Abstract
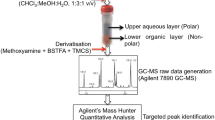
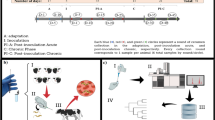
Differentiation between viable and non-viable hookworm ova in environmental samples is necessary in order to implement strategies to mitigate re-infections in endemic regions. In this study, an untargeted metabolic profiling method was developed that utilised gas chromatography-mass spectrometry (GC-MS) in order to investigate hookworm ova viability. Ancylostoma caninum was used to investigate the metabolites within viable and non-viable ova. Univariate and multivariate statistical analyses of the data resulted in the identification of 53 significant metabolites across all hookworm ova samples. The major compounds observed in viable and non-viable hookworm ova were tetradecanoic acid, commonly known as myristic acid [fold change (FC) = 0.4], and dodecanoic acid, commonly known as lauric acid (FC = 0.388). Additionally, the viable ova had self-protecting metabolites such as prostaglandins, a typical feature absent in non-viable ova. The results of this study demonstrate that metabolic profiling using GC-MS methods can be used to determine the viability of canine hookworm ova. Further studies are needed to assess the applicability of metabolic profiling using GC-MS to detect viable hookworm ova in the mixed (viable and non-viable) populations from environmental samples and identify the metabolites specific to human hookworm species.






Similar content being viewed by others
References
Beale DJ, Dunn MS, Morrison PD, Porter NA, Marlow DR (2012) Characterisation of bulk water samples from copper pipes undergoing microbially influenced corrosion by diagnostic metabolomic profiling. Corros Sci 55:272–279
Beale DJ, Marney D, Marlow DR, Morrison PD, Dunn MS, Key C, Palombo EA (2013) Metabolomic analysis of Cryptosporidium parvum oocysts in water: a proof of concept demonstration. Environ Pollut 174:201–203
Bethony J, Brooker S, Albonico M, Geiger SM, Loukas A, Diemert D, Hotez PJ (2006) Soil-transmitted helminth infections: ascariasis, trichuriasis, and hookworm. Lancet 367:1521–1532
Bowman DD, Little MD, Reimers RS (2003) Precision and accuracy of an assay for detecting Ascaris eggs in various biosolid matrices. Water Res 37:2063–2072
Durairaj RB (2005) Resorcinol: chemistry, technology and applications, Springer Science and Business Media
Fairfax KC, Vermeire JJ, Harrison LM, Bungiro RD, Grant W, Husain SZ, Cappello M (2009) Characterisation of a fatty acid and retinol binding protein orthologue from the hookworm Ancylostoma ceylanicum. Int J Parasitol 39:1561–1571
Freeman MC, Clasen T, Brooker SJ, Akoko DO, Rheingans R (2013) The impact of a school-based hygiene, water quality and sanitation intervention on soil-transmitted helminth reinfection: a cluster-randomized trial. Am J Trop Med Hyg 89:875–883
Gyawali P, Ahmed W, Sidhu JPS, Jagals P, Toze S (2015a) Comparison of concentration methods for rapid detection of hookworm ova in wastewater matrices using quantitative PCR. Exp Parasitol 159:160–167
Gyawali P, Sidhu JPS, Ahmed W, Jagals P, Toze S (2015b) Rapid concentration and sensitive detection of hookworm ova from wastewater matrices using a real-time PCR method. Exp Parasitol 159:5–12
Hotez PJ, Gurwith M (2011) Europe’s neglected infections of poverty. Int J Infect Dis 15:e611–e619
Hotez PJ, Alvarado M, Basanez MG, Bolliger I, Bourne R, Boussinesq M, Brooker SJ, Brown AS, Buckle G, Budke CM, Carabin H, Coffeng LE, Fevre EM, Furst T, Halasa YA, Asrasaria R, Johns NE, Keiser J, King CH, Lozano R, Murdoch ME, O’hanlon S, Pion SD, Pullan RL, Ramaiah KD, Roberts T, Shepard DS, Smith JL, Stolk WA, Undurraga EA, Utzinger J, Wang M, Murray CJ, Naghavi M (2014) The Global Burden of Disease Study 2010: interpretation and implications for the neglected tropical diseases. PLoS Negl Trop Dis 8:e2865
Jia TW, Melville S, Utzinger J, King CH, Zhou XN (2012) Soil-transmitted helminth reinfection after drug treatment: a systematic review and meta-analysis. PLoS Negl Trop Dis 6:e1621
Karkashan A, Khallaf B, Morris J, Thurbon N, Rough D, Smith SR, Deighton M (2015) Comparison of methodologies for enumerating and detecting the viability of Ascaris eggs in sewage sludge by standard incubation-microscopy, the BacLight Live/Dead viability assay and other vital dyes. Water Res 68:533–544
Knopp S, Steinmann P, Keiser J, Utzinger J (2012) Nematode infections: soil-transmitted helminths and Trichinella. Infect Dis Clin N Am 26:341–358
Kouremenos KA, Beale DJ, Antti H, Palombo EA (2014) Liquid chromatography time of flight mass spectrometry based environmental metabolomics for the analysis of Pseudomonas putida bacteria in potable water. J Chromatogr B Anal Technol Biomed Life Sci 966:179–186
Lammler G, Sahai BN, Herzog H (1969) Anthelmintic efficacy of 2, 6-dihydroxybenzoic acid-4′-bromanilide (Hoe 296 V) against mature and immature Paramphisto mum microbothrium in goats. Acta Vet Acad Sci Hung 19:447–451
Lier T, Do DT, Johansen MV, Nguyen TH, Dalsgaard A, Asfeldt AM (2014) High reinfection rate after preventive chemotherapy for fishborne zoonotic trematodes in Vietnam. PLoS Negl Trop Dis 8:e2958
Markov GV, Baskaran P, Sommer RJ (2014) The same or not the same: lineage-specific gene expansions and homology relationships in multigene families in nematodes. J Mol Evol 80:18–36
McCarthy JS, Lustigman S, Yang GJ, Barakat RM, Garcia HH, Sripa B, Willingham AL, Prichard RK, Basanez MG (2012) A research agenda for helminth diseases of humans: diagnostics for control and elimination programmes. PLoS Negl Trop Dis 6:e1601
Ng JS, Ryan U, Trengove RD, Maker GL (2012) Development of an untargeted metabolomics method for the analysis of human faecal samples using Cryptosporidium-infected samples. Mol Biochem Parasitol 185:145–150
Olszewski KL, Morrisey JM, Wilinski D, Burns JM, Vaidya AB, Rabinowitz JD, Llinas M (2009) Host-parasite interactions revealed by Plasmodium falciparum metabolomics. Cell Host Microbe 5:191–199
Orhon D, Cokgor EU, Katipoglu T, Insel G, Karahan O (2010) Fate of 2,6-dihydroxybenzoic acid and its inhibitory impact on the biodegradation of peptone under aerobic conditions. Bioresour Technol 101:2665–2671
Preidis GA, Hotez PJ (2015) The newest “omics” metagenomics and metabolomics-enter the battle against the neglected tropical diseases. PLoS Negl Trop Dis 9:e0003382
Song C, Chiasson MA, Nursimulu N, Hung SS, Wasmuth J, Grigg ME, Parkinson J (2013) Metabolic reconstruction identifies strain‐specific regulation of virulence in Toxoplasma gondii. Mol Syst Biol 9:e708
Timanova A, Muller S, Marti T, Bankov I, Walter RD (1999) Ascaridia galli fatty acid‐binding protein, a member of the nematode polyprotein allergens family. Eur J Biochem 261:569–576
Traub RJ, Hobbs RP, Adams PJ, Behnke JM, Harris PD, Thompson RC (2007) A case of mistaken identity-reappraisal of the species of canid and felid hookworms (Ancylostoma) present in Australia and India. Parasitology 134:113–119
Truscott JE, Hollingsworth TD, Brooker SJ, Anderson RM (2014) Can chemotherapy alone eliminate the transmission of soil transmitted helminths. Parasit Vectors 7:e266
Tyagi R, Rosa BA, Lewis WG, Mitreva M (2015) Pan-phylum comparison of nematode metabolic potential. PLoS Negl Trop Dis 9:e0003788
Vranova V, Lojkova L, Rejsek K, Formanek P (2013) Significance of the natural occurrence of L-versus D-pipecolic acid: a review. Chirality 25:823–831
WHO (2012) Soil-transmitted helminthiases: eliminating soil-transmitted helminthiases as a public health problem in children: progress report 2001–2010 and strategic plan 2011–2020
WHO (2015) Investing to overcome the global impact of neglected tropical diseases: third WHO report on neglected diseases 2015. WHO Press, World Health Organization, Geneva
Willger SD, Grahl N, Cramer RA Jr (2009) Aspergillus fumigatus metabolism: clues to mechanisms of in vivo fungal growth and virulence. Med Mycol 47:S72–S79
Xia J, Mandal R, Sinelnikov IV, Broadhurst D, Wishart DS (2012) MetaboAnalyst 2.0—a comprehensive server for metabolomic data analysis. Nucleic Acids Res 40:W127–W133
Yap P, Du ZW, Wu FW, Jiang JY, Chen R, Zhou XN, Hattendorf J, Utzinger J, Steinmann P (2013) Rapid re-infection with soil-transmitted helminths after triple-dose albendazole treatment of school-aged children in Yunnan, People’s Republic of China. Am J Trop Med Hyg 89:23–31
Acknowledgments
The authors would like to appreciate financial support from the Water Corporation, WA and Land and Water, Commonwealth Scientific and Industry Research Organisation (CSIRO). Thanks to Dr. Simon Toze for his guidance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gyawali, P., Beale, D.J., Ahmed, W. et al. Determination of Ancylostoma caninum ova viability using metabolic profiling. Parasitol Res 115, 3485–3492 (2016). https://doi.org/10.1007/s00436-016-5112-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-016-5112-4