) of Yakutia of the Russian Federation. Life Sci J 11:587–590. https://doi.org/10.7537/marslsj111114.105
Robinson A et al (2020) Responses of the putative trachoma vector, Musca sorbens, to volatile semiochemicals from human faeces. PLoS Neglected Trop Dis 14:1–15. https://doi.org/10.1371/journal.pntd.0007719
Salerno G, Frati F, Conti E, Peri E, Colazza S, Cusumano A (2019) Mating status of an herbivorous stink bug female affects the emission of oviposition-induced plant volatiles exploited by an egg parasitoid. Front Physiol 10:398. https://doi.org/10.3389/fphys.2019.00398
Uefune M, Shiojiri K, Takabayashi J (2017) Oviposition of diamondback moth Plutella xylostella females is affected by herbivore-induced plant volatiles that attract the larval parasitoid Cotesia vestalis. Arthropod-Plant Inte 11:235–239. https://doi.org/10.1007/s11829-016-9484-2
Wang WT et al (2014) Population genetic structure of Gasterophilus pecorum in the Kalamaili Nature Reserve, Xinjiang, based on mitochondrial cytochrome oxidase (COI) gene sequence. Med Vet Entomol 28:75–82. https://doi.org/10.1111/mve.12073
Wang WT et al (2016) Diversity and Infection of Gasterophilus spp. in Mongol-Xinjiang Region and Qinghai Tibet Region. Sci Silvae Sin 52:134–139 (in Chinese with English abstract)
Wei JQ, Mo JC, Wang XJ, Lin W (2008) Responses of housefly adults to different food. Chin J Vector Biol Control 19:12–13 (in Chinese with English abstract)
Witzgall P, Stelinski L, Gut L, Thomson D (2008) Codling moth management and chemical ecology. Annu Rev Entomol 53(1):503–522. https://www.annualreviews.org/doi/10.1146/annurev.ento.53.103106.093323
Zang S et al (2017) Food patch particularity and forging strategy of reintroduced Przewalski's horse in North Xinjiang, China. Turk J Zool 41:924–930. https://doi.org/10.3906/zoo-1509-9
Zhang K et al (2021) The impact of temperature on the life cycle of Gasterophilus pecorum in northwest China. Parasit Vectors 14:129. https://doi.org/10.1186/s13071-021-04623-7
Zhang YJ et al (2015) Water use patterns of sympatric Przewalski’s horse and Khulan: interspecific comparison reveals niche differences. PLoS One 10:e0132094. https://doi.org/10.1371/journal.pone.0132094
Zhou R et al (2020) Identification of volatile components from oviposition and non-oviposition plants of Gasterophilus pecorum (Diptera: Gasterophilidae). Sci Rep 10:15731. https://doi.org/10.1038/s41598-020-72378-9
Zhou R et al (2021) Analysis of volatiles from feces of released Przewalski’s horse (Equus przewalskii) in Gasterophilus pecorum (Diptera: Gasterophilidae) spawning habitat. Sci Rep 11:15671. https://doi.org/10.1038/s41598-021-95162-9
Zong XL et al (2020) Ecology of bat flies in Singapore: a study on the diversity, infestation bias and host specificity (Diptera: Nycteribiidae). Int J Parasitol-Par 12:29–33. https://doi.org/10.1016/j.ijppaw.2020.04.010
Acknowledgements
We are grateful to the staff of the Kalamaili Ungulate Nature Reserve for their support and valuable technical assistance.
Funding
This work was supported by the National Science Foundation of China (No. 31670538), the Species Project (No. 2018123) of Department for Wildlife and Forest Plants Protection, NFGA of China, and the Forestry Fund of LiBin (01210823).
Author information
Authors and Affiliations
Contributions
K. L. and K. Z. conceived the study; K. Z., R. Z., and H. Q. H. conducted the experiment; K. Z., W. M., Y. J. Q., and B. L. L. carried out the statistics; K. Z. drafted the manuscript; D. Z., K. L., and H. J. C revised the manuscript. All authors reviewed the manuscript and approved the final version.
Corresponding authors
Ethics declarations
Ethics approval
The sample collection and field investigation were approved by the management department of Kalamaili Ungulate Nature Reserve. The study was performed in accordance with the relevant guidelines and regulations regarding animal welfare. All experimental protocols were approved by the Ethic and Animal Welfare Committee, Beijing Forestry University (EAWC-BJFU-2018015).
Conflict of interest
The authors declare no competing interests.
Additional information
Section Editor: Boris Krasnov
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, K., Zhou, R., Huang, H. et al. Host feces, olfactory beacon guiding aggregation of intestinal parasites Gasterophilus pecorum (Diptera: Gasterophilidae). Parasitol Res 121, 2601–2613 (2022). https://doi.org/10.1007/s00436-022-07577-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-022-07577-6