Abstract
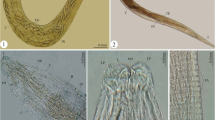
Philophthalmus is a cosmopolitan genus of digeneans that includes ocular parasites of birds and mammals. Despite broad distribution and veterinary importance of these digeneans, there are still gaps in knowledge about their diversity and biology, especially in South America. Herein, we conducted morphological, life cycle, and molecular studies of megalurous cercariae found in aquatic gastropod molluscs Aylacostoma chloroticum and A. tuberculatum collected in the São Francisco River, Brazil. Adult parasites reared experimentally in the eyes of chicks are described here as Philophthalmus aylacostoma n. sp. The new species differs from its congeners known in the Americas by a combination of traits, including the sucker width ratio, the oral sucker to pharynx width ratio, egg size, and the type of vitellarium in adult forms. The new species is morphologically closest to Philophthalmus megalurus, from which it differs by the smaller body and larger eggs, as well as by the measurements of cercariae and the family of snails that act as the intermediate host. Molecular phylogenetic analysis based on 28S rDNA and comparison of cox1 sequences confirm that P. aylacostoma n. sp. is distinct from four previously sequenced named species of the genus. Moreover, cox1 sequences revealed conspecificity of our specimens with an isolate of Philophthalmus sp. previously reported, also in thiarid snails, in Paraná River, Brazil. The interspecific divergence in cox1 between the new species and other species with sequences available for comparison varied between 12 and 15%.




Similar content being viewed by others
References
Alicata JE (1962) Life cycle and developmental stages of Philophthalmus gralli in the intermediate and final hosts. J Parasitol 48:47–54
Bennett J, Presswell B (2019) Morphology and molecules resolve the identity and life cycle of an eye trematode, Philophthalmus attenuatus n. sp. (Trematoda: Philophthalmidae) infecting gulls in New Zealand. Parasitol Res 118:1501–1509. https://doi.org/10.1007/s00436-019-06289-8
Born-Torrijos A, Poulin R, Raga JA, Holzer AS (2014) Estimating trematode prevalence in snail hosts using a single-step duplex PCR: how badly does cercarial shedding underestimate infections rates? Parasit Vectors 7:243. https://doi.org/10.1186/1756-3305-7-243
Bowles J, Hope M, Tiu WU, Liu X, McManus DP (1993) Nuclear and mitochondrial genetic markers highly conserved between Chinese and Philippine Schistosoma japonicum. Acta Trop 55:217–229. https://doi.org/10.1016/0001-706X(93)90079-Q
Chalkowski K, Morgan A, Lepczyk CA, Zohdy S (2021) Spread of an avian eye fluke, Philophthalmus gralli, through biological invasion of an intermediate host. J Parasitol 107:336–348. https://doi.org/10.1645/20-72
Cheng TC, Thakur AS (1967) Thermal activation and inactivation of Philophthalmus gralli metacercariae. J Parasitol 53:212–213
Ching HL (1961) The development and morphological variation of Philophthalmus gralli Mathis & Leger, 1910 with a comparison of species of Philophthalmus Looss, 1899. Proc Helminthol Soc Wash 28:130–138
Coelho PN, Fernandez MA, Cesar DAS, Ruocco AMC, Henry R (2018) Updated distribution and range expansion of the gastropod invader Melanoides tuberculata (Müller, 1774) in Brazilian waters. BioInvasions Rec 7:405–409. https://doi.org/10.3391/bir.2018.7.4.08
Díaz MT, Hernández LE, Bashirullah AK (2002) Experimental life cycle of Philophthalmus gralli (Trematoda: Philophthalmidae) in Venezuela. Rev Biol Trop 50:629–641
Dronen NO, Fried B (2008) Comparative study of the ages classes two species of Philophthalmus Looss, 1899 (Philophthalmidae: Philophthalminae). Comp Parasitol 75:12–23. https://doi.org/10.1654/4307.1
Freitas JFT (1955) Sobre dois trematódeos parasitos de aves: Philophthalmum lachrymosus Braun, 1902 e Renicola mirandaribeiroi n. sp. Arq Mus Nac 42:585–610
Heneberg P, Casero M, Waap H, Sitko J, Azevedo F, Těěínský (2018) An outbreak of philophthalmosis in Larus michahellis and Larus fuscus gulls in Iberian Peninsula. Parasitol Int 67:253–261. https://doi.org/10.1016/j.parint.2017.12.007
Huffman JE, Fried B (1983) Trematodes from Goniobasis virginica (Gastropoda: Pleuroceridae) in Lake Musconetcong, New Jersey. J Parasitol 69:429. https://doi.org/10.2307/3281254
Kanev I, Nollen PM, Vissilev I, Dimitrov V (1993) Redescription of Philophthalmus lucipetus (Rudolphi, 1819) (Trematoda: Philophthalmidae) with a discussion of its identity and characteristics. Ann Naturhist Mus Wien 94(95B):11–34
Kanev I, Radev V, Fried B (2005) Family Philophthalmidae Looss, 1899. In: Jones A, Bray RA, Gibson DI (eds) Keys to the Trematoda, 2nd edn. CAB International, Wallingford, pp 87–97
Keeney DB, King TM, Rowe DL, Poulin R (2009) Contrasting mtDNA diversity and population structure in a direct-developing marine gastropod and its trematode parasites. Mol Ecol 22:4591–4603. https://doi.org/10.1111/j.1365-294X.2009.04388.x
Krygier BB, Macy RW (1969) The eye fluke Philophthalmus megalurus (Cort) (Trematoda: Philophthalmidae) in the Dipper, Cinclus mexicanus, in Oregon. J Parasitol 55:78
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874. https://doi.org/10.1093/molbev/msw054
Lamothe-Argumedo R, Díaz-Camacho SP, Nawa Y (2003) The first human case in Mexico of conjunctivitis caused by the avian parasite, Philophthalmus lacrimosus. J Parasitol 89:183–185. https://doi.org/10.1645/0022-3395(2003)089[0183:TFHCIM]2.0.CO;2
Leung TLF, Donald KM, Keeney DB, Koehler AV, Peoples RC, Poulin R (2009) Trematode parasites of Otago Harbour (New Zealand) soft-sediment intertidal ecosystems: life cycles, ecological roles and DNA barcodes, New Zeal. J Mar Fresh Res 43:857–865
Literák I, Heneberg P, Sitko J, Wetzel EJ, Callirgos JM, Čapek M, Basto DV, Papoušek (2013) Eye trematode infection in small passerines in Peru caused by Philophthalmus lucipetus, an agent with a zoonotic potential spread by an invasive freshwater snail. Parasitol Int 62:390–396. https://doi.org/10.1016/j.parint.2013.04.001
McMillan TA (1971) Life history of Philophthalmus megalurus (Cort, 1914) in Western Oregon. Dissertation, Portland State University
McMillan TA, Macy RW (1972) The life cycle of Philophthalmus megalurus (Cort, 1914) (Trematoda: Philophthalmidae) in Western Oregon. J Parasitol 58:22
Miura O, Kuris AM, Torchin ME, Hechinger RF, Dunham EJ, Chiba S (2005) Molecular-genetic analyses reveal cryptic species of trematodes in the intertidal gastropod, Batillaria cumingi (Crosse). Int J Parasitol 35:793–801. https://doi.org/10.1016/j.ijpara.2005.02.014
Muniz-Pereira LC, Amato SB (1993) Philophthalmus gralli (Digenea: Philophthalmidae) parasite of Anas bahamensis and Amazonetta brasiliensis, from lagoons of Maricá county, Rio de Janeiro, Brazil. Mem Inst Oswaldo Cruz 88:567–569. https://doi.org/10.1590/S0074-02761993000400012
Nasir P, Díaz T (1972) Avian flukes of Venezuela. Riv Parassitol 33:245–276
Nollen PM (1983) The effects of crowding on adults of Philophthalmus gralli (Trematoda) grown in chickens. J Parasitol 69:196–199. https://doi.org/10.2307/3281298
Nollen PM, Kanev I (1995) The taxonomy and biology of philophthalmid eyeflukes. Adv Parasitol 36:205–269. https://doi.org/10.1016/S0065-308X(08)60492-3
Onaca FMT, da Graça RJ, Fabrin TMC, Takemoto RM, Oliveira AV (2020) Molecular characterization and identification of digenean larval stages in Aylacostoma chloroticum (Prosobranchia: Thiaridae) from a neotropical floodplain. J Helminthol 94:e73. https://doi.org/10.1017/s0022149x19000725
Ostrowski de Núñez M, Quintana MG (2008) The life cycle of Stephanoprora aylacostoma n. sp. (Digenea: Echinostomatidae), parasite of the threatened snail Aylacostoma chloroticum (Prosobranchia, Thiaridae), in Argentina. Parasitol Res 102:647–655. https://doi.org/10.1007/s00436-007-0806-2
Ostrowski de Núñez M, Quintana MG, Laczkó ACM (2020) The life cycle of Heterophyes yacyretana n. sp. (Digenea, Heterophyidae), parasitic in the endemic snail Aylacostoma chloroticum (Caenogastropoda, Thiaridae) in Argentina. J Parasitol 106:625–632. https://doi.org/10.1645/19-168
Penner LR, Fried B (1963) Philophthalmus hegeneri sp. n., an ocular trematode from birds. J Parasitol 49:974–977. https://doi.org/10.2307/3275734
Pinto HA, Melo AL (2010) Melanoides tuberculata as intermediate host of Philophthalmus gralli in Brazil. Rev Inst Med Trop Sao Paulo 52:323–327. https://doi.org/10.1590/S0036-46652010000600007
Preston DL, Layden TJ, Segui LM, Falke LP, Brant SV, Novak M (2021) Trematode parasites exceed aquatic insect biomass in Oregon stream food webs. J Anim Ecol 90:766–755. https://doi.org/10.1111/1365-2656.13409
Quintana MG, Ostrowski de Núñez O (2014) The life cycle of Pseudosellacotyla lutzi (Digenea: Cryptogonomidae), in Aylacostoma chloroticum (Prosobranchia: Thiaridae), and Hoplias malabaricus (Characiformes: Erythrinidae), in Argentina. J Parasitol 100:805–811. https://doi.org/10.1645/13-379.1
Quintana MG, Ostrowski de Núñez M (2016) The life cycle of Neocladocystis intestinalis (Vaz, 1932) (Digenea: Cryptogonimidae), in Aylacostoma chloroticum (Prosobranchia: Thiaridae), and Salminus brasiliensis (Characiformes: Characidae), in Argentina. Parasitol Res 115:2589–2595. https://doi.org/10.1007/s00436-016-5004-7
Radev V, Kanev I, Nollen PM, Gold D (1999) Life history and identification of Philophthalmus lucipetus from Israel. J Parasitol 85:291–294. https://doi.org/10.2307/3285636
Radev V, Kanev I, Gold D (2000) Life cycle and identification of an eyefluke from Israel transmitted by Melanoides tuberculata. J Parasitol 86:773–776. https://doi.org/10.1645/0022-3395(2000)086[0773:LCAIOA]2.0.CO;2
Ronquist F, Teslenko M, van der Mark P, Ayres DL, Hohna S, Larget B, Liu L, Suchard MA, Huelsenbeck JP (2012) MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61:539–542. https://doi.org/10.1093/sysbio/sys029
Schunck F, Piacentini VQ, Souza EA, Sousa AEBA, Rego MA, Albano C, Nunes MFC, Favaro FL, Neto IS, Mariano EF, Lima DM, Las-Casas FMG, Rodrigues RC, Neto FPF (2012) Birds of the Lower Middle São Francisco River. Rev Bras Ornitol 20:312–328
Simone LRL (2006) Land and Freshwater Molluscs of Brazil: an illustrated inventory on the Brazilian malacofauna, including neighbor regions of the South America, respect to the terrestrial and freshwater ecosystems. EGB FAPESP, São Paulo
Tkach VV, Littlewood DTJ, Olson PD, Kinsella JM, Swiderski (2003) Molecular phylogenetic analysis of the Microphalloidea Ward, 1901 (Trematoda: Digenea). Syst Parasitol 56:1–15. https://doi.org/10.1023/A:1025546001611
Tkach VV, Kudlai O, Kostadinova A (2016) Molecular phylogeny and systematics of the Echinostomatoidea Looss, 1899 (Platyhelminthes: Digenea). Int J Parasitol 46:171–185
Vogler RE (2012) Aylacostoma chloroticum Hylton Scott, 1954: antecedentes de la especie. Amici Molluscarum 20:43–46
West (1961) Studies on the biology of Philophthalmus gralli Mathis and Leger, 1910 (Trematoda: Digenea). Am Midl Nat 66:363–383. https://doi.org/10.2307/2423036
Acknowledgements
We thank the members of the Center of Advanced Treatment and Research on Tegumentary Leishmaniasis, Januária City, especially Jailton Xavier and Prof. Stefan Geiger, for the laboratory infrastructure facilities used during the malacological surveys. We are indebted to Dr. Monica Fernandez and Dr. Silvana Thiengo (Instituto Oswaldo Cruz, Rio de Janeiro), and Dr Luiz Simone (Universidade de São Paulo) for their help in the taxonomic identification of molluscs. This work was also supported by National Council for Scientific and Technological Development (CNPq), Brazil (research scholarship to HAP and doctoral scholarship to EAPM), and U. S. National Science Foundation grants DEB-1120734 to VVT.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
Applicable national and/or institutional guidelines for the care and use of animals were followed in all procedures performed.
Conflict of interest
The authors declare no competing interests.
Additional information
Section Editor: Konstans Wells
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pulido-Murillo, E.A., Tkach, V.V. & Pinto, H.A. The life cycle of Philophthalmus aylacostoma n. sp. (Trematoda: Philophthalmidae), a new eye fluke species transmitted by Aylacostoma spp. (Gastropoda: Thiaridae) in Brazil. Parasitol Res 121, 933–944 (2022). https://doi.org/10.1007/s00436-022-07447-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-022-07447-1