Abstract
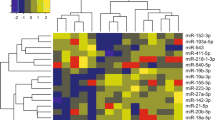
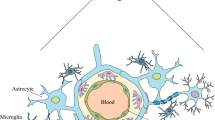
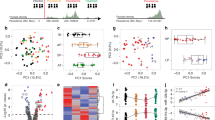
The appearance of increasing drug resistance in apicomplexan intracellular Plasmodium falciparum presents a significant challenge. P. falciparum infection results in cerebral malaria (CM), causing irreversible damage to the brain leading to high mortality cases. To enhance the clinical outcome of the disease, further research is required to identify new molecular targets involved in disease manifestations. Presently, the role of non-coding microRNAs (miRNAs) derived from different cells implicated in CM pathogenesis is still barely understood. Despite the absence of miRNA machinery in Plasmodium, host-parasite interactions can lead to disease severity or impart resistance to malaria. Cytoadherence and sequestration of parasitized RBCs dysregulate the miRNA profile of brain endothelial cells, leukocytes, monocytes, and platelets, disrupting blood–brain barrier integrity and activating inflammatory signaling pathways. The abundance of miRNA in blood plasma samples of CM patients directly correlates to cerebral symptoms compared to non-CM patients and healthy individuals. Moreover, the differential host-miRNA signatures distinguish P. falciparum from P. vivax infection. Here, we review the diverse functions of host-miRNA, either protective, pathogenic, or a combination of the two, which may act as prognostic markers and novel antimalarial drug targets.


Similar content being viewed by others
References
Al-Quraishy S, Dkhil MA, Delic D, Abdel-Baki AA, Wunderlich F (2012) Organ-specific testosterone-insensitive response of miRNA expression of C57BL/6 mice to Plasmodium chabaudi malaria. Parasitol Res 111(3):1093–1101. https://doi.org/10.1007/s00436-012-2937-3
Androulidaki A et al (2009) The kinase Akt1 controls macrophage response to lipopolysaccharide by regulating microRNAs. Immunity 31(2):220–231. https://doi.org/10.1016/j.immuni.2009.06.024
Assis PA et al (2020) Integrative analysis of microRNA and mRNA expression profiles of monocyte-derived dendritic cells differentiation during experimental cerebral malaria. J Leukoc Biol 108(4):1183–1197. https://doi.org/10.1002/JLB.1MA0320-731R
Babatunde KA et al (2018) Malaria infected red blood cells release small regulatory RNAs through extracellular vesicles. Sci Rep 8(1):884. https://doi.org/10.1038/s41598-018-19149-9
Bai H, Wu S (2019) miR-451: a novel biomarker and potential therapeutic target for cancer. Onco Targets Ther 12:11069–11082. https://doi.org/10.2147/OTT.S230963
Barker KR et al (2017) miR-155 modifies inflammation, endothelial activation and blood-brain barrier dysfunction in cerebral malaria. Mol Med 23:24–33. https://doi.org/10.2119/molmed.2016.00139
Bartel DP (2009) MicroRNAs: target recognition and regulatory functions. Cell 136(2):215–233. https://doi.org/10.1016/j.cell.2009.01.002
Bartel DP (2018) Metazoan microRNAs. Cell 173(1):20–51. https://doi.org/10.1016/j.cell.2018.03.006
Bartel DP (2004) MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116(2):281–297. https://doi.org/10.1016/s0092-8674(04)00045-5
Bezman NA, Chakraborty T, Bender T, Lanier LL (2011) miR-150 regulates the development of NK and iNKT cells. J Exp Med 208(13):2717–2731. https://doi.org/10.1084/jem.20111386
Birnbaum J et al (2020) A Kelch13-defined endocytosis pathway mediates artemisinin resistance in malaria parasites. Science 367(6473):51–59. https://doi.org/10.1126/science.aax4735
Bogdan C (2012) Natural killer cells in experimental and human leishmaniasis. Front Cell Infect Microbiol 2:69. https://doi.org/10.3389/fcimb.2012.00069
Brewster DR, Kwiatkowski D, White NJ (1990) Neurological sequelae of cerebral malaria in children. Lancet 336(8722):1039–1043. https://doi.org/10.1016/0140-6736(90)92498-7
Burel JG et al. (2017) Dichotomous miR expression and immune responses following primary blood-stage malaria. JCI Insight 2(15). https://doi.org/10.1172/jci.insight.93434
Cannella D et al (2014) miR-146a and miR-155 delineate a microRNA fingerprint associated with Toxoplasma persistence in the host brain. Cell Rep 6(5):928–937. https://doi.org/10.1016/j.celrep.2014.02.002
Chakrabarti M, Garg S, Rajagopal A, Pati S, Singh S (2020) Targeted repression of Plasmodium apicortin by host microRNA impairs malaria parasite growth and invasion. Dis Model Mech 13(6). https://doi.org/10.1242/dmm.042820
Chakraborty C, Sharma AR, Sharma G, Doss CGP, Lee SS (2017) Therapeutic miRNA and siRNA: moving from bench to clinic as next generation medicine. Mol Ther Nucleic Acids 8:132–143. https://doi.org/10.1016/j.omtn.2017.06.005
Chamnanchanunt S et al (2015) Downregulation of plasma miR-451 and miR-16 in Plasmodium vivax infection. Exp Parasitol 155:19–25. https://doi.org/10.1016/j.exppara.2015.04.013
Chapman LM, Ture SK, Field DJ, Morrell CN (2017) miR-451 limits CD4(+) T cell proliferative responses to infection in mice. Immunol Res 65(4):828–840. https://doi.org/10.1007/s12026-017-8919-x
Chen XM, Splinter PL, O’Hara SP, LaRusso NF (2007) A cellular micro-RNA, let-7i, regulates Toll-like receptor 4 expression and contributes to cholangiocyte immune responses against Cryptosporidium parvum infection. J Biol Chem 282(39):28929–28938. https://doi.org/10.1074/jbc.M702633200
Coelho CH, Doritchamou JYA, Zaidi I, Duffy PE (2017) Advances in malaria vaccine development: report from the 2017 malaria vaccine symposium. NPJ Vaccines 2:34. https://doi.org/10.1038/s41541-017-0035-3
Cohen A, Zinger A, Tiberti N, Grau GER, Combes V (2018) Differential plasma microvesicle and brain profiles of microRNA in experimental cerebral malaria. Malar J 17(1):192. https://doi.org/10.1186/s12936-018-2330-5
Combes V et al (2005) ABCA1 gene deletion protects against cerebral malaria: potential pathogenic role of microparticles in neuropathology. Am J Pathol 166(1):295–302. https://doi.org/10.1016/S0002-9440(10)62253-5
Dandewad V, Vindu A, Joseph J, Seshadri V (2019) Import of human miRNA-RISC complex into Plasmodium falciparum and regulation of the parasite gene expression. J Biosci 44(2). https://doi.org/10.1007/s12038-019-9870-x
Das S, Manna S, Saha B, Hati AK, Roy S (2019) Novel pfkelch13 gene polymorphism associates with artemisinin resistance in Eastern India. Clin Infect Dis 69(7):1144–1152. https://doi.org/10.1093/cid/ciy1038
Deen J (2021) Coinfections and Malaria. In: Hommel M, Kremsner PG (eds) Encyclopedia of malaria. Springer, New York, pp 1–10
Delic D, Dkhil M, Al-Quraishy S, Wunderlich F (2011) Hepatic miRNA expression reprogrammed by Plasmodium chabaudi malaria. Parasitol Res 108(5):1111–1121. https://doi.org/10.1007/s00436-010-2152-z
Dieng MM et al (2020) Integrative genomic analysis reveals mechanisms of immune evasion in P. falciparum malaria. Nat Commun 11(1):5093
Dkhil MA, Al-Quraishy SA, Abdel-Baki AS, Delic D, Wunderlich F (2016) Differential miRNA expression in the liver of Balb/c mice protected by vaccination during crisis of Plasmodium chabaudi blood-stage malaria. Front Microbiol 7:2155. https://doi.org/10.3389/fmicb.2016.02155
Dobano C et al (2019) Concentration and avidity of antibodies to different circumsporozoite epitopes correlate with RTS, S/AS01E malaria vaccine efficacy. Nat Commun 10(1):2174. https://doi.org/10.1038/s41467-019-10195-z
El-Assaad F et al (2011) Differential microRNA expression in experimental cerebral and noncerebral malaria. Infect Immun 79(6):2379–2384. https://doi.org/10.1128/IAI.01136-10
Fabian MR, Sonenberg N, Filipowicz W (2010) Regulation of mRNA translation and stability by microRNAs. Annu Rev Biochem 79:351–379. https://doi.org/10.1146/annurev-biochem-060308-103103
Feliciano P (2012) miRNAs and malaria resistance. Nat Genet 44(10):1079–1079. https://doi.org/10.1038/ng.2428
Gatsiou A, Boeckel JN, Randriamboavonjy V, Stellos K (2012) MicroRNAs in platelet biogenesis and function: implications in vascular homeostasis and inflammation. Curr Vasc Pharmacol 10(5):524–531. https://doi.org/10.2174/157016112801784611
Gnanguenon V et al (2015) Malaria vectors resistance to insecticides in Benin: current trends and mechanisms involved. Parasit Vectors 8:223. https://doi.org/10.1186/s13071-015-0833-2
Gupta H et al (2021) Plasma microRNA profiling of Plasmodium falciparum biomass and association with severity of malaria disease. Emerg Infect Dis 27(2):430–442. https://doi.org/10.3201/eid2702.191795
Hamidi F et al (2021) Inhibition of anti-inflammatory cytokines, IL-10 and TGF-beta, in Leishmania major infected macrophage by miRNAs: a new therapeutic modality against leishmaniasis. Microb Pathog 153:104777. https://doi.org/10.1016/j.micpath.2021.104777
Henriksson M, Luscher B (1996) Proteins of the Myc network: essential regulators of cell growth and differentiation. Adv Cancer Res 68:109–182. https://doi.org/10.1016/s0065-230x(08)60353-x
Hentzschel F et al (2014) AAV8-mediated in vivo overexpression of miR-155 enhances the protective capacity of genetically attenuated malarial parasites. Mol Ther 22(12):2130–2141. https://doi.org/10.1038/mt.2014.172
Hu G et al (2009) MicroRNA-98 and let-7 confer cholangiocyte expression of cytokine-inducible Src homology 2-containing protein in response to microbial challenge. J Immunol 183(3):1617–1624. https://doi.org/10.4049/jimmunol.0804362
Hu R et al (2014) miR-155 promotes T follicular helper cell accumulation during chronic, low-grade inflammation. Immunity 41(4):605–619. https://doi.org/10.1016/j.immuni.2014.09.015
Ingham VA, Pignatelli P, Moore JD, Wagstaff S, Ranson H (2017) The transcription factor Maf-S regulates metabolic resistance to insecticides in the malaria vector Anopheles gambiae. BMC Genomics 18(1):669. https://doi.org/10.1186/s12864-017-4086-7
Iorio MV et al (2005) MicroRNA gene expression deregulation in human breast cancer. Cancer Res 65(16):7065–7070. https://doi.org/10.1158/0008-5472.CAN-05-1783
Jiang D, Wu S, Xu L, Xie G, Peng H-J (2021) Anti-Infection Roles of mir155-5p Packaged in Exosomes Secreted by Dendritic Cells Infected with Toxoplasma Gondii. https://doi.org/10.21203/rs.3.rs-287510/v1
Jiang S, Li X, Wang X, Ban Q, Hui W, Jia B (2016) MicroRNA profiling of the intestinal tissue of Kazakh sheep after experimental Echinococcus granulosus infection, using a high-throughput approach. Parasite 23:23. https://doi.org/10.1051/parasite/2016023
Jiang S et al (2010) MicroRNA-155 functions as an OncomiR in breast cancer by targeting the suppressor of cytokine signaling 1 gene. Cancer Res 70(8):3119–3127. https://doi.org/10.1158/0008-5472.CAN-09-4250
John CC et al (2008) Cerebral malaria in children is associated with long-term cognitive impairment. Pediatrics 122(1):e92–e99. https://doi.org/10.1542/peds.2007-3709
Johnson SM et al (2005) RAS is regulated by the let-7 microRNA family. Cell 120(5):635–647. https://doi.org/10.1016/j.cell.2005.01.014
Kaur H, Sehgal R, Kumar A, Sehgal A, Bansal D, Sultan AA (2018) Screening and identification of potential novel biomarker for diagnosis of complicated Plasmodium vivax malaria. J Transl Med 16(1):272. https://doi.org/10.1186/s12967-018-1646-9
Khan S et al (2020) Immune escape and immune camouflage may reduce the efficacy of RTS, S vaccine in Malawi. Hum Vaccin Immunother 16(2):214–227. https://doi.org/10.1080/21645515.2018.1560772
Khraiwesh B et al (2010) Transcriptional control of gene expression by microRNAs. Cell 140(1):111–122. https://doi.org/10.1016/j.cell.2009.12.023
Laishram DD et al (2012) The complexities of malaria disease manifestations with a focus on asymptomatic malaria. Malar J 11:29. https://doi.org/10.1186/1475-2875-11-29
LaMonte G et al (2012) Translocation of sickle cell erythrocyte microRNAs into Plasmodium falciparum inhibits parasite translation and contributes to malaria resistance. Cell Host Microbe 12(2):187–199. https://doi.org/10.1016/j.chom.2012.06.007
Li JJ et al (2018) Identification of potential whole blood MicroRNA biomarkers for the blood stage of adult imported falciparum malaria through integrated mRNA and miRNA expression profiling. Biochem Biophys Res Commun 506(3):471–477. https://doi.org/10.1016/j.bbrc.2018.10.072
Liang Y, Ridzon D, Wong L, Chen C (2007) Characterization of microRNA expression profiles in normal human tissues. BMC Genomics 8:166. https://doi.org/10.1186/1471-2164-8-166
Liu X, Meng J (2018) Tanshinone IIA ameliorates lipopolysaccharide-induced inflammatory response in bronchial epithelium cell line BEAS-2B by down-regulating miR-27a. Biomed Pharmacother 104:158–164. https://doi.org/10.1016/j.biopha.2018.05.021
Ludwig N et al (2016) Distribution of miRNA expression across human tissues. Nucleic Acids Res 44(8):3865–3877. https://doi.org/10.1093/nar/gkw116
Makenga G et al (2020) Prevalence of malaria parasitaemia in school-aged children and pregnant women in endemic settings of sub-Saharan Africa: a systematic review and meta-analysis. Parasite Epidemiol Control 11:e00188. https://doi.org/10.1016/j.parepi.2020.e00188
Mantel PY et al (2016) Infected erythrocyte-derived extracellular vesicles alter vascular function via regulatory Ago2-miRNA complexes in malaria. Nat Commun 7:12727. https://doi.org/10.1038/ncomms12727
Martin-Alonso A et al (2018) Differentially expressed microRNAs in experimental cerebral malaria and their involvement in endocytosis, adherens junctions, FoxO and TGF-beta signalling pathways. Sci Rep 8(1):11277. https://doi.org/10.1038/s41598-018-29721-y
Masaki S, Ohtsuka R, Abe Y, Muta K, Umemura T (2007) Expression patterns of microRNAs 155 and 451 during normal human erythropoiesis. Biochem Biophys Res Commun 364(3):509–514. https://doi.org/10.1016/j.bbrc.2007.10.077
Narasimhan M, Patel D, Vedpathak D, Rathinam M, Henderson G, Mahimainathan L (2012) Identification of novel microRNAs in post-transcriptional control of Nrf2 expression and redox homeostasis in neuronal, SH-SY5Y cells. PLoS ONE 7(12):e51111. https://doi.org/10.1371/journal.pone.0051111
Neilsen PM et al (2013) Mutant p53 drives invasion in breast tumors through up-regulation of miR-155. Oncogene 32(24):2992–3000. https://doi.org/10.1038/onc.2012.305
Nielsen CM, Vekemans J, Lievens M, Kester KE, Regules JA, Ockenhouse CF (2018) RTS, S malaria vaccine efficacy and immunogenicity during Plasmodium falciparum challenge is associated with HLA genotype. Vaccine 36(12):1637–1642. https://doi.org/10.1016/j.vaccine.2018.01.069
Nimsarkar P, Ingale P, Singh S (2020) Systems studies uncover miR-146a as a target in Leishmania major infection model. ACS Omega 5(21):12516–12526. https://doi.org/10.1021/acsomega.0c01502
Nsanzabana C (2019) Resistance to artemisinin combination therapies (ACTs): do not forget the partner drug! Trop Med Infect Dis 4(1). https://doi.org/10.3390/tropicalmed4010026
O’Connell RM, Chaudhuri AA, Rao DS, Baltimore D (2009) Inositol phosphatase SHIP1 is a primary target of miR-155. Proc Natl Acad Sci U S A 106(17):7113–7118. https://doi.org/10.1073/pnas.0902636106
O’Hara SP et al (2010) NFkappaB p50-CCAAT/enhancer-binding protein beta (C/EBPbeta)-mediated transcriptional repression of microRNA let-7i following microbial infection. J Biol Chem 285(1):216–225. https://doi.org/10.1074/jbc.M109.041640
Pombo DJ et al (2002) Immunity to malaria after administration of ultra-low doses of red cells infected with Plasmodium falciparum. Lancet 360(9333):610–617. https://doi.org/10.1016/S0140-6736(02)09784-2
Pordzik J et al (2018) The potential role of platelet-related microRNAs in the development of cardiovascular events in high-risk populations, including diabetic patients: a review. Front Endocrinol (lausanne) 9:74. https://doi.org/10.3389/fendo.2018.00074
Qayum AA et al (2016) IL-10-induced miR-155 targets SOCS1 to enhance IgE-mediated mast cell function. J Immunol 196(11):4457–4467. https://doi.org/10.4049/jimmunol.1502240
Raman J et al (2020) High levels of imported asymptomatic malaria but limited local transmission in KwaZulu-Natal, a South African malaria-endemic province nearing malaria elimination. Malar J 19(1):152. https://doi.org/10.1186/s12936-020-03227-3
Rangel G et al (2017) Prediction of miRNA that modulate significant host response genes as potential biomarkers in cerebral malaria infection
Rathjen T, Nicol C, McConkey G, Dalmay T (2006) Analysis of short RNAs in the malaria parasite and its red blood cell host. FEBS Lett 580(22):5185–5188. https://doi.org/10.1016/j.febslet.2006.08.063
Ruiz-Tagle C, Naves R, Balcells ME (2020) Unraveling the role of microRNAs in Mycobacterium tuberculosis infection and disease: advances and pitfalls. Infect Immun 88(3). https://doi.org/10.1128/IAI.00649-19
Salam N, Mustafa S, Hafiz A, Chaudhary AA, Deeba F, Parveen S (2018) Global prevalence and distribution of coinfection of malaria, dengue and chikungunya: a systematic review. BMC Public Health 18(1):710. https://doi.org/10.1186/s12889-018-5626-z
Santos ML et al (2020) A distinct fingerprint of inflammatory mediators and miRNAs in Plasmodium vivax severe thrombocytopenia. bioRxiv. https://doi.org/10.1101/2020.08.20.260463
Sheedy FJ, O’Neill LA (2008) Adding fuel to fire: microRNAs as a new class of mediators of inflammation. Ann Rheum Dis 67(Suppl 3):iii50-5. https://doi.org/10.1136/ard.2008.100289
SIMON-OKE IA, Ogunseem MF, Afolabi OJ, Awosolu O (2019) 2019:6 %J Journal of Biomedicine and Translational Research. https://doi.org/10.14710/jbtr.v5i1.3711
Sonkoly E, Stahle M, Pivarcsi A (2008) MicroRNAs and immunity: novel players in the regulation of normal immune function and inflammation. Semin Cancer Biol 18(2):131–140. https://doi.org/10.1016/j.semcancer.2008.01.005
Sun L et al (2018) Different erythrocyte microRNA Profiles in low- and high-altitude individuals. Front Physiol 9:1099. https://doi.org/10.3389/fphys.2018.01099
Sun L, Yu Y, Niu B, Wang D (2020) Red blood cells as potential repositories of microRNAs in the circulatory system. Front Genet 11:442. https://doi.org/10.3389/fgene.2020.00442
Testa U, Pelosi E, Castelli G, Labbaye C (2017) miR-146 and miR-155: two key modulators of immune response and tumor development. Noncoding RNA 3(3). https://doi.org/10.3390/ncrna3030022
Thomson R et al (2019) pfhrp2 and pfhrp3 gene deletions that affect malaria rapid diagnostic tests for Plasmodium falciparum: analysis of archived blood samples from 3 African countries. J Infect Dis 220(9):1444–1452. https://doi.org/10.1093/infdis/jiz335
Tili E et al (2011) Mutator activity induced by microRNA-155 (miR-155) links inflammation and cancer. Proc Natl Acad Sci U S A 108(12):4908–4913. https://doi.org/10.1073/pnas.1101795108
Urbich C et al (2012) MicroRNA-27a/b controls endothelial cell repulsion and angiogenesis by targeting semaphorin 6A. Blood 119(6):1607–1616. https://doi.org/10.1182/blood-2011-08-373886
van de Bunt M et al (2013) The miRNA profile of human pancreatic islets and beta-cells and relationship to type 2 diabetes pathogenesis. PLoS ONE 8(1):e55272. https://doi.org/10.1371/journal.pone.0055272
van Loon W, Gai PP, Hamann L, Bedu-Addo G, Mockenhaupt FP (2019) MiRNA-146a polymorphism increases the odds of malaria in pregnancy. Malar J 18(1):7. https://doi.org/10.1186/s12936-019-2643-z
van Loon W et al (2020) MiRNA-146a polymorphism was not associated with malaria in Southern India. Am J Trop Med Hyg 102(5):1072–1074. https://doi.org/10.4269/ajtmh.19-0845
Varikuti S et al. (2019) MicroRNA 155 contributes to host immunity against Leishmania donovani but is not essential for resolution of infection. Infect Immun 87(8). https://doi.org/10.1128/IAI.00307-19
Wah ST, Hananantachai H, Patarapotikul J, Ohashi J, Naka I, Nuchnoi P (2019) MicroRNA-27a and microRNA-146a SNP in cerebral malaria. Mol Genet Genomic Med 7(2):e00529. https://doi.org/10.1002/mgg3.529
Wang T, Wu F, Yu D (2019) miR-144/451 in hematopoiesis and beyond. ExRNA 1(1):16. https://doi.org/10.1186/s41544-019-0035-8
Wang Z et al (2017) Red blood cells release microparticles containing human argonaute 2 and miRNAs to target genes of Plasmodium falciparum. Emerg Microbes Infect 6(8):e75. https://doi.org/10.1038/emi.2017.63
World Health Organization (2017) False-negative RDT results and implications of new reports of P. falciparum histidine-rich protein 2/3 gene deletions. https://apps.who.int/iris/bitstream/10665/258972/1/WHO-HTM-GMP-2017.18-eng.pdf
World Health Organization (2020) World malaria report 2020: 20 years of global progress and challenges. In World malaria report 2020: 20 years of global progress and challenges. https://apps.who.int/iris/rest/bitstreams/1321872/retrieve
Xiao C et al (2007) MiR-150 controls B cell differentiation by targeting the transcription factor c-Myb. Cell 131(1):146–159. https://doi.org/10.1016/j.cell.2007.07.021
Xue X, Zhang Q, Huang Y, Feng L, Pan W (2008) No miRNA were found in Plasmodium and the ones identified in erythrocytes could not be correlated with infection. Malar J 7:47. https://doi.org/10.1186/1475-2875-7-47
Yewhalaw D et al (2011) Multiple insecticide resistance: an impediment to insecticide-based malaria vector control program. PLoS ONE 6(1):e16066. https://doi.org/10.1371/journal.pone.0016066
Zhang M et al (2014) MicroRNA-27a/b regulates cellular cholesterol efflux, influx and esterification/hydrolysis in THP-1 macrophages. Atherosclerosis 234(1):54–64. https://doi.org/10.1016/j.atherosclerosis.2014.02.008
Zhou X, Li X, Wu M (2018) miRNAs reshape immunity and inflammatory responses in bacterial infection. Signal Transduct Target Ther 3:14. https://doi.org/10.1038/s41392-018-0006-9
Funding
The V.R. lab is supported by the research grant from Department of Science & Technology (DST), INSPIRE-Faculty Project (DST/INSPIRE/04/2018/003541), Ministry of Science and Technology, Government of India.
Author information
Authors and Affiliations
Contributions
A.M. and V.R. contributed equally towards literature survey, data interpretation, and preparation of the figures. V.R. contributed to the overall supervision, design of the review, critical analysis of the intellectual content, revision of the manuscript, and funding acquisition. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Section Editor: Kevin S.W. Tan
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Abhinab Mohanty and Vinoth Rajendran are joint first author - contributed equally to this paper.
Rights and permissions
About this article
Cite this article
Mohanty, A., Rajendran, V. Mammalian host microRNA response to plasmodial infection: role as therapeutic target and potential biomarker. Parasitol Res 120, 3341–3353 (2021). https://doi.org/10.1007/s00436-021-07293-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-021-07293-7