Abstract
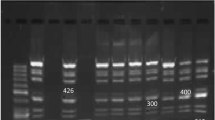
Trichomoniasis is the most common curable sexually transmitted disease worldwide. Resistance to metronidazole in treating trichomoniasis is a problematic health issue. We aimed to determine the minimum lethal concentration (MLC) of metronidazole for Trichomonas vaginalis isolates detected in Mansoura, Egypt and studied the genotypic profile of these isolates. Vaginal swab specimens were obtained from 320 symptomatic and 100 asymptomatic females, for whom clinical examination, vaginal discharge wet mount, Giemsa stain, and culture in modified Diamond’s media were performed. Metronidazole susceptibility testing by an aerobic tube assay was performed. Both sensitive and resistant isolates were examined by PCR amplification followed by restriction fragment length polymorphism (RFLP). Trichomonas vaginalis was identified in 49/420 (11.7%) using either culture or PCR, while wet mount and Giemsa stain detected the parasite in 8.1 and 7.6% of participants, respectively. After 48 h incubation, most isolates were sensitive to metronidazole with a minimal lethal concentration (MLC) of 1 μg/ml. Mild resistance was observed in two isolates with MLCs of 64 μg\ml and mild to moderate resistance was observed in an additional two isolates with MLCs of 128 μg/ml. The four isolates that demonstrated low to moderate metronidazole resistance displayed a unique genotype band pattern by RFLP compared to the other 45 samples that were metronidazole sensitive. Our results highlight the presence of in vitro metronidazole tolerance in a few T. vaginalis isolates in Mansoura, Egypt that may lead to the development of drug resistance as well as the possibility of an identifying RFLP pattern in the isolates.

Similar content being viewed by others
References
Ali V, Nozaki T (2007) Current therapeutics, their problems, and sulfur-containing-amino-acid metabolism as a novel target against infections by “amitochondriate” protozoan parasites. Clin Microbiol Rev 20:164–187
Borchardt KA, Zhang MZ, Shing H, Flink K (1997) A comparison of the sensitivity of the InPouch TV, Diamond’s and Trichosel media for detection of Trichomonas vaginalis. Genitourin Med 73:297–298
Bradic M, Warring SD, Tooley GE, Scheid P, Secor WE, Land KM, Huang PJ, Chen TW, Lee CC, Tang P, Sullivan SA, Carlton JM (2017) Genetic indicators of drug resistance in the highly repetitive genome of Trichomonas vaginalis. Genome Biol Evol 9:1658–1672
Byun JM, Jeong DH, Kim YN, Lee KB, Sung MS, Kim KT (2015) Experience of successful treatment of patients with metronidazole-resistant Trichomonas vaginalis with zinc sulfate: a case series. Taiwan J Obstet Gynecol 54:617–620
Cherpes TL, Wiesenfeld HC, Melan MA, Kant JA, Cosentino LA, Meyn LA, Hillier SL (2006) The associations between pelvic inflammatory disease, Trichomonas vaginalis infection, and positive herpes simplex virus type 2 serology. Sex Transm Dis 33:747–752
Conrad MD, Gorman AW, Schillinger JA, Fiori PL, Arroyo R, Malla N, Dubey ML, Gonzalez J, Blank S, Secor WE, Carlton JM (2012) Extensive genetic diversity, unique population structure and evidence of genetic exchange in the sexually transmitted parasite Trichomonas vaginalis. PLoS Negl Trop Dis 6:e1573
Fouts AC, Kraus SJ (1980) Trichomonas vaginalis: reevaluation of its clinical presentation and laboratory diagnosis. J Infect Dis 141:137–143
Gandhi NR, Brust JC, Moodley P, Weissman D, Heo M, Ning Y, Moll AP, Friedland GH, Sturm AW, Shah NS (2014) Minimal diversity of drug-resistant Mycobacterium tuberculosis strains, South Africa. Emerg Infect Dis 20:426–433
Grama DF, Casarotti LS, Silva LS, Mendon DF, Viana C, Cury MC (2013) Prevalence of Trichomonas vaginalis and risk factors in women treated at public health units in Brazil: a transversal study. Trans R Soc Trop Med Hyg 107:584–591
Hegazy MM, El-Tantawy NL, Soliman MM, El-Sadeek ES, El-Nagar HS (2012) Performance of rapid immunochromatographic assay in the diagnosis of Trichomoniasis vaginalis. Diagn Microbiol Infect Dis 74:49–53
Hussein AH, Saleh MH, Nagaty IM, Ghieth KA, El-Azab NA (2015) Prevalence, clinical criteria and sociodemographic predictors of Trichomonas vaginalis infection in suspected Egyptian women, using direct diagnostic techniques. Iran J Parasitol 10:432–440
Hussien EM, El-Sayed HZ, El-Moamly AA, Helmy MM, Shaban MM (2005) Molecular characterization of Egyptian Trichomonas vaginalis clinical isolates by HSP70 restriction fragment length polymorphism. J Egypt Soc Parasitol 35:699–710
Kalra B, Kalra S (2017) Vulvovaginitis and diabetes. J Pak Med Assoc 67:143–145
Kazemi F, Hooshyar H, Zareikar B, Bandehpour M, Arbabi M, Talari S, Alizadeh R, Kazemi B (2010) Study on ITS1 gene of Iranian Trichomonas vaginalis by molecular methods. Iran J Parasitol 5:9–14
Kissinger P, Secor WE, Leichliter JS, Clark RA, Schmidt N, Curtin E, Martin DH (2008) Early repeated infections with Trichomonas vaginalis among HIV-positive and HIV-negative women. Clin Infect Dis 46:994–999
Kulda J, Vojtĕchovská M, Tachezy J, Demes P, Kunzová E (1982) Metronidazole resistance of Trichomonas vaginalis as a cause of treatment failure in trichomoniasis—a case report. Br J Vener Dis 58:394–399
Meri T, Jokiranta TS, Suhonen L, Meri S (2000) Resistance of Trichomonas vaginalis to metronidazole: report of the first three cases from Finland and optimization of in vitro susceptibility testing under various oxygen concentrations. J Clin Microbiol 38:763–767
Narcisi EM, Secor WE (1996) In vitro effect of tinidazole and furazolidone on metronidazole-resistant Trichomonas vaginalis. Antimicrob Agents Chemother 40:1121–1125
Negm AY, El-Haleem DA (2004) Detection of trichomoniasis in vaginal specimens by both conventional and modern molecular tools. J Egypt Soc Parasitol 34:589–600
O’Rourke M, Ison CA, Renton AM, Spratt BG (1995) Opa-typing: a high resolution tool for studying the epidemiology of gonorrhoea. Mol Microbiol 17:865–875
Paulish-Miller TE, Augostini P, Schuyler JA, Smith WL, Mordechai E, Adelson ME, Gygax SE, Secor WE, Hilbert DW (2014) Trichomonas vaginalis metronidazole resistance is associated with single nucleotide polymorphisms in the nitroreductase genes ntr4Tv and ntr6Tv. Antimicrob Agents Chemother 58:2938–2943
Poole DN, McClelland RS (2013) Global epidemiology of Trichomonas vaginalis. Sex Transm Infect 89:418–422
Rabiee S, Bazmani A, Matini M, Fallah M (2012) Comparison of resistant and susceptible strains of Trichomonas vaginalis to metronidazole using PCR method. Iran J Parasitol 7:24–30
Radonjic IV, Dzamic AM, Mitrovic SM, Arsic Arsenijevic VS, Popadic DM, Kranjcic Zec IF (2006) Diagnosis of Trichomonas vaginalis infection: the sensitivities and specificities of microscopy, culture and PCR assay. Eur J Obstet Gynecol Reprod Biol 126:116–120
Schwebke JR, Barrientes FJ (2006) Prevalence of Trichomonas vaginalis isolates with resistance to metronidazole and tinidazole. Antimicrob Agents Chemother 50:4209–4210
Stark JR, Judson G, Alderete JF, Mundodi V, Kucknoor AS, Giovannucci EL, Platz EA, Sutcliffe S, Fall K, Kurth T, Ma J, Stampfer MJ, Mucci LA (2009) Prospective study of Trichomonas vaginalis infection and prostate cancer incidence and mortality: Physicians’ Health Study. J Natl Cancer Inst 101:1406–1411
Snipes LJ, Gamard PM, Narcisi EM, Beard C, Lehmann T, Secor WE (2000) Molecular epidemiology of metronidazole resistance in a population of Trichomonas vaginalis clinical isolates. J Clin Microbiol 38:3004–3009
Yarlett N, Hof H, Yarlett NC (1987) Activities of metronidazole and niridazole against Trichomonas vaginalis clinical isolates. J Antimicrob Chemother 19:767–770
Younis EZ, Elamami AH (2016) Trichomonas vaginalis infection in women with Type 2 diabetes mellitus and vaginal discharge in Benghazi, Libya. Ibnosina J Med Biomed Sci 8:109–113
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This study was approved by the Institutional Review Board of the Faculty of Medicine, Mansoura University, Egypt (IRB reference no: 16.06.83).
Statement of informed consent
Prior to enrollment, informed consent was gained from each participant. Women with confirmed trichomoniasis were treated and followed up.
Rights and permissions
About this article
Cite this article
Abdel-Magied, A.A., El-Kholya, ES.I., Abou El-Khair, S.M. et al. The genetic diversity of metronidazole susceptibility in Trichomonas vaginalis clinical isolates in an Egyptian population. Parasitol Res 116, 3125–3130 (2017). https://doi.org/10.1007/s00436-017-5627-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-017-5627-3