Abstract
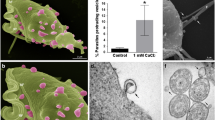
Trichomonas vaginalis is responsible for the prevalence of trichomoniasis, which may be one of the most epidemic nonviral sexually transmitted pathogens. Extracellular traps (ET) are a unique form of innate immunity against infection; they bind to and kill microorganisms. However, the effect of T. vaginalis on ET release in the human monocytic cell line THP-1 remains unclear. In the present study, the morphology of ET derived from THP-1 in response to T. vaginalis was observed by scanning electron microscopy (SEM). The results demonstrated ET entangling T. vaginalis. Then, the colocalization of histone (H3) and myeloperoxidase (MPO) with DNA was observed via fluorescence confocal microscopy. Colocalization revealed the classic characteristics of DNA decorated with H3 and MPO. T. vaginalis significantly increased reactive oxygen species (ROS) and THP-1-derived ET. In addition, we measured the levels of lactic dehydrogenase (LDH) and the phosphorylation of the P38 and ERK1/2 MAPK signaling pathways. The results indicated that the formation of ET induced by T. vaginalis was related to phosphorylation of the P38 and ERK1/2 MAPK signaling pathways but not to LDH levels. These data confirmed the phenomenon of THP-1-derived ET being triggered by T. vaginalis in vitro; this process may play a pivotal role in innate immunity during defense against T. vaginalis infection.







Similar content being viewed by others

References
Abi Abdallah DS, Lin C, Ball CJ, King MR, Duhamel GE, Denkers EY (2012) Toxoplasma gondii triggers release of human and mouse neutrophil extracellular traps. Infect Immun 80(2):768–777. https://doi.org/10.1128/IAI.05730-11
Aulik NA, Hellenbrand KM, Czuprynski CJ (2012) Mannheimia haemolytica and its leukotoxin cause macrophage extracellular trap formation by bovine macrophages. Infect Immun 80(5):1923–1933. https://doi.org/10.1128/IAI.06120-11
Baker VS, Imade GE, Molta NB, Tawde P, Pam SD, Obadofin MO, Sagay SA, Egah DZ, Iya D, Afolabi BB, Baker M, Ford K, Ford R, Roux KH, Keller TCS (2008) Cytokine-associated neutrophil extracellular traps and antinuclear antibodies in Plasmodium falciparum infected children under six years of age. Malar J 7:41. https://doi.org/10.1186/1475-2875-7-41
Behrendt JH, Ruiz A, Zahner H, Taubert A, Hermosilla C (2010) Neutrophil extracellular trap formation as innate immune reactions against the apicomplexan parasite Eimeria bovis. Vet Immunol Immunopathol 133(1):1–8. https://doi.org/10.1016/j.vetimm.2009.06.012
Bonne-Annee S et al (2014) Extracellular traps are associated with human and mouse neutrophil and macrophage mediated killing of larval Strongyloides stercoralis. Microbes Infect 16(6):502–511. https://doi.org/10.1016/j.micinf.2014.02.012
Brinkmann V, Zychlinsky A (2007) Beneficial suicide: why neutrophils die to make NETs. Nature Reviews Microbiology 5(8):577–582
Brinkmann V et al (2004) Neutrophil extracellular traps kill bacteria. Science 303(5663):1532–1535. https://doi.org/10.1126/science.1092385
Bruns S, Kniemeyer O, Hasenberg M, Aimanianda V, Nietzsche S, Thywißen A, Jeron A, Latgé JP, Brakhage AA, Gunzer M (2010) Production of extracellular traps against Aspergillus fumigatus in vitro and in infected lung tissue is dependent on invading neutrophils and influenced by hydrophobin RodA. PLoS Pathog 6(4):e1000873. https://doi.org/10.1371/journal.ppat.1000873
Cargnello M, Roux PP (2011) Activation and function of the MAPKs and their substrates, the MAPK-activated protein kinases. Microbiol Mol Biol Rev 75(1):50–83. https://doi.org/10.1128/MMBR.00031-10
Caro TM, Hermosilla C, Silva LMR, Cortes H, Taubert A (2014) Neutrophil extracellular traps as innate immune reaction against the emerging apicomplexan parasite Besnoitia besnoiti. PloS one 9(3):e91415. https://doi.org/10.1371/journal.pone.0091415
Chuah C, Jones MK, Burke ML, Owen HC, Anthony BJ, McManus DP, Ramm GA, Gobert GN (2013) Spatial and temporal transcriptomics of Schistosoma japonicum-induced hepatic granuloma formation reveals novel roles for neutrophils. J Leukoc Biol 94(2):353–365. https://doi.org/10.1189/jlb.1212653
Fiori PL, Diaz N, Cocco AR, Rappelli P, Dessi D (2013) Association of Trichomonas vaginalis with its symbiont Mycoplasma hominis synergistically upregulates the in vitro proinflammatory response of human monocytes. Sex Transm Infect 89(6):449–454. https://doi.org/10.1136/sextrans-2012-051006
Guimaraes-Costa AB, Nascimento MTC, Froment GS, Soares RPP, Morgado FN, Conceicao-Silva F, Saraiva EM (2009) Leishmania amazonensis promastigotes induce and are killed by neutrophil extracellular traps. Proc Natl Acad Sci U S A 106(16):6748–6753. https://doi.org/10.1073/pnas.0900226106
Guimaraes-Costa AB, Nascimento MT, Wardini AB, Pinto-da-Silva LH, Saraiva EM (2012) ETosis: a microbicidal mechanism beyond cell death. J Parasitol Res 2012:929743:1–11. https://doi.org/10.1155/2012/929743
Hakkim A, Fuchs TA, Martinez NE, Hess S, Prinz H, Zychlinsky A, Waldmann H (2011) Activation of the Raf-MEK-ERK pathway is required for neutrophil extracellular trap formation. Nat Chem Biol 7(2):75–77. https://doi.org/10.1038/nchembio.496
Han IH, Goo SY, Park SJ, Hwang SJ, Kim YS, Yang MS, Ahn MH, Ryu JS (2009) Proinflammatory cytokine and nitric oxide production by human macrophages stimulated with Trichomonas vaginalis. Korean J Parasitol 47(3):205–212. https://doi.org/10.3347/kjp.2009.47.3.205
Kissinger P (2015) Trichomonas vaginalis: a review of epidemiologic, clinical and treatment issues. BMC Infectious Diseases 15(1):307. https://doi.org/10.1186/s12879-015-1055-0
McClelland RS et al (2007) Infection with Trichomonas vaginalis increases the risk of HIV-1 acquisition. J Infect Dis 195(5):698–702. https://doi.org/10.1086/511278
Menezes CB, Tasca T (2016) Trichomoniasis immunity and the involvement of the purinergic signaling. Biom J 39(4):234–243. https://doi.org/10.1016/j.bj.2016.06.007
Mercer F, Ng SH, Brown TM, Boatman G, Johnson PJ (2018) Neutrophils kill the parasite Trichomonas vaginalis using trogocytosis. PLoS Biol 16(2):e2003885. https://doi.org/10.1371/journal.pbio.2003885
Munoz-Caro T, Lendner M, Daugschies A, Hermosilla C, Taubert A (2015a) NADPH oxidase, MPO, NE, ERK1/2, p38 MAPK and Ca2+ influx are essential for Cryptosporidium parvum-induced NET formation. Dev Comp Immunol 52(2):245–254. https://doi.org/10.1016/j.dci.2015.05.007
Munoz-Caro T et al (2015b) Eimeria bovis-triggered neutrophil extracellular trap formation is CD11b-, ERK 1/2-, p38 MAP kinase- and SOCE-dependent. Vet Res 46:23. https://doi.org/10.1186/s13567-015-0155-6
Munoz-Caro T, Silva LM, Ritter C, Taubert A, Hermosilla C (2014) Besnoitia besnoiti tachyzoites induce monocyte extracellular trap formation. Parasitol Res 113(11):4189–4197. https://doi.org/10.1007/s00436-014-4094-3
Perez D et al (2016) Eimeria ninakohlyakimovae induces NADPH oxidase-dependent monocyte extracellular trap formation and upregulates IL-12 and TNF-alpha, IL-6 and CCL2 gene transcription. Vet Parasitol 227:143–150. https://doi.org/10.1016/j.vetpar.2016.07.028
Quinlivan EB, Patel SN, Grodensky CA, Golin CE, Tien HC, Hobbs MM (2012) Modeling the impact of Trichomonas vaginalis infection on HIV transmission in HIV-infected individuals in medical care. Sex Transm Dis 39(9):671–677. https://doi.org/10.1097/OLQ.0b013e3182593839
Reichel M, Muñoz-Caro T, Sanchez Contreras G, Rubio García A, Magdowski G, Gärtner U, Taubert A, Hermosilla C (2015) Harbour seal (Phoca vitulina) PMN and monocytes release extracellular traps to capture the apicomplexan parasite Toxoplasma gondii. Dev Comp Immunol 50(2):106–115. https://doi.org/10.1016/j.dci.2015.02.002
Roux PP, Blenis J (2004) ERK and p38 MAPK-activated protein kinases: a family of protein kinases with diverse biological functions. Microbiol Mol Biol Rev 68(2):320–344. https://doi.org/10.1128/MMBR.68.2.320-344.2004
Silva LM et al (2014) The apicomplexan parasite Eimeria arloingi induces caprine neutrophil extracellular traps. Parasitol Res 113(8):2797–2807. https://doi.org/10.1007/s00436-014-3939-0
Sousa-Rocha D, Thomaz-Tobias M, Diniz LF, Souza PS, Pinge-Filho P, Toledo KA (2015) Trypanosoma cruzi and its soluble antigens induce NET release by stimulating toll-like receptors. PLoS One 10(10):e0139569. https://doi.org/10.1371/journal.pone.0139569
Urban CF, Reichard U, Brinkmann V, Zychlinsky A (2006) Neutrophil extracellular traps capture and kill Candida albicans yeast and hyphal forms. Cell Microbiol 8(4):668–676
Wardini AB, Guimaraes-Costa AB, Nascimento MTC, Nadaes NR, Danelli MGM, Mazur C, Benjamim CF, Saraiva EM, Pinto-da-Silva LH (2010) Characterization of neutrophil extracellular traps in cats naturally infected with feline leukemia virus. J Gen Virol 91(Pt 1):259–264. https://doi.org/10.1099/vir.0.014613-0
Wei Z, Hermosilla C, Taubert A, He X, Wang X, Gong P, Li J, Yang Z, Zhang X (2016) Canine neutrophil extracellular traps release induced by the apicomplexan parasite Neospora caninum in vitro. Front Immunol 7:436. https://doi.org/10.3389/fimmu.2016.00436
Wei Z, Wang Y, Zhang X, Wang X, Gong P, Li J, Taubert A, Hermosilla C, Zhang X, Yang Z (2018) Bovine macrophage-derived extracellular traps act as early effectors against the abortive parasite Neospora caninum. Vet Parasitol 258:1–7. https://doi.org/10.1016/j.vetpar.2018.06.002
Yang Z, Wei Z, Hermosilla C, Taubert A, He X, Wang X, Gong P, Li J, Zhang X (2017) Caprine monocytes release extracellular traps against Neospora caninum in vitro. Front Immunol 8:2016. https://doi.org/10.3389/fimmu.2017.02016
Funding
This study was financially supported through a grant from the “National Key Basic Research Program (973 program) of China” (Grant No. 2015CB150300).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Handling editor: Julia Walochnik
Rights and permissions
About this article
Cite this article
Fei, L., Zhengkai, W., Weina, J. et al. Trichomonas vaginalis triggers the release of THP-1 extracellular traps. Parasitol Res 118, 267–274 (2019). https://doi.org/10.1007/s00436-018-6139-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-018-6139-5