Abstract
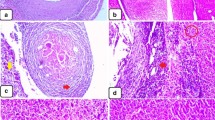
To enhance the therapeutic effects of albendazole (ABZ) on Echinococcus granulosus protoscoleces and metacestodes, ABZ-loaded nanostructured lipid carriers (ABZ–NLCs) are prepared by the hot high-speed homogenization method. Protoscoleces and microcysts were treated in vitro with free ABZ and ABZ–NLCs (concentrations of 1, 5, and 10 μg/ml), and the corresponding effects were monitored by methylene blue exclusion test and scanning and transmission electron microscopy. Chemoprophylactic treatment was performed on Balb/C mice 1 day before intraperitoneal injection of viable protoscoleces. The drugs were administered daily by intragastric inoculation for a period of 30 days. The prophylactic efficacy was assessed based on the number and weight of cysts developed in treated mice. The ultrastructural alterations in cysts were examined by transmission electron microscopy. After 18 days, all the protoscoleces incubated with 10 μg/ml ABZ–NLCs were killed, while 51.25 ± 4.03% of the protoscoleces incubated with 10 μg/ml free ABZ were still viable. Microcysts treated with ABZ–NLCs underwent degenerative alterations in a shorter time than when free ABZ was applied. The mean weight of the cysts recovered from mice of ABZ–NLCs group was significantly lower than that of the free ABZ group (P < 0.05), yielding prophylactic efficacy of 92.45% and 38.53%, respectively. The cysts treated with ABZ–NLCs showed marked ultrastructural changes in the germinal layer. This study demonstrated that both in vitro and in vivo treatments with ABZ–NLCs are significantly more efficient than treatment with free ABZ against E. granulosus protoscoleces, metacestodes, and prevention of cyst development in mice.





Similar content being viewed by others
References
Ahmadi N, Rostamizadeh K, Modarresi-Alam AR (2018) therapeutic anti-inflammatory potential of different formulations based on coenzyme Q10-loaded nanostructured lipid carrier: in vitro, ex vivo, and in vivo evaluations. Eur J Lipid Sci Technol 120:1800232
Ahmadnia S, Moazeni M, Mohammadi-Samani S, Oryan A (2013) In vivo evaluation of the efficacy of albendazole sulfoxide and albendazole sulfoxide loaded solid lipid nanoparticles against hydatid cyst. Exp Parasitol 135:314–319
Albanese A, Tang PS, Chan WC (2012) The effect of nanoparticle size, shape, and surface chemistry on biological systems. Annu Rev Biomed Eng 14:1–16
Bhatt A, Pethe A (2010) Nanotechnology: a promising drug delivery for poorly water soluble drugs. J Pharm Res 3:1748–1751
Casado N, Pérez-Serrano J, Denegri G, Rodriguez-Caabeiro F (1996) Development of a chemotherapeutic model for the in vitro screening of drugs against Echinococcus granulosus cysts: the effects of an albendazole-albendazole sulphoxide combination. Int J Parasitol 26:59–65
Casado N, Urrea-París M, Moreno M, Rodriguez-Caabeiro F (2001) Combined praziquantel and albendazole chemoprophylaxis in experimental hydatidosis. Parasitol Res 87:787–789
Casado N, Moreno M, Urrea-París M, Rodriguez-Caabeiro F (2002) Could ivermectin have a synergic effect with albendazole in hydatidosis therapy? Parasitol Res 88:153–159
Ceballos L, Elissondo C, Bruni SS, Confalonieri A, Denegri G, Alvarez L, Lanusse C (2010) Chemoprophylactic activity of flubendazole in cystic echinococcosis. Chemotherapy 56:386–392
Ceballos L, Elissondo C, Bruni SS, Denegri G, Lanusse C, Alvarez L (2011) Comparative performances of flubendazole and albendazole in cystic echinococcosis: ex vivo activity, plasma/cyst disposition, and efficacy in infected mice. Antimicrob Agents Chemother 55:5861–5867
Craig PS, McManus DP, Lightowlers MW, Chabalgoity JA, Garcia HH, Gavidia CM, Gilman RH, Gonzalez AE, Lorca M, Naquira C (2007) Prevention and control of cystic echinococcosis. Lancet Infect Dis 7:385–394
Daniel-Mwambete K, Torrado S, Cuesta-Bandera C, Ponce-Gordo F, Torrado J (2004) The effect of solubilization on the oral bioavailability of three benzimidazole carbamate drugs. Int J Pharm 272:29–36
Das S, Ng WK, Tan RB (2012) Are nanostructured lipid carriers (NLCs) better than solid lipid nanoparticles (SLNs): development, characterizations and comparative evaluations of clotrimazole-loaded SLNs and NLCs? Eur J Pharm 47:139–151
Eckert J, Deplazes P (2004) Biological, epidemiological, and clinical aspects of echinococcosis, a zoonosis of increasing concern. Clin Microbiol Rev 17:107–135
Elissondo M, Dopchiz M, Brasesco M, Denegri G (2004) Echinococcus granulosus: first report of microcysts formation from protoscoleces of cattle origin using the in vitro vesicular culture technique. Parasite 11:415–418
Elissondo M, Dopchiz M, Ceballos L, Alvarez L, Bruni SS, Lanusse C, Denegri G (2006) In vitro effects of flubendazole on Echinococcus granulosus protoscoleces. Parasitol Res 98:317–323
Elissondo M, Ceballos L, Dopchiz M, Andresiuk V, Alvarez L, Bruni SS, Lanusse C, Denegri G (2007) In vitro and in vivo effects of flubendazole on Echinococcus granulosus metacestodes. Parasitol Res 100:1003–1009
Elissondo MC, Ceballos L, Alvarez L, Bruni SS, Lanusse C, Denegri G (2009) Flubendazole and ivermectin in vitro combination therapy produces a marked effect on Echinococcus granulosus protoscoleces and metacestodes. Parasitol Res 105:835
Farhadi M, Fazaeli A, Haniloo A (2015) Genetic characterization of livestock and human hydatid cyst isolates from northwest Iran, using the mitochondrial cox1 gene sequence. Parasitol Res 114:4363–4370
Farhadi M, Haniloo A, Rostamizadeh K, Faghihzadeh S (2018) Efficiency of flubendazole-loaded mPEG-PCL nanoparticles: a promising formulation against the protoscoleces and cysts of Echinococcus granulosus. Acta Trop 187:190–200
Freitas C, Müller RH (1998) Effect of light and temperature on zeta potential and physical stability in solid lipid nanoparticle (SLNTM) dispersions. Int J Pharm 168:221–229
Gamboa GVU, Pensel PE, Elissondo MC, Bruni SFS, Benoit J-P, Palma SD, Allemandi DA (2019) Albendazole-lipid nanocapsules: optimization, characterization and chemoprophylactic efficacy in mice infected with Echinococcus granulosus. Exp Parasitol 198:79–86
Hemphill A, Spicher M, Stadelmann B, Müller J, Naguleswaran A, Gottstein B, Walker M (2007) Innovative chemotherapeutical treatment options for alveolar and cystic echinococcosis. Parasitology 134:1657–1670
Iqbal MA, Md S, Sahni JK, Baboota S, Dang S, Ali J (2012) Nanostructured lipid carriers system: recent advances in drug delivery. J Drug Target 20:813–830
Jelowdar A, Rafiei A, Abbaspour MR, Rashidi I, Rahdar M (2017) Efficacy of combined albendazol and praziquntel and their loaded solid lipid nanoparticles components in chemoprophylaxis of experimental hydatidosis. Asian Pac J Trop Biomed 7:549–554
Kaur IP, Bhandari R, Bhandari S, Kakkar V (2008) Potential of solid lipid nanoparticles in brain targeting. J Control Release 127:97–109
Liu C, Zhang H, Yin J, Hu W (2015) In vivo and in vitro efficacies of mebendazole, mefloquine and nitazoxanide against cyst echinococcosis. Parasitol Res 114:2213–2222
Marcato PD, Durán N (2008) New aspects of nanopharmaceutical delivery systems. J Nanosci Nanotechnol 8:2216–2229
Moreno M, Urrea-Paris M, Casado N, Rodriguez-Caabeiro F (2001) Praziquantel and albendazole in the combined treatment of experimental hydatid disease. Parasitol Res 87:235–238
Moreno M, Casado N, Urrea-París M, Rodríguez-Caabeiro F (2002) Could ivermectin have a synergic effect with albendazole in hydatidosis therapy? Parasitol Res 88:563–567
Moreno-Esparza R, Jung H, Medina L, García L, Fuentes I (1998) Absorption studies of albendazole and some physicochemical properties of the drug and its metabolite albendazole sulphoxide. J Pharm Pharmacol 50:43–48
Moro P, Schantz PM (2009) Echinococcosis: a review. Int J Infect Dis 13:125–133
Naguleswaran A, Spicher M, Vonlaufen N, Ortega-Mora LM, Torgerson P, Gottstein B, Hemphill A (2006) In vitro metacestodicidal activities of genistein and other isoflavones against Echinococcus multilocularis and Echinococcus granulosus. Antimicrob Agents Chemother 50:3770–3778
Patra JK, Das G, Fraceto LF, Campos EVR, del Pilar R-T, Acosta-Torres LS, Diaz-Torres LA, Grillo R, Swamy MK, Sharma S (2018) Nano based drug delivery systems: recent developments and future prospects. J Nanobiotechnol 16:71
Patzschke K, Pütter J, Wegner L, Horster F, Diekmann H (1979) Serum concentrations and renal excretion in humans after oral administration of praziquantel—results of three determination methods—. Eur J Drug Metab Pharmacokinet 4:149–156
Pensel PE, Castro S, Allemandi D, Bruni SS, Palma SD, Elissondo MC (2014) Enhanced chemoprophylactic and clinical efficacy of albendazole formulated as solid dispersions in experimental cystic echinococcosis. Vet Parasitol 203:80–86
Pensel PE, Gamboa GU, Fabbri J, Ceballos L, Bruni SS, Alvarez LI, Allemandi D, Benoit JP, Palma SD, Elissondo MC (2015) Cystic echinococcosis therapy: albendazole-loaded lipid nanocapsules enhance the oral bioavailability and efficacy in experimentally infected mice. Acta Trop 152:185–194
Pérez-Serrano J, Casado N, Rodriguez-Caabeiro F (1994) The effects of albendazole and albendazole sulphoxide combination-therapy on Echinococcus granulosus in vitro. Int J Parasitol 24:219–224
Pérez-Serrano J, Denegri G, Casado N, Rodríguez-Caabeiro F (1997) In vivo effect of oral albendazole and albendazole sulphoxide on development of secondary echinococcosis in mice. Int J Parasitol 27:1341–1345
Rafiei A, Pipelzadeh MH, Jahanshahi A, Salim MRE (2009) Comparing the effectiveness of albendazole and combination of albendazole and praziquantel in experimental hydatidosis. Arch Clin Infect Dis 4:9–12
Sahni JK, Baboota S, Ali J (2011) Promising role of nanopharmaceuticals in drug delivery. Pharm times 43:16–18
Sangsen Y, Laochai P, Chotsathidchai P, Wiwattanapatapee R (2015) Effect of solid lipid and liquid oil ratios on properties of nanostructured lipid carriers for oral curcumin delivery. Adv Mat Res 1060. Trans Tech Publ, p 62–65
Smyth J, Howkins A, Barton M (1966) Factors controlling the differentiation of the hydatid organism, Echinococcus granulosus, into cystic or strobilar stages in vitro. Nature 211:1374–1377
Stojkovic M, Zwahlen M, Teggi A, Vutova K, Cretu CM, Virdone R, Nicolaidou P, Cobanoglu N, Junghanss T (2009) Treatment response of cystic echinococcosis to benzimidazoles: a systematic review. PLoS Negl Trop Dis 3:e524
Torabi N, Dobakhti F, Faghihzadeh S, Haniloo A (2018) In vitro and in vivo effects of chitosan-praziquantel and chitosan-albendazole nanoparticles on Echinococcus granulosus Metacestodes. Parasitol Res 117:1–9
Urrea-Paris M, Moreno M, Casado N, Rodriguez-Caabeiro F (2000) In vitro effect of praziquantel and albendazole combination therapy on the larval stage of Echinococcus granulosus. Parasitol Res 86:957–964
Vitorino C, Carvalho FA, Almeida AJ, Sousa JJ, Pais AA (2011) The size of solid lipid nanoparticles: an interpretation from experimental design. Colloids Surf B 84:117–130
Acknowledgements
We would like to extend our gratitude to Dr. Pirhajati for his helpful comments on transmission electron microscopy analysis.
Funding
This research was financially supported by a grant (no. A-12–95-11) from the Research Vice Chancellery of Zanjan University of Medical Sciences (ZUMS).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
This research was approved by ethics committee of ZUMS (Code: IR.ZUMS.REC.1396.314).
Conflict of interest
The authors declare no competing interests.
Additional information
Section Editor: Christoph G. Grevelding
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Farhadi, M., Haniloo, A., Rostamizadeh, K. et al. In vitro evaluation of albendazole-loaded nanostructured lipid carriers on Echinococcus granulosus microcysts and their prophylactic efficacy on experimental secondary hydatidosis. Parasitol Res 120, 4049–4060 (2021). https://doi.org/10.1007/s00436-021-07343-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-021-07343-0